2. Beijing Allgens Medical Science and Technology Co., Ltd., Beijing 100176, China;
3. Department of Neurosurgery, Tsinghua University Yuquan Hospital, Beijing 100040, China
Objectives: In this case report, we describe the design, fabrication and clinical outcomes of a novel bioresorbable, mineralized collagen burr-hole plug for the reconstruction of craniotomy burr-holes.
Methods: Mineralized collagen burr-hole plugs were fabricated via a biomimetic mineralization process. The biomimetic mineralized collagen has a similar chemical composition and microstructure to natural bone tissue, thereby possessing good biocompatibility and osteoconductivity. The mineralized collagen burr-hole plugs were implanted into three patients, and clinical outcomes were evaluated at one-year follow-ups.
Results: All bone defects healed very well using the mineralized collagen burr-hole plugs, and there were no adverse reactions at the surgical sites.
Conclusions: The clinical outcomes indicated that the mineralized collagen was effective for reconstructing burr-holes in the skull after craniotomy.
Autologous bone,considered the "gold standard" bone graft material,can be used to reconstruct cranial burr‐holes. There are two primary methods for collecting autologous bone in a neurosurgical procedure. One is by harvesting cortical bone discs from the inner table of the craniotomy flap by trepanation[4, 5],and the other is by collecting bone dust during cranial trepanation to mix with biological glue to form a paste[6]. However,the amount of autologous bone is limited in patients,and the craniotomy flap becomes thinner after cortical bone discs are trephined. Furthermore,the collection of autologous bone further increases the complexity of the surgery.
A variety of synthetic materials have been widely applied in bone defect repair,such as titanium alloys,calcium phosphate bioceramics,polyethylene, polyetheretherketone (PEEK),polymethyl methacrylate (PMMA),and bioresorbable polyesters[7, 8, 9]. In recent decades,many of these biomaterials have also been used for skull repair,including to fill the burr‐holes. Polyethylene (PE) has been fabricated into a porous structure and then processed into specific shapes to cover the burr‐holes[3] as well as to fill the gap along the craniotomy flap[10]. A biocompatible hydroxyapatite (HA) has been fabricated into porous (low porosity of 15%-30%) or porous‐dense bi‐layer structures to fill and cover burr‐holes[1, 11, 12]. A biodegradable burrhole cover was also developed using bioresorbable polycaprolactone (PCL)[13]. Previous case studies have reported that such PCL implants gradually degraded with new bone regeneration at the burr‐hole site[13, 14]. However,all of these synthetic materials are different from natural bone tissues,and adverse reactions might be induced by using burr‐hole covers made of such materials[15].
A novel bioresorbable burr‐hole plug was designed and fabricated for the reconstruction of craniotomy burr‐holes using mineralized collagen. The mineralized collagen is a biomaterial prepared via an in vitro biomimetic process,which causes it to possess a chemical composition and microstructure similar to those of natural bone tissue. The mineralized collagen’s biocompatibility and promotion effects on bone regeneration were demonstrated in our previous studies and clinical outcomes[16, 17]. In this case report,we describe the clinical outcomes of three patients in whom the mineralized collagen burr‐hole plugs were implanted. The clinical outcomes were evaluated at the one‐year follow‐up.
2 Materials and methods 2.1 Design and fabrication of the mineralized collagen burr‐hole plug The design of the burr‐hole plug took into consideration the shape of the bone defect and operational convenience,so as to meet the requirement of craniotomy. The plug was a whole entity and contained two parts: a flat cap and a low cylinder. The cylinder’s diameter and height matched those of the burr‐hole, thus completely filling the bone defect on the skull. The cap provided a 3 mm brim that extended out from the cylinder,which controlled the implant depth of the plug. In order to fit the plug to the outline of the skull,the top surface of the cap and the bottom surface of the cylinder were designed to cambered surfaces (Figure 1).

|
| Figure 1 The mineralized collagen burr‐hole plug. |
The mineralized collagen burr‐hole plugs were produced in conformance with GMP specifications by Allgens Medical Science and Technology Co.,Ltd. (Beijing,China),and the products have been approved for marketing by the China Food and Drug Administration (CFDA,registration certificate number: 20143462075). The preparation process contained two primary procedures for biomimetic mineralization and integral molding. Biomimetic mineralization was performed as previously described by adding a solution containing Ca2+ and PO4 3- into an acidic collagen solution to form mineralized collagen deposition[ 18]. During this biomimetic mineralization process,the crystal nucleation and growth of hydroxyapatite were directed by collagen molecular templates,which is similar to the mineralization process of natural bone tissue. The deposition was centrifuged and concentrated to collect the slurry, which was then cast in a mold with a specific cavity and frozen to form the desired shape. The mineralized collagen burr‐hole plug fabrication process was completed by freeze‐drying and sterilization.
2.2 Surgical procedures and postoperative follow‐ups 2.2.1 Case 1: Bone defect repair after subarachnoid cyst removal at the left temporal region Case 1 was a 12‐year‐old male. Four,2.0 cm × 1.0 cm bone defects were produced after surgery for cyst removal at the left temporal region. The right two defects were implanted with the mineralized collagen burr‐hole plugs,and the left two defects were set as blank controls without any treatment. The clinical effects were evaluated by plain radiographs at the 1st, 3rd,and 10th months after surgery. 2.2.2 Case 2: Bone defect repair after evacuation of intracranial hematoma at the skull Case 2 was an 18‐year‐old male. A piece of cranial bone was removed to evacuate an intracranial hematoma, forming a 2.0 cm × 3.0 cm bone defect in the skull. A mineralized collagen burr‐hole plug was implanted into the bone defect,as shown in Figure 2. The clinical effects were evaluated by computed tomography (CT) at the 1st,6th,and 12th months after the surgery.
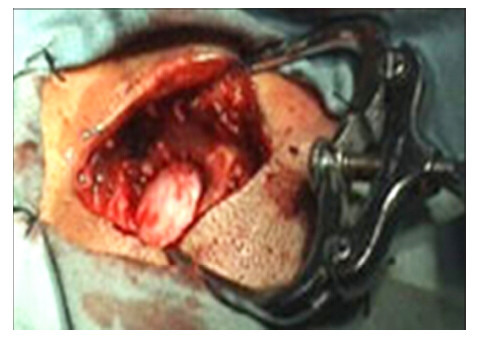
|
| Figure 2 Intraoperative scene as the burr‐hole plug was implanted. |
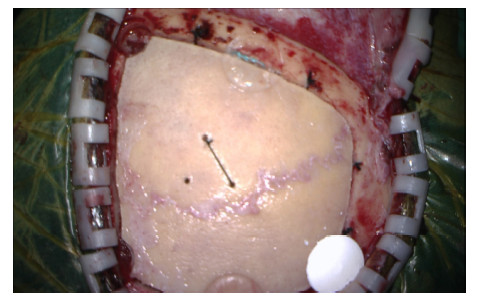
|
| Figure 3 The mineralized collagen burr‐hole plug was implanted into one bone defect after cranial bone flap reposition. |
This study was approved by the Committee of Medical Ethics of the hospital and performed with the informed consent for each patient and his family.
3 Results and discussion For each patient,there were no inflammatory responses, itching,or exudation at the surgical sites,and the wounds healed well. 3.1 Case 1 Figure 4 shows plain radiographs from the patient’s follow‐up. At the sites treated with mineralized collagen burr‐hole plugs (noted by triangles),bone mineral density gradually increased and approximated the host temporal bone. The interfaces between the implants and the host bone became fuzzy,indicating a remarkable osteogenesis effect. However,the bone mineral density of the blank control sites (noted by arrows) remained obviously lower than that of the surrounding normal bone tissue.
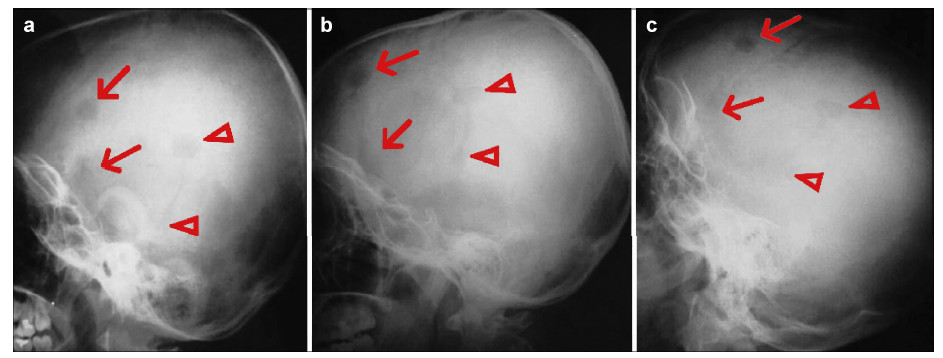
|
| Figure 4 Post‐operative plain radiographs from case 1 (a. 1‐month follow‐up; b. 3‐month follow‐up; c. 10‐month follow‐up). |
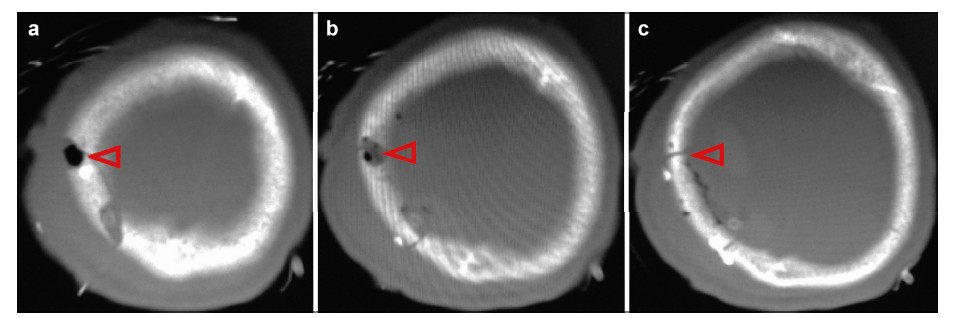
|
| Figure 5 Post‐operative CT scans of case 2 (a. 1‐month follow‐up; b. 6‐month follow‐up; c. 12‐month follow‐up). |
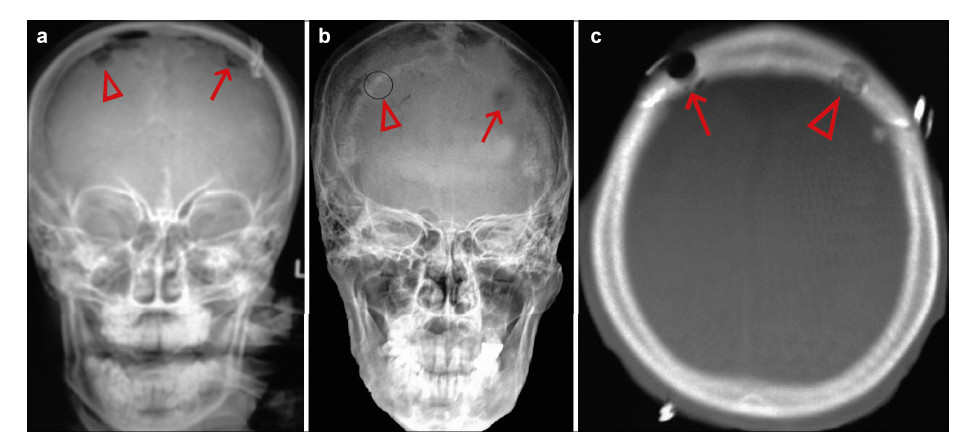
|
| Figure 6 Post‐operative plain radiographs and CT scan of case 3 (a. plain radiograph at 1‐month follow‐up; b. plain radiograph at the 6‐month follow‐up; c. CT scan at the 6‐month follow‐up). |
There are increasing clinical safety requirements for sealing the cranial bone defects and protecting organs and tissues inside the skull in order to reduce the risks of complications due to the holes,such as cerebrospinal fluid leakage and infection[3]. Cerebrospinal fluid leakage is a potential complication that can result from dural wounds during neurosurgical surgeries[19]. Cerebrospinal fluid leakage could result in intracranial hypotension,which is considered to be the main pathogenic mechanism underlying intracranial hemorrhage[20]. Craniotomy burr‐holes expose the dura mater and lead to the risk of physical damage or infection on the dura mater. Therefore,filling such burr‐holes through the use of adequate materials could provide a barrier between the unhealed scalp and the dura mater,so as to protect the dura mater from physical damage or infection.
On the other hand,cosmetic outcomes were generally attended by physicians and patients in cranioplasty surgeries[21, 22, 23]. A malformed appearance on the skull may produce psychosocial effects,leading to the need for cranioplasty,regardless of the defect size[24, 25]. Therefore,the cosmetic advantages of repairing burrholes or other small defects are very reasonable and necessary.
In light of the clinical requirement for the repair of burr‐holes,some synthetic materials have been used in an attempt to fabricate a related filler. Such materials have included metal[26],silicone rubber[15], PE[3],PCL[13, 14],HA[1, 12, 27],and so on. Synthetic biomaterials provide a sufficient source for cranial defect repair,including those as small as burr‐holes. PE burr‐hole filler is non‐bioresorbable and would permanently remain in the surgical sites. However, the biocompatibility of PE is unsatisfactory,and foreign body reactions to this material have been reported in many parts of the human body[28, 29, 30]. Bioceramics made of HA are considered to be biocompatible and have been widely used as bone substitutes[7]. However,the brittleness of the HA increases the risks when it is used for cranioplasty[30, 31]. The immunotoxicity of silicone rubber,which is also a commonly used biomaterial,is still being discussed[32, 33]. In a clinical application,a silicone rubber burr‐hole cover induced a serious foreign body reaction in which a seroma was formed,and the implant had to be removed surgically[15]. PCL is a bioresorbable polymer that has been used for bone substitution, drug delivery,suture,and so on[34, 35]. Although PCL is biodegradable,its degradation rate is much slower than the rate of bone regeneration[36]. Thus,the slowbiodegrading PCL hardly provides sufficient space for the formation of new bone,and the osteoconductivity is therefore limited.
There are two commonly used methods for utilizing autologous bone for filling burr‐holes in craniotomy. One method is drilling cortical bone discs from the inside of the craniotomy flap[5],and the other is collecting bone dust during cranial trepanation[6]. There have been comparative studies of the two methods. Cortical bone discs have a lower resorption degree and showed better bone repair results and better cosmetic outcomes compared to those for the bone dust[37]. Similar to those orthopedic surgeries employing autologous bone,the amount of autologous bone is very limited for patients[38],and the craniotomy flap becomes thinner after cortical bone discs are trephined. Moreover,harvesting autologous bone further increases the complexity of the surgery.
Mineralized collagen is a biomimetic biomaterial with the same chemical composition and microstructure to those of the natural bone matrix. Within the mineralized collagen,HA nano‐crystals are arranged in an orderly manner with the collagen fibrils,and the crystallographic c‐axis of the HA is parallel to the longitudinal direction of the collagen fibrils[39]. Some medical device products made of mineralized collagen have been commercialized. In this case report, due to the high similarity between the mineralized collagen and the natural bone matrix,the mineralized collagen was beneficial for the adhesion,growth, and proliferation of the osteocytes[40, 41]. Mineralized collagen is bioresorbable and is gradually replaced by new bone tissue via a creep substitution process at the implantation sites,thus inducing the regeneration of the bone defects[18]. Previous clinical outcomes demonstrated that biomimetic mineralized collagen promoted new bone formation at the bone defect sites at various sites of human bones[42, 43, 44]. The outcomes in this case report indicate that mineralized collagen plug is effective for the reconstruction of burr‐holes at the skull after craniotomy and improved the cosmetic outcomes of the patients.
4 Conclusions A novel bioresorbable mineralized collagen burr‐hole plug was designed and fabricated for the reconstruction of craniotomy burr‐holes. The mineralized collagen burr‐hole plugs were implanted into three patients, and clinical outcomes were evaluated at one‐year follow‐ups. All the bone defects healed very well with the mineralized collagen burr‐hole plugs,and there were no inflammatory responses,itching,or exudation at the surgical sites. The clinical outcomes indicate that mineralized collagen is effective for the reconstruction of burr‐holes in the skull after craniotomy.This work was supported in part by the National Basic Research Program (973 Program) of China (No. 2011CB606205),the "Twelfth Five‐Year" National Science and Technology Support Program (No. 2012BAI17B02) funded by the Ministry of Science and Technology of China,and the National Natural Science Fund funded by the National Natural Science Foundation of China (Nos. 21371106 and 51402167).
List of symbols| China Food and Drug Administration | CFDA |
| hydroxyapatite | HA |
| polyethylene | PE |
| polyetheretherketone | PEEK |
| polymethyl methacrylate | PMMA |
| polycaprolactone | PCL |
| [1] | Easwer HV, Rajeev A, Varma HK, Vijayan S, Bhattacharya RN. Cosmetic and radiological outcome following the use of synthetic hydroxyapatite porous-dense bilayer burr-hole buttons. Acta Neurochir (Wien) 2007, 149(5): 481-486. |
| Click to display the text | |
| [2] | Dujovny M, Dujovny N, Viñas F, Park HK, Lopez F. Burr hole cover for ventriculoperitoneal shunts and ventriculostomy: technical note. Neurol Res 2002, 24(5): 483-484. |
| Click to display the text | |
| [3] | Dujovny M, Aviles A, Cuevas P. Bone-like polyethelyne burr-hole cover. Neurol Res 2005, 27(3): 333-334. |
| Click to display the text | |
| [4] | Boström S, Kourtopoulos H, Nilsson I. Reconstruction of craniotomy burr-holes with autologous bone blugs made by a new hole-saw. Acta Neurochir (Wien) 1990, 105(3-4): 132-134. |
| Click to display the text | |
| [5] | Cokluk C, Senel A, Iyigün O, Aydin K, Rakunt C, Celik F. Reconstruction of burr hole by using autologous buttonshaped graft harvested from inner table of craniotomy flap: Technique and clinical result. Minim Invasive Neurosurg 2003, 46(6): 372-373. |
| Click to display the text | |
| [6] | Matsumoto K, Kohmura E, Kato A, Hayakawa T. Restoration of small bone defects at craniotomy using autologous bone dust and fibrin glue. Surg Neurol 1998, 50(4): 344-346. |
| Click to display the text | |
| [7] | Dorozhkin SV. Bioceramics of calcium orthophosphates. Biomaterials 2010, 31(7): 1465-1485. |
| Click to display the text | |
| [8] | Guo BL, Lei B, Li P, Ma PX. Functionalized scaffolds to enhance tissue regeneration. Regen Biomater 2015, 2(1): 47-57. |
| Click to display the text | |
| [9] | Hammouche S, Hammouche D, McNicholas M. Biodegradable bone regeneration synthetic scaffolds: In tissue engineering. Curr Stem Cell Res Ther 2012, 7(2): 134-142. |
| Click to display the text | |
| [10] | Goh DH, Kim GJ, Park J. Medporcraniotomy gap wedge designed to fill small bone defects along cranial bone flap. J Korean Neurosurg Soc 2009, 46(3): 195-198. |
| Click to display the text | |
| [11] | Koyama J, Hongo K, Iwashita T, Kobayashi S. A newly designed key-hole button. J Neurosurg 2000, 93(3): 506-508. |
| Click to display the text | |
| [12] | Kashimura H, Ogasawara K, Kubo Y, Yoshida K, Sugawara A, Ogawa A. A newly designed hydroxyapatite ceramic burrhole button. Vasc Health Risk Manag 2010, 6: 105-108. |
| Click to display the text | |
| [13] | Schantz JT, Lim TC, Ning C, Teoh SH, Tan KC, Wang SC, Hutmacher DW. Cranioplasty after trephination using a novel biodegradable burr hole cover: Technical case report. Neurosurgery 2006, 58(1): ONS-E176. |
| Click to display the text | |
| [14] | Low SW, Ng YJ, Yeo TT, Chou N. Use of Osteoplug polycaprolactone implants as novel burr-hole covers. Singapore Med J 2009, 50(8): 777-780. |
| Click to display the text | |
| [15] | Winkler PA, Herzog C, Weiler C, Krishnan KG. Foreignbody reaction to silastic burr-hole covers with seroma formation: case report and review of the literature. Pathol Res Pract 2000, 196(1): 61-66. |
| Click to display the text | |
| [16] | Liao SS, Cui FZ. In vitro and in vivo degradation of mineralized collagen-based composite scaffold: nanohydroxyapatite/collagen/poly(L-lactide). Tissue Eng 2004, 10(1-2): 73-80. |
| Click to display the text | |
| [17] | Liao SS, Guan K, Cui FZ, Shi SS, Sun TS. Lumbar spinal fusion with a mineralized collagen matrix and rhBMP-2 in a rabbit model. Spine (Phila Pa 1976) 2003, 28(17): 1954-1960. |
| Click to display the text | |
| [18] | Liao SS, Cui FZ, Zhang W, Feng QL. Hierarchically biomimetic bone scaffold materials: Nano-HA/collagen/PLA composite. J Biomed Mater Res B Appl Biomater 2004, 69(2): 158-165. |
| Click to display the text | |
| [19] | Huh J. Burr hole drainage: could be another treatment option for cerebrospinal fluid leakage after unidentified dural tear during spinal surgery? J Korean Neurosurg Soc 2013, 53(1): 59-61. |
| Click to display the text | |
| [20] | Sciubba DM, Kretzer RM, Wang PP. Acute intracranial subdural hematoma following a lumbar CSF leak caused by spine surgery. Spine (Phila Pa 1976) 2005, 30(24): E730- E732. |
| Click to display the text | |
| [21] | Plum AW, Tatum SA. A comparison between autograft alone, bone cement, and demineralized bone matrix in cranioplasty. Laryngoscope 2015, 125(6): 1322-1327. |
| Click to display the text | |
| [22] | O'Reilly EB, Barnett S, Madden C, Welch B, Mickey B, Rozen S. Computed-tomography modeled polyether ether ketone (PEEK) implants in revision cranioplasty. J Plast Reconstr Aesthet Surg 2015, 68(3): 329-338. |
| Click to display the text | |
| [23] | Sorour M, Caton WL, Couldwell WT. Technique for methyl methacrylate cranioplasty to optimize cosmetic outcome. Acta Neurochir (Wien) 2014, 156(1): 207-209. |
| Click to display the text | |
| [24] | Dujovny M, Aviles A, Agner C, Fernandez P, Charbel FT. Cranioplasty: cosmetic or therapeutic? Surg Neurol 1997, 47(3): 238-241. |
| Click to display the text | |
| [25] | Grantham EC, Landis HP. Cranioplasty and the posttraumatic syndrome. J Neurosurg 1948, 5(1): 19-22. |
| Click to display the text | |
| [26] | Ohata K, Haque M, Tsuruno T, Morino M, Soares SBJ, Hakuba A. Craniotomy repair with titanium miniplates. J Clin Neurosci 1998, 5(1): 81-86. |
| Click to display the text | |
| [27] | Yamashima T. Reconstruction of surgical skull defects with hydroxylapatite ceramic buttons and granules. Acta Neurochir (Wien) 1988, 90(3-4): 157-162. |
| Click to display the text | |
| [28] | Timoney PJ, Clark JD, Frederick PA, Krakauer M, Compton C, Horbinski C, Spkol J, Nunery WR. Foreign body granuloma following orbital reconstruction with porous polyethylene. Ophthal Plast Reconstr Surg 2014, DOI: 10.1097/IOP.0000000000000328. |
| Click to display the text | |
| [29] | Spaans AJ, van Heeswijk EJM, Arnold DE, Beumer A. Foreign body reaction associated with polyethylene mesh interposition used for treatment of trapeziometacarpal osteoarthritis: Report of 8 cases. J Hand Surg 2014, 39(10): 2016-2019. |
| Click to display the text | |
| [30] | Gosau M, Draenert FG, Ihrler S. Facial augmentation with porous polyethylene (Medpor)—Histological evidence of intense foreign body reaction. J Biomed Mater Res B Appl Biomater 2008, 87(1): 83-87. |
| Click to display the text | |
| [31] | Ducic Y. Titanium mesh and hydroxyapatite cement cranioplasty: a report of 20 cases. J Oral Maxillofac Surg 2002, 60(3): 272-276. |
| Click to display the text | |
| [32] | Heggers JP, Kossovsky N, Parsons RW, Robson MC, Pelley RP, Raine TJ. Biocompatibility of silicone implants. Ann Plast Surg 1983, 11(1): 38-45. |
| Click to display the text | |
| [33] | VandeVord PJ, Gupta N, Wilson RB, Vinuya RZ, Schaefer CJ, Canady AI, Wooley PH. Immune reactions associated with silicone-based ventriculo-peritoneal shunt malfunctions in children. Biomaterials 2004, 25(17): 3853-3860. |
| Click to display the text | |
| [34] | Berner A, Woodruff MA, Lam CXF, Arafat MT, Saifzadeh S, Steck R, Ren J, Nerlich M, Ekaputra AK, Gibson I, et al. Effects of scaffold architecture on cranial bone healing. Int J Oral Maxillofac Surg 2014, 43(4): 506-513. |
| Click to display the text | |
| [35] | Coombes AGA, Rizzi SC, Williamson M, Barralet JE, Downes S, Wallace WA. Precipitation casting of polycaprolactone for applications in tissue engineering and drug delivery. Biomaterials 2004, 25(2): 315-325. |
| Click to display the text | |
| [36] | Woodruff MA, Hutmacher DW. The return of a forgotten polymer-Polycaprolactone in the 21st century. Prog Polym Sci 2010, 35(10): 1217-1256. |
| Click to display the text | |
| [37] | Worm PV, Ferreira NP, Faria MB, Ferreira MP, Kraemer JL, Collares MVM. Comparative study between cortical bone graft versus bone dust for reconstruction of cranial burr holes. Surg Neurol Int 2010, 1: 91. |
| Click to display the text | |
| [38] | Betz RR. Limitations of autograft and allograft: new synthetic solutions. Orthopedics 2002, 25(5 Suppl): S561-S570. |
| Click to display the text | |
| [39] | Cui FZ, Li Y, Ge J. Self-assembly of mineralized collagen composites. Mater Sci Engineer: R: Rep 2007, 57(1-6): 1-27. |
| Click to display the text | |
| [40] | Du C, Cui FZ, Zhu XD, de Groot K. Three-dimensional nano-HAp/collagen matrix loading with osteogenic cells in organ culture. J Biomed Mater Res 1999, 44(4): 407-415. |
| Click to display the text | |
| [41] | Xia Y, Peng SS, Xie LZ, Lian XJ, Zhang XJ, Cui H, Song TX, Zhang FM, Gu N, Cui FZ. A novel combination of nanoscaffolds with micro-scaffolds to mimic extracellularmatrices improve osteogenesis. J Biomater Appl 2013, 29(1): 59-71. |
| Click to display the text | |
| [42] | Yu X, Xu L, Cui FZ, Qu Y, Lian XJ, Wang XM, Wang Y. Clinical evaluation of mineralized collagen as a bone graft substitute for anterior cervical intersomatic fusion. J Biomater Tissue Engineer 2012, 2(2): 170-176. |
| Click to display the text | |
| [43] | Lian K, Lu H, Guo XD, Cui FZ, Qiu ZY, Xu SY. The mineralized collagen for the reconstruction of intra-articular calcaneal fractures with trabecular defects. Biomatter 2013, 3(4): e272850. |
| Click to display the text | |
| [44] | Kou JM, Fu TY, Jia XJ, Hou JW, Gao C, Ma YZ, Qiu ZY, Cui FZ. Clinical observations on repair of non-infected bone nonunion by using mineralized collagen graft. J Biomater Tissue Engineer 2014, 4(12): 1107-1112. |
| Click to display the text |



