2. Department of Skalozyb-Biotechnology, Cell Therapy Center EmCell, Kiev 04210, Ukraine;
3. Department of Experimental Medicine, Second University of Naples via S. Maria di Costantinopoli 16, Naples 80138, Italy
Objectives: To study the effect of fetal stem cell (FSC) therapy on Grade Ⅰ and Ⅱ respiratory failure in patients with amyotrophic lateral sclerosis (ALS) and muscular dystrophy (MD).
Methods: A comparative study was conducted on 41 patients with Grade Ⅰ or Ⅱ respiratory failure (RF) resulting from ALS or MD. The patients were divided into 4 groups according to the underlying disease and the degree of RF. Patients underwent combined treatment, including the experimental application of FSC therapy, and were examined before FSC treatment, and 6 months and 12 months after treatment.
Results: FSC treatment improved both subjective and objective breathing parameters as early as 6 months post-treatment. A significant increase in the forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1) was reported by all patients with Grade Ⅰ RF linked to ALS and MD compared to baseline. Patient respiratory improvement was maintained over the next 6 months. Grade Ⅱ RF patients with MD reported a significant improvement in FVC 12 months after treatment.
Conclusions: Evidence for respiratory improvement was observed as early as 6 months in all patients after combined treatment including FSC therapy, and this was maintained for a further 6 months after therapy. In MD patients with Grade Ⅱ RF, treatment resulted in a significant FVC and FEV1 increase within 6 months and downgrading to Grade Ⅰ RF within a year after FSC treatment.
RF is defined as inadequate gas exchange by the respiratory system,or its maintenance only through excessive work,often resulting in apnea. RF is often found in patients with ALS and muscular dystrophy (MD),and is the main cause of death in ALS patients[7].
MD represents a group of chronic hereditary diseases that can affect skeletal muscles leading to progressive weakness and muscular degeneration. This degeneration can spread to the upper body, gradually involving all of the main muscle groups, including those required for respiration. This disease characteristically causes damage to the voluntary and smooth muscles and myocardium,ultimately leading to respiratory excursion,and a decompensation state. In MD,RF is caused by muscle tissue damage resulting from a mutation in the dystrophin gene[1, 8, 9]. Muscle fibers become necrotic and are substituted by fatty and connective tissue,which results in impaired respiration[7]. RF lowers the overall life expectancy in MD patients irrespective of its type,and its progression results in cardiopulmonary decompensation in most cases[9, 10].
Respiratory disturbances are fatal in both ALS and MD. Although respiratory exercises and oxygen therapy are recommended for these patients,an effective method to prevent ALS and MD progression and the inevitable development of RF remains to be found. Suggested alternative therapeutic methods include fetal stem cell (FSC) transplant,and several studies have explored this possibility.
Specialists at the A. P. Romodanov Institute for Neurosurgery (Ukrainian National Academy of Medical Science) developed the stem cell technology used for the treatment of Duchenne MD. The biological formulation consisted of stem cells and myoblasts that were injected directly into the muscle tissue, preventing myocyte destruction. This in turn led to disease remission[11]. It is assumed that stem cells contain specific muscle markers and are capable of dystrophin expression[12, 13, 14]. Other researchers have used both intravenous and intramuscular modes of FSC administration[15]. Hematopoietic and mesenchymal FSCs can reach "niches" throughout the circulation and then become satellite cells during the course of development. These in turn usually transform into muscle fibers. The extent to which they migrate depends on the degree of muscle impairment[16].
Fetal myoblasts have several advantages over embryonic ones[17, 18]. They have a triangular shape and can divide several times in a non‐differentiated state, and then differentiate into multinuclear myocytes in response to growth factors[17, 18]. Fetal neuronal stem cells have the potential to differentiate into neural cells inside the impaired nervous system[11, 16, 20]. FSCs can differentiate into virtually any type of neuron (as well as non‐neuronal cells) when administered to patients with neurological diseases. This has been proven in multiple studies with labeled nuclei[1]. However,the engraftment rate of the transplanted cells is low (5%-20%),and is influenced by the method used to isolate stem cells from the fetal brain[19, 20]. Another possible therapeutic role for FSCs is in preventing mitochondrial dysfunction and hence nerve cell degeneration[2]. It is also assumed that FSCs contain specific muscle markers and can express the dystrophin gene[12, 13, 14].
Based on these findings,this study was designed to evaluate changes in spirometry parameters after FSC therapy for Grade Ⅰ and Ⅱ RF in ALS and MD patients.
2 Materials and methods 2.1 Subjects The study involved 41 patients with ALS or MD complicated by RF. The cohort consisted of 19 patients (46.3%) with ALS aged 25-60 years (mean age,39.4 ± 2.57 years),including 13 men (68.4%) and 6 women (31.6%),and 22 patients (53.7%) with MD aged 15-60 years (mean age,36.3 ± 2.46 years),including 15 men (68.2%) and 7 women (31.8%).The 41 patients were divided into 4 groups according to their underlying disease and degree of RF:
(1) Group Ⅰ: ALS patients with Grade Ⅰ RF - 9 patients (22.0%).
(2) Group Ⅱ: ALS patients with Grade Ⅱ RF - 10 patients (24.4%).
(3) Group Ⅲ: MD patients with Grade Ⅰ RF - 11 patients (26.8%).
(4) Group Ⅳ: MD patients with Grade Ⅱ RF - 11 patients (26.8%).
MD was diagnosed on the basis of clinical findings, past history,electroneuromyography (ENMG) results that were suggestive of MD,laboratory test findings (abnormal alanine‐aminotransferase (ALT),aspartateaminotransferase (AST),creatine phosphokinase (CPK), and lactate dehydrogenase (LDH) activity),genetic test results,and histological findings. ALS was confirmed on the basis of clinical and ENMG findings. The cervical form was diagnosed in 6 patients (31.6%),the cervicothoracic form in 5 patients (26.3%),and the bulbar form in 8 patients (42.1%). RF was diagnosed according to clinical symptoms (apnea,acrocyanosis,and a heart rate frequently exceeding 90 bpm),and measurements such as blood oxygen saturation (Grade Ⅰ RF,93-98%; Grade Ⅱ RF,86%-92%; Grade Ⅲ RF,< 75%),forced vital capacity (FVC) (Grade Ⅰ RF,70%-85%; Grade Ⅱ RF, 50%-70%; Grade Ⅲ RF,< 50%),and forced expiratory volume in 1 second (FEV1) (Grade Ⅰ RF,74%-55%; Grade Ⅱ RF,54%-35%; Grade Ⅲ RF,< 35%).
Lung ventilation capacity was measured using a BTL‐08 Spiro spirograph (2010) in accordance with the approved standards[21],and arterial oxyhemoglobin (SpO2) was controlled using a pulse Oxymeter YX300 Armed (2011).
2.2 Stem cells We used suspensions containing FSCs harvested from 5-9‐week‐old human fetuses. One suspension was made using stem cells from the fetal liver,while the other consisted of stem cells from fetal brain. The detailed protocol for making these suspensions has been previously published[22]. Briefly,fetal material was harvested in the surgery room,in compliance with all aseptic and antiseptic requirements. After obtaining written informed consent all tissues were collected from the donor,mainly from the liver and brain tissue of 5-9‐week‐old human fetuses aborted for family planning reasons and found to have no developmental abnormalities or hemic infections. The tissue was then placed into sterile transport medium consisting of Hank’s Balanced Salt Solution and antibiotics (penicillin-streptomycin,100 U-100 mg/ml) (Sigma Chemical Company,St. Louis,MO,USA). The tissues were aseptically separated and homogenized in Hank’s solution. The stem cell suspension was then filtered. Dimethyl sulfoxide 10% diluted in DMEM was used as a cryoprotectant. Cryopreservation of cell suspensions was performed in a controlled‐rate freezer chamber pursuant to the selected program.In order to ensure safety,both the donors and the ready‐made stem cells in suspension from fetal liver were tested for the presence of bacteria,fungi,parasites and viruses such as human immunodeficiency virus 1 and 2,hepatitis B virus,hepatitis C virus,cytomegalovirus, herpes simplex virus 1 and 2,Epstein-Barr virus,rubella,syphilis (Treponema pallidum),toxoplasmosis (Toxoplasma gondii),Mycoplasma genitalium bacterium,Ureaplasma urealyticum,Chlamydia trachomatis,and Ureaplasma parvum.
Suspensions containing stem cells from fetal liver and brain were stored in liquid nitrogen at -196 °C in a properly arranged cryobank. Transplantation of the suspension containing cryopreserved fetal stem cells was preceded by an infusion of diphenylhydramine 10 mg and prednisone 30 mg on day 1 and a specially prepared solution on day 2. On day 1,stem cells from fetal liver were transplanted. A suspension containing cryopreserved stem cells was administered via an intravenous drip‐feed in a volume of 1.75 ± 0.51 mL with a nucleated cell count of 58.74 × 106/mL per transplantation. Fetal brain stem cells were subcutaneously administered on day 2 in a volume of 2.12 ± 0.49 mL with a nucleated cell count of 7.9 × 106/mL per transplantation. The number of CD34+ cells was determined using flow cytofluorometry (Becton Dickinson,Franklin Lakes,NJ,USA) with fluorescently tagged antibodies (Santa Cruz Biotechnology,Dallas, TX,USA).
All patients also underwent routine therapy including muscle supporting medicines (L‐carnitine,vitamins, lipoic acid,amino acids,vasoactive medications,and biostimulators),and antioxidants in order to reduce muscle cell membrane damage.
Neurological evaluation,laboratory tests (ALT,AST, CPK,and LDH levels) and instrumental examinations were performed before FSCT,and 6 and 12 months after treatment. Before the FSCT,all patients or their legal representatives signed the informed consent. This study was approved by the local ethics committee and included in the public registry of the Ministry of Education and Science of Ukraine,Ukrainian Institute of Scientific,Technical and Economic Information. The study registration number is 0113U000957. Approval was obtained by the local ethics committee of Kyiv City Clinical Hospital for Accident and Emergency Care.
2.3 Statistical analysis Average values and their standard deviations were calculated,and significant differences were determined using Statistika 6.0 Student’s T‐criteria. Differences were regarded as statistically significant if P < 0.05. 3 Results Objective parameters of respiration (FVC,FEV1,and SpO2) in ALS patients are presented in Table 1. After FSCT,the SpO2 in ALS patients with Grade Ⅰ RF (Group Ⅰ) had increased by 1.8 ppm (P < 0.05) 6 months after treatment and by 4.0 ppm (P < 0.05) one year after treatment in comparison with baseline. The FVC had increased by 17.3 ppm (P < 0.05) and the FEV1 had increased by 18.1 ppm (P < 0.05) in the same group 6 months after FSCT. An increase in respiratory volume was also apparent in Group Ⅰ 12 months after treatment; the FVC was elevated by 18.5 ppm (P < 0.05) and the FEV1 by 19.5 ppm (P < 0.05). 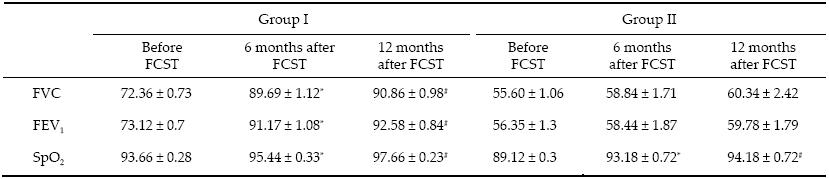 |
| Note: *P < 0.05, statistically significant difference in values prior to therapy and 6 months after FSCT; #P < 0.05, statistically significant difference in values prior to treatment and 12 months after FSCT; ALS-amyotrophic lateral sclerosis; FCST-fetal stem cell treatment; FVC-forced vital capacity; FEV1-forced expiratory volume in 1 second; SpO2-arterial oxyhemoglobin saturation. |
Thus,in combined treatment of ALS patients with Grade Ⅰ RF,FSCs produced a positive clinical effect as demonstrated by an increase in respiratory volume. Furthermore,respiratory improvements were evident within 6 months of the start of treatment and were maintained one year after the treatment.
In ALS patients with Grade Ⅱ RF (Group Ⅱ),SpO2 increased by 4.2 ppm (P < 0.05) over 6 months and by 5.2 ppm (P < 0.05) one year after treatment. The baseline respiratory volumes were significantly lower in these patients owing to the rapid progression of the disease, although despite this the FVC increased by 3.2 ppm (P > 0.05) and the FEV1 by 2.1 ppm (P > 0.05) 6 months after FSCT,and within 12 months the FCV had increased by 4.7 ppm (P > 0.05) and the FEV1 by 3.43 ppm (P > 0.05) in comparison with the baseline. Thus,FSCs tend to increase lung ventilation capacity and preserve respiratory capacity in these patients.
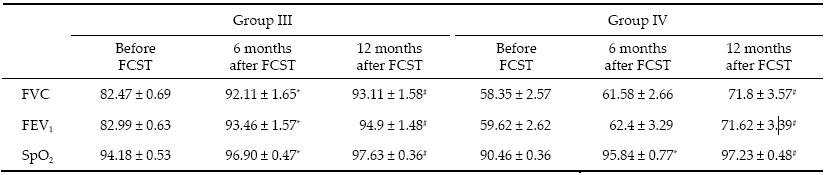
Spirometry findings and oxygen saturation levels of the MD patients are presented in Table 2. The most obvious effect of stem cell therapy was seen in MD patients with Grade Ⅰ RF (Group Ⅲ). FCV increased by 9.6 ppm (P < 0.05) and FEV1 by 10.47 ppm (P < 0.05) over the 6 months following FSCT,and after 12 months they had increased by 10.64 ppm (P < 0.05) and 11.9 ppm (P < 0.05),respectively. Oxygen saturation levels within this group had also increased by 2.7 ppm (P < 0.05) over the 6 months following therapy,and by 3.4 ppm (P < 0.05) over 12 months. Thus,in the combined treatment of Grade Ⅰ RF in MD,FSCs significantly improved respiration with respect to FVC and FEV1. In these patients,normalization of breathing was possible within 6 months after the start of treatment and was maintained through the subsequent 6 months.
 |
| Note: *P < 0.05, statistically significant difference between values prior to treatment and 6 months after FSCT; #P < 0.05, statistically significant difference between values prior to therapy and 12 months after FSCT; FCST-fetal stem cell treatment; FVC-forced vital capacity; FEV1-forced expiratory volume in 1 second; MD-muscular dystrophy; SpO2-arterial oxyhemoglobin saturation. |
Improvements were also apparent in MD patients (Group Ⅳ) with Grade Ⅱ RF. Six months after FSCT, the FCV had increased by 3.2 ppm (P > 0.05) and FEV1 by 2.8 ppm (P > 0.05). Twelve months after therapy the increase in respiratory capacity was significantly higher than the baseline: FEV increased by 13.5 ppm (P < 0.05) and FEV1 by 12 ppm (P < 0.05). The post‐FSCT SpO2 increased by 5.4 ppm (P < 0.05) over 6 months and by 6.8 ppm (P < 0.05) over 12 months. Thus,after FSCT, MD patients with Grade Ⅱ RF generally had improved respiration through a significant increase in respiratory capacity,which allowed the patient to be downgraded to Grade Ⅰ RF.
4 Discussion The experimental application of FSCs continues to be debated,and is not supported in the USA and some European countries. However,in many countries, including Ukraine,this method of therapy can be used experimentally provided that consent for abortion and donation of the fetal material is obtained based on legal,moral,and ethical principles. In view of very promising clinical results using FSCs,which are able to differentiate into specialized functional cells,including various muscular tissues,their routine use may now be justifiable.In our study,the improvements in the patient’s condition,including their respiratory function,were probably due to the distribution of multipotent stem cells throughout the body,allowing them to repair defects both locally and systemically. This is seen in MD patients in general,and Duchenne MD in particular[14]. Moreover,stem cells could promote muscle regeneration through their properties of self‐renewal and differentiation,as demonstrated in several pre‐clinical models[23]. The rationale for the use of FSCs derived from liver and brain tissue, administered on different days,is based on our previous experience[24]. In Duchenne MD,FSCs could exert a beneficial effect based not only on their ability to differentiate into functional cells,but also through a trophic paracrine effect,as they can secrete diffusible growth factors. It is also possible that transplanted FSCs could restore dystrophin synthesis.
In addition to improvements in breathing capacity, FSCT helped improve the patient’s physical and emotional state,with improved self‐confidence and social functioning. Consequently,patients receiving FSC treatment considered that their overall quality of life had greatly improved.
5 Conclusions In combined treatment of Grade Ⅰ RF in ALS patients FSCT resulted in respiratory improvement as early as 6 months after treatment,and this was maintained over the next 6 months. Although there was no significant respiratory capacity improvement in ALS patients with Grade Ⅱ RF,their ventilation capacity tended to increase.In combined treatment of MD patients with Grade Ⅰ RF FSCT resulted in respiratory function normalization within 6 months. In MD patients with Grade Ⅱ RF,treatment resulted in significant FVC and FEV1 increases within 6 months and downgrading to Grade Ⅰ RF within a year.
Conflict of interests The paper is intended to be an original paper; all authors of the manuscript,except DS,are members of Cell Therapy Center EmCell,Kyiv,Ukraine. The authors have approved the manuscript and agree to its submission. There are no matters that constitute a conflict of interest among the authors who contributed to this manuscript.| [1] | Boulis NM, Federici T, Glass JD, Lunn JS, Sakowski SA, Feldman EL. Translational stem cell therapy for amyotrophic lateral sclerosis. Nat Rev Neurol 2011, 8(3): 172-176. |
| Click to display the text | |
| [2] | Clelland CD, Choi M, Romberg C, Clemenson GDJ, Fragniere A, Tyers P, Jessberger S, Saksida LM, Barker RA, Gage FH. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 2009, 325(5937): 210-213. |
| Click to display the text | |
| [3] | Jessberger S, Nakashima K, Clemenson GDJ, Mejia E, Mathews E, Ure K, Ogawa S, Sinton CM, Gage FH, Hsieh J. Epigenetic modulation of seizure-induced neurogenesis and cognitive decline. J Neurosci 2007, 27(22): 5967-5975. |
| Click to display the text | |
| [4] | Johnson L, Miller JW, Gkazi AS, Vance C, Topp SD, Newhouse SJ, Al-Chalabi A, Smith BN, Shaw CE. Screening for OPTN mutations in a cohort of British amyotrophic lateral sclerosis patients. Neurobiol Aging 2012, 33(12): 2948. |
| Click to display the text | |
| [5] | Perlson E, Jeong GB, Ross JL, Dixit R, Wallace KE, Kalb RG, Holzbaur ELF. A switch in retrograde signaling from survival to stress in rapid-onset neurodegeneration. J Neurosci 2009, 29(31): 9903-9917. |
| Click to display the text | |
| [6] | Péault B, Rudnicki M, Torrente Y, Cossu G, Tremblay J, Partridge T, Gussoni E, Kunkel LM, Huard J. Stem and progenitor cells in skeletal muscle development, maintenance, and therapy. Mol Therapy 2007, 15(5): 867-877. |
| Click to display the text | |
| [7] | Evtushenko SK, Shaymurzin MR, Evtushenko OS. New modern technology in therapy of neuromuscular diseases for slowing its progression. Int Neurology J 2009, 4(26): 9-18. (in Russian) |
| [8] | Ilancheran S, Michalska A, Peh G, Wallace EM, Pera M, Manuelpillai U. Stem cells derived from human fetal membranes display multilineage differentiation potential. Biol Reprod 2007, 77(3): 577-588. |
| Click to display the text | |
| [9] | Kang PB, Lidov HG, White AJ, Mitchell M, Balasubramanian A, Estrella E, Bennett RR, Darras BT, Shapiro FD, Bambach BJ, Kurtzberg J, Gussoni E, Kunkel LM. Inefficient dystrophin expression after cord blood transplantation in Duchenne muscular dystrophy. Muscle Nerve 2010, 41(6): 746-750. |
| Click to display the text | |
| [10] | Lawlor MW, Alexander MS, Viola MG, Meng H, Joubert R, Gupta V, Motohashi N, Manfready RA, Hsu CP, Huang P, Bu-Bello A, Kunkel LM, Beggs AH, Gussoni E. Myotubularin-deficient myoblasts display increased apoptosis, delayed proliferation, and poor cell engraftment. Am J Pathol 2012, 181(3): 961-968. |
| Click to display the text | |
| [11] | Lapan AD, Rozkalne A, Gussoni E. Human fetal skeletal muscle contains a myogenic side population that expresses the melanoma cell-adhesion molecule. Hum Mol Genet 2012, 21(16): 3668-3680. |
| Click to display the text | |
| [12] | Cossu G, Biressi S. Satellite cells, myoblasts and other occasional myogenic progenitors: Possible origin, phenotypic features and role in muscle regeneration. Semin Cell Dev Biol 2007, 16(4-5): 623-631. |
| Click to display the text | |
| [13] | Johnson BV, Shindo N, Rathjen PD, Rathjen J, Keough RA. Understanding pluripotency-How embryonic stem cells keep their options open. Mol Hum Reprod 2008, 14(9): 513-520. |
| Click to display the text | |
| [14] | Knobloch M, Jessberger S. Perspectives on adult neurogenesis. Eur J Nurosci 2011, 33(6): 1013-1017. |
| Click to display the text | |
| [15] | Acevedo-Arozena A, Kalmar B, Essa S, Ricketts T, Joyce P, Kent R, Rowe C, Parker A, Gray A, Hafezparast M, Thorpe JR, Greensmith L, Fisber EM. A comprehensive assessment of the SOD1SOD1G93A low-copy transgenic mouse, which models human amyotrophic lateral sclerosis. Dis Model Mech 2011, 4(5): 686-700. |
| Click to display the text | |
| [16] | Kukharchuk AL, Radchenko VV. Embryonic progenitor cells, immunological tolerance and aging. Transplantology 2008, 10(1): 51-54. |
| [17] | Kim J, Chu JL, Shen XH, Wang JL, Orkin SH. An extended transcriptional network for pluripotency of embryonic stem cells. Cell 2008, 132(6): 1049-1061. |
| Click to display the text | |
| [18] | Messina G, Biressi S, Monteverde S, Magli A, Cassano M, Perani L, Roncaglia E, Tagliafico E, Starnes L, Campbell CE, Grossi M, Goldhamer DJ, Gronostajski RM, Cossu G. Nfix regulates fetal-specific transcription in developing skeletal muscle. Cell 2010, 140(4): 554-566. |
| Click to display the text | |
| [19] | Zaporozhan VN, Bazhora YI. Stem Cells. Odessa: Odessa University, 2007, pp. 132-154. |
| [20] | Zozulya YA, Lysyany NI, Tsymbalyuk VI, et al. Neurogenic Differentiation of Stem Cells. - Kyiv: LLC UIPK. EksOb. 2007: 154-161. |
| [21] | Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, Crapo R, Enright P, van der Grinten CPM, Gustafsson P, et al. Standardisation of spirometry. Eur Respir J 2005, 26(2): 319-338. |
| Click to display the text | |
| [22] | Bradstreet JJ, Sych N, Antonucci N, Klunnik M, Ivankova O, Matyashchuk I, Demchuk M, Siniscalco D. Efficacy of fetal stem cell transplantation in autism spectrum disorders: an open-labeled pilot study. Cell Transplant 2014, 23(S1): S105-S112. |
| Click to display the text | |
| [23] | Siniscalco D, Sych N. Regenerative Potential of Stem Cells for Duchenne Muscular Dystrophy. J Regen Med 2013, 2: 2. |
| Click to display the text | |
| [24] | Sych N, Klunnik M, Ivankova O, Matyaschuk I, Demchuk M, Novytska A, Arkhipenko I, Shalita I, Siniscalco D. Efficacy of fetal stem cells in Duchenne muscular dystrophy therapy. J Neurorestoratol 2014, 2: 37-46. |
| Click to display the text |



