2. Department of Neurosurgery, Tsinghua University Yuquan Hospital, Beijing 100049, China
3. Medical Center, Tsinghua University, Beijing 100084, China
4. Beijing Neurosurgical Institute, Capital Medical University, Beijing 100050, China
Objectives: Adult medulloblastoma is a rare tumor, and few retrospective studies on medulloblastoma have been published to date. A standard treatment regimen has not yet been established. Accordingly, this study was designed to determine the treatment outcomes and prognostic factors for patients with adult medulloblastoma.
Methods: We retrospectively reviewed all cases of adult medulloblastoma at the Beijing Tiantan Hospital between 2004 and 2007. There were 33 patients in total. Tumor removal followed by radiotherapy was performed in all patients. Five patients received maintenance chemotherapy after radiotherapy. Statistical analysis was performed using the log-rank test, Kaplan–Meier method, and Cox regression analysis.
Results: The median follow-up period was 73 months. The 5-year progression-free survival (PFS) and overall survival (OS) rates were 45.5%±8.7% and 51.5%±8.7%, respectively. Recurrence or progression was observed in 21 patients, and 14 patients were still alive. Tumor location at the midline with infiltration to the floor of the fourth ventricle was associated with the worst PFS and OS (P= 0.032). Hemispheric desmoplastic/nodular medulloblastomas (DNMBs) were associated with a better outcome than midline DNMB. The average-risk group tended to have a better 5-year PFS rate than the high-risk group (P= 0.065).
Conclusions: Tumor location is an important prognostic marker for adulthood MB. We propose a molecular and clinical staging system should be established for adulthood MB. Further prospective clinical trials should be performed to draw a conclusion.
A standard treatment regimen for adult medulloblastomas has not yet been established. Currently, most adult patients are treated according to protocols for pediatric patients or based on experience, at an institutional level[5]. Adult medulloblastoma is distinct from pediatric medulloblastoma in terms of molecular features and clinical outcomes[6]. Due to the small number of adult medulloblastoma cases, conducting clinical trials is difficult and data are relatively limited. The clinical and pathological prognostic factors in adult patients have been discussed in only a few reports. We therefore performed a retrospective study of adult medulloblastoma patients treated between 2004 and 2007 at Beijing Tiantan Hospital to investigate the prognostic factors in these patients.
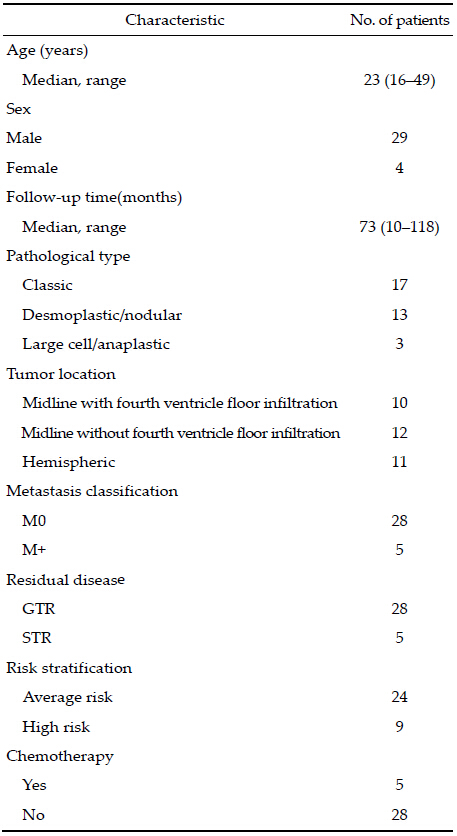
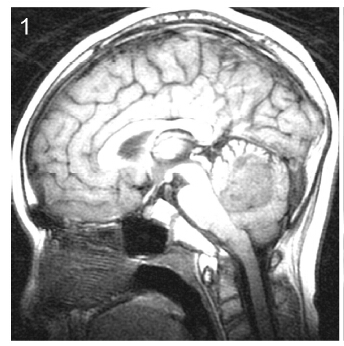
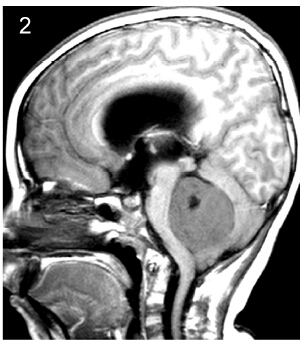
2 Patients and methods This study was approved by Ethics Committee of Tiantan Hospital, Capital Medical University. We retrospectively reviewed the medical records of all patients operated at our hospital between January 2004 and January 2007 who were diagnosed with medulloblastoma and were aged 16 years or older at diagnosis. Follow-up data were collected by telephone or follow-up visits at the clinic.Tumor location was defined as central or lateral on the basis of preoperative computed tomography and magnetic resonance neuroimaging findings and surgical reports (Figures 1-3). Because infiltration of the floor of the fourth ventricle was only observed in cases of centrally located tumors, we classified the tumors into three subtypes: those located at the midline without infiltration of the fourth ventricle floor, those located at the midline with infiltration of the fourth ventricle floor, and those located laterally.
|
| Figure 1 Subtype 1, Subtype 1: midline-located tumor without fourth ventricle floor infiltration. |
|
| Figure 2 Subtype 2, midline-located tumor with fourth ventricle floor infiltration. |
|
| Figure 3 Subtype 3, laterally located tumor. |
| Figures 1-3 Kaplan–Meier estimates for PFS according to clinical parameters. (A) Average risk patients (n = 24) had a better survival (P = 0.065) compared with high risk patients (n = 9). (B) PFS didn’t differ between patients who had presurgical metastasis or not (5 versus 28, P = 0.635). (C) Patients who received subtotal removal had a trend toward worse PFS (5 versus 28, P = 0.066). (D) Midline located MB with infiltration to 4th ventricular floor had a worse prognosis compared to hemispheric MB (12 versus 11, P = 0.025). Other two subgroups didn’t had a significant difference in PFS. (E) DNMB subgroup had a good prognosis, and LC/A MB had a worst outcome, whereas no significant difference could be found between three subgroups (P = 0.342). (F) PFS of hemispheric DNMB was higher than midline DNMB (7 versus 6, P = 0.030). |
Histological examination was performed by two neuropathologists according to the 2007 World Health Organization classification, without knowledge of patients’ prognosis. Tumors were classified into three pathological subtypes: classic medulloblastoma (CMB), desmoplastic/nodular medulloblastoma (DNMB) and medulloblastoma with extensive nodularity (MBEN), large cell and anaplastic (LC/A) medulloblastoma.
The extent of surgery was judged based on perioperative magnetic resonance imaging (MRI) images and medical records. In our hospital, postsurgical enhanced MRI was routinely performed before discharge. Gross total resection (GTR) was defined as residual tumors <1.5 cm2, and subtotal resection (STR) was defined as residual tumors >1.5 cm2 on postsurgical MRI images. The Chang system was used for disease classification as M0 or M+. In our hospital, postoperative lumber puncture for cytological analysis was not routinely performed. Accordingly, the M0 stage in our cohort includes M0 and M1 stages. Patients were classified into the high-risk group (M+ (metastatic) disease and/or residual tumor >1.5 cm2) and average-risk group (no metastasis or residualtumor).
All patients received postoperative radiotherapy. A boost dose was delivered to the entire posterior fossa, and a 15–20 mm margin was added. Five patients received maintenance chemotherapy after radiotherapy. Patients who did not receive any radiotherapy or chemotherapy were excluded.
Statistical analyses were performed using SPSS software (15.0). OS was defined as time from the date of tumor removal to the date of final follow-up or death. Progression-free survival (PFS) was measured from the date of tumor removal to the date of first relapse at any site, death, or the last follow-up. Results are presented as percentage &177; standard error of the mean. The following variables were evaluated for their prognostic value: M stage (M0 vs. M+), extent of surgery (GTR vs. STR), sex, risk stratification (average vs. high), pathological subtype, and tumor location. PFS and OS were calculated using the Kaplan–Meier method. Survival curves were compared between groups using the log-rank test (univariate analysis). Statistical significance was defined when the p-value was less than 0.05. Statisticals analyses were performed using SPSS Statistics 19 (IBM Corp., Armonk, NY, USA). Cox regression analysis was used to identify independent prognostic factors.
3 Results 3.1 Patient and tumor characteristics Between January 2004 and January 2007, 53 patients older than 16 years underwent tumor removal at Beijing Tiantan Hospital. Two patients did not receive radiotherapy or chemotherapy. One tumor was located in the occipital lobe and was not diagnosed as medulloblastoma. Seventeen patients were lost to follow-up. Ultimately, a total of 33 patients were included in our cohort (Table 1). The median age at surgery was 23 years (range, 16–49 years). Four patients were women (12.1%). Twenty-two tumors were located at the midline, and 11, in the cerebellar hemisphere. According to surgical reports, 12 of 22 midline tumors were found to have infiltrated the floor of the fourth ventricle.All 33 pathological slides were reviewed again. Seventeen patients had the classic subtype, 13 had DNMB, and 3 had LC/A medulloblastoma. None of the tumors were classifiable as MBEN.
Among 17 CMBs, 3 were located at the midline without fourth ventricle floor infiltration, 9 were located at the midline with floor infiltration, and 5 were located in the cerebellar hemisphere. Among 13 DNMBs, 5 were located at the midline without floor infiltration, 2 were located at the midline with floor infiltration, and 6 were located in the cerebellar hemisphere. Six of 11 lateral tumors were DNMB, compared with 7 of 22 midline tumors. All 3 patients with the LC/A subtype had tumors located at the midline, with one tumor infiltrating the fourth ventricle floor.
3.2 Surgery and risk stratification GTR was achieved in 28 of 33 tumors, according to medical reports and postsurgical enhanced MRI images. Residual tumors >1.5 cm2 were found on postsurgical MRI images in only 5 cases. Of these 5 tumors, 1 was located in the vermis without floor infiltration; 2, in the vermis with floor infiltration; and 2, in the cerebellar hemisphere.Five patients had tumor metastasis according to perioperative MRI images and surgical reports. Four were classified as having M2 stage disease and 1 was classified as having M3 stage disease. Of the 5 tumors with M+ stage classification, 3 were CMB and 2 were DNMB. Of the 5 patients with metastasis diagnosed preoperatively, 4 showed floor infiltration and ultimately relapsed.
Twenty-four patients were assigned to the averagerisk group, and 9 were assigned to the high-risk group. Of the 9 patients in the high-risk group, 4 had residual tumors >1.5 cm2, 4 had central nervous system (CNS) metastasis, and 1 had both.
3.3 Radiotherapy All but two patients received irradiation to the entire craniospinal axis postoperatively at different centers. One patient received radiotherapy to the posterior fossa only and one did not receive spine irradiation. The median interval between surgery and initiation of radiotherapy was 20.0 days (range, 13–43 days). The median duration of radiation was 39.5 days (range, 32–50 days). The median dose to the posterior fossa was 55.8 Gy (range, 49.2–59.6 Gy, 1.80 Gy/fraction, 5 fractions/week). The median dose to the whole brain was 34.2 Gy (range, 30.0–40.0 Gy). The median dose the spine was 34.2 Gy (range, 23.4–36.5 Gy). 3.4 Chemotherapy Five patients received 2 to 6 cycles of maintenance chemotherapy after radiotherapy. Four of 5 patients were assigned to the high-risk group, and one was assigned to the average-risk group. The chemotherapy regimen was cisplatin/vincristine in 3 patients, cisplatin/ vincristine/etoposide in 1 patient, and unknown in 1 patient. 3.5 Survivalanalysis After a median follow-up of 73 months (range, 10–118 months), recurrence or progression was observed in 21 patients and 14 patients were still alive. The estimated 5-year PFS and OS rates for all patients were 45.5% &177;8.7% and 51.5%&177;8.7%, respectively. The 5-year PFS and OS rates of different subsets of patients are showed in Table 2. No significant differences were observed according to sex, metastasis, extent of surgery, or chemotherapy. Tumors located at the midline with infiltration to the fourth ventricle were associated with a significantly worse PFS than laterally located tumors (P =0.025). 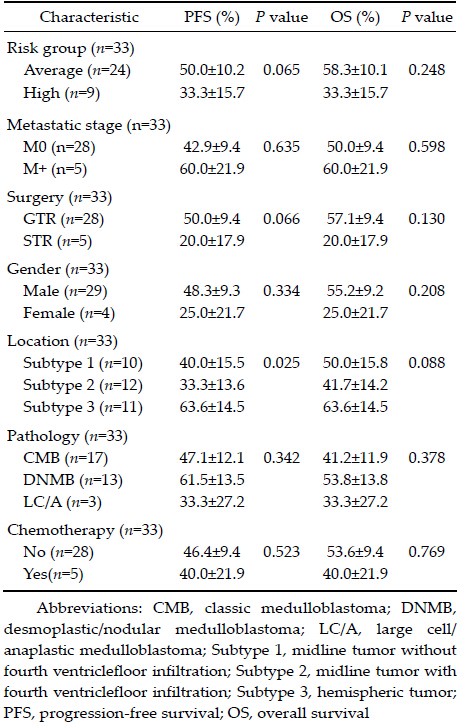 |
At the last follow-up, relapse was noted 6 of 10 midline tumors without fourth ventricle floor infiltration, 11 of 12 midline tumors with fourth ventricle floor infiltration, and 4 of 10 hemispheric tumors.
The average-risk group showed a trend toward better 5-year PFS than the high-risk group (P = 0.065). At the last follow-up, relapse or progression was noted in 11 of 12 high-risk group patients, and 6 patients had died. In the high-risk group, 2 of 4 patients who received maintenance chemotherapy were alive at the last follow-up; only 1 of 5 patients who received radiotherapy alone were alive (P = 0.057).
In Cox regression analysis, tumor location was the only independent predictor of prognosis. Tumors located at the midline with infiltration of the floor of the fourth ventricle had the worst PFS and OS (P = 0.032). No significant difference was observed according to sex, metastasis, pathology, or residual tumor (Table 2).
4 Discussion Most medulloblastoma patients are diagnosed at the age of 5 to 10 years, accounting for 25% of cases of pediatric brain tumors[7]. Because medulloblastomas in adults are rare, few prospective clinical trials have been performed. Because of the very low incidence of medulloblastoma in the adult population, actual survival rates and prognostic factors are difficult to assess[8]. Most authors have reported a 5-year OS rate of <80%, while this was reported as 75% by Brandes et al[9]. who studied 36 patients and as 74% by Call et al[10]. who studied 66 adult patients. A SEER database reported a 5-year OS rate of 65% in a population-based study. In the present study, the estimated 5-year OS rate was 51.5%, lower than those previously reported. The differences could be attributable to the effect of metastasis, residual tumor, adjuvant chemotherapy, tumor location, and histological subtypes on patient outcomes[10, 11, 12, 13].Tumor infiltration of the floor of the fourth ventricle was the only independent prognostic factor in our series (P = 0.032); only few studies have reported this finding[14]. Tumors with floor infiltration in our cohort tended to be associated with CNS metastasis and relapse. Of the 5 cases with preoperatively determined metastasis, 4 were associated with floor infiltration and ultimately showed relapse. Of the 12 tumors with floor infiltration in our series, 11 ultimately relapsed. Midline-located tumors without floor infiltration showed a similar prognosis to laterally located tumors in our cohort. To our knowledge, this is the first study to classify midline-located tumors according to infiltration of the floor of the fourth ventricle. This may explain the better prognosis of hemispheric tumors compared to midline tumors, as reported previously[15, 16]. Some authors[5, 17] have postulated that the worse prognosis of patients with floor infiltration is attributable to incomplete tumor resection, although we did not find an obvious correlation between infiltration and residual tumor in our series. GTR was performed in 10 of 12 patients with floor infiltration in our cohort.
The different subtypes had different origins and outcomes. The WNT subtype has been reported to be located within the fourth ventricle and infiltrate the dorsal brainstem, whereas the SHH subtype has been reported to be mainly hemispheric[4, 18]. Wefers et al.[3] found that type c and type d medulloblastoma most often contacted both cuneate and cochlear nuclei and always infiltrated the fourth ventricle. Adult medulloblastoma is distinct from pediatric medulloblastoma in terms of molecular staging and clinical outcomes. In adult patients, type d tumors have been reported to have the worst outcome, type c tumors are reported to be less common, and WNT tumors are reported to be associated with a worse prognosis than their pediatric counterparts[6]. This may explain why tumor infiltration of the floor of the fourth ventricle was the only independent prognostic factor in our series (P = 0.032). Further study on tumor origin and molecular classification should be performed to draw a definitive conclusion.
We found that patients with GTR tended to have better PFS (P = 0.066) in univariate analysis,although GTR was not an independent prognostic factor in multivariate cox regression analysis. Incomplete resection has been found to affect outcomein some studies[10, 15],whereas no difference in survivalhas been noted in others[19]. This is because near total removal was performed in the majority of patients and because modern diagnostic tools are more sensitive in detecting residual tumor[14]. According to the University of California,San Francisco,risk criteria,a degree of resection of less than 75% is a significant prognostic factor[20]. Attempting maximal tumor resection should be advised in most patients as long as severe surgeryrelated sequelae can be avoided [10, 14].
Histology may be important in medulloblastoma[21]. The age distribution of DNMB is bimodal,with peaks in early childhood and adulthood. The incidence of DNMB was high in our cohort as well (n = 13,39.4%); 6 of the 13 tumors were laterally located and showed no difference in survival compared with CMB. Rutkowski et al.[22] demonstrated the role of DNMB as an independent and significant prognostic factor for favorable outcomes in childhood. However,the role of DNMB in adult patients remains unclear[5]. Adult DNMB has been found to predominantly belong to the SHH subtype and shows a distinct genomic and transcriptional profile from infant DNMB[23]. Adult SHH medulloblastomas are more often located in the lateral hemispheres,whereas infant SHH medulloblastomasare more often located centrally[23]. Friedrich et al.[15] found that DNMB was associated with a better prognosis if the tumors were located centrally. In our cohort,hemispheric DNMB was associated with a better outcome than midline DNMB. At the lastfollow-up,6 of 7 patients with midline DNMB showed relapse or progression,and 5 patients were dead. In contrast,only 1 of 6 patients with hemispheric DNMB in our cohort showed relapse. Thus,adult DNMB could be classified into risk groups according to tumor location. Further studies are warranted to confirm whether the clinical risk can be reflected at a molecular level.
Long-term follow-up is important because late relapse has been reported to be more common in adulthood[17]. In their study based on the SEER database,Smoll[12] reported a “fork-like” Kaplan- Meier survival curve and an “age-by-follow-up interaction” only after 4 years. Differences in survival between adult and pediatric patients could emerge only 4 years after diagnosis and adultsare reported to have poorer survival[12]. This was confirmed in our study,especially in the high-risk group. In our cohort, 3 of 9 high-risk group patients relapsed after 4 years and only one was alive without relapse at the time of follow-up; in contrast,only 3 of 24 average-risk group patients relapsed after 4 years and 11 were alive without relapse. Many authors have reported no significant difference between average-risk and high-risk group patients and no differencebetween adult and pediatric patients[9]. However,this may be in part due to the lack of long-term follow-up.
Whether chemotherapy should be administered to adult medulloblastoma patientsis currently debated[9, 10]. Adjuvant chemotherapy has been recommended in recent years to patients in both the average- and high-risk groups[10, 15, 16]. Our data also showed a trend toward better OS for high-risk group patients who received maintenance chemotherapy. Call et al.[10] recently demonstrated that chemotherapy was associated with a decreased risk of relapse in the posterior fossa in high-risk patients with classic histology (n= 25). Trend toward better PFS and OS was also noted in this small group. Friedrich et al.[15] published the outcomes of adult medulloblastomas patients treated in the HIT2000 trial. Patients treated with a reduced dose (23.4 Gy,n = 9) and maintenance chemotherapy had a similar PFS and OS to those treated with the standard dose (35.2Gy,n = 47),with or without chemotherapy. Brandes et al.[9] published the results of the only prospective trial on averagerisk adult medulloblastoma patients who received postsurgical radiotherapy alone. This long-term followup study showed that the incidence of recurrence increased markedly after 7.6 years. It should be noted that the number of patients who received adjuvant chemotherapy in our study and in previous studies is small,making it difficult to identify a significant difference. The true value of chemotherapy in adult medulloblastoma has to be studied further in prospective clinical trials.
5 Conclusions Here,we report the first series of adult medulloblastoma in China. Tumor infiltration to the fourth ventricle floor was the only independent prognostic factor. Hemispheric DNMB was associated with a better outcome than midline DNMB. Patients with GTR tended to have a better PFS. Late relapse was more common in adult medulloblastoma,especially in the high-risk group. Further prospective clinical trials should be performed to confirm our findings.| [1] | Northcott PA, Shih DJH, Remke M, Cho YJ, Kool M, Hawkins C, Eberhart CG, Dubuc A, Guettouche T, Cardentey Y, et al. Rapid, reliable, and reproducible molecular sub-grouping of clinical medulloblastoma samples. Acta Neuropathol 2012, 123(4): 615-626. |
| Click to display the text | |
| [2] | Taylor MD, Northcott PA, Korshunov A, Remke M, Cho YJ, Clifford SC, Eberhart CG, Parsons DW, Rutkowski S, Gajjar A, et al. Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol 2012, 123(4): 465-472. |
| Click to display the text | |
| [3] | Wefers AK, Warmuth-Metz M, Pöschl J, von Bueren AO, Monoranu C-M, Seelos K, Peraud A, Tonn J-C, Koch A, Pietsch T, et al. Subgroup-specific localization of human medulloblastoma based on pre-operative MRI. Acta Neuropathol 2014, 127(6): 931-933. |
| Click to display the text | |
| [4] | Teo WY, Shen JH, Su JMF, Yu A, Wang J, Chow WY, Li XN, Jones J, Dauser R, Whitehead W, et al. Implications of tumor location on subtypes of medulloblastoma. Pediatric Blood Cancer 2013, 60(9): 1408-1410. |
| Click to display the text | |
| [5] | Rieken S, Gaiser T, Mohr A, Welzel T, Witt O, Kulozik AE, Wick W, Debus J, Combs SE. Outcome and prognostic factors of desmoplastic medulloblastoma treated within a multidisciplinary treatment concept. BMC Cancer 2010, 10: 450. |
| Click to display the text | |
| [6] | Remke M, Hielscher T, Northcott PA, Witt H, Ryzhova M, Wittmann A, Benner A, von Deimling A, Scheurlen W, Perry A, et al. Adult medulloblastoma comprises three major molecular variants. J Clin Oncol: Offic J Am Soc Clin Oncol 2011, 29(19): 2717-2723. |
| Click to display the text | |
| [7] | Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 2007, 114(2): 97-109. |
| Click to display the text | |
| [8] | Silvani A, Gaviani P, Lamperti E, Botturi A, DiMeco F, Franzini A, Ferroli P, Fariselli L, Milanesi I, Erbetta A, et al. Adult medulloblastoma: multiagent chemotherapy with cisplatinum and etoposide: a single institutional experience. J Neuro-Oncol 2012, 106(3): 595-600. |
| Click to display the text | |
| [9] | Brandes AA, Franceschi E, Tosoni A, Blatt V, Ermani M. Long-term results of a prospective study on the treatment of medulloblastoma in adults. Cancer 2007, 110(9): 2035-2041. |
| Click to display the text | |
| [10] | Call JA, Naik M, Rodriguez FJ, Giannini C, Wu WT, Buckner JC, Parney IF, Laack NN. Long-term outcomes and role of chemotherapy in adults with newly diagnosed medulloblastoma. Am JClin Oncol 2014, 37(1): 1-7. |
| Click to display the text | |
| [11] | Brandes AA, Franceschi E. Shedding light on adult medulloblastoma: current management and opportunities for advances. Am Soc Clin Oncol Educat Book/ASCO Am Soc Clin Oncol Meet 2014, 34: e82-e87 |
| Click to display the text | |
| [12] | Smoll NR. Relative survival of childhood and adult medulloblastomas and primitive neuroectodermal tumors (PNETs). Cancer 2012, 118(5): 1313-1322. |
| Click to display the text | |
| [13] | Menon G, Krishnakumar K, Nair S. Adult medulloblastoma: clinical profile and treatment results of 18 patients. J Clin Neurosci 2008, 15(2): 122-126. |
| Click to display the text | |
| [14] | Herrlinger U, Steinbrecher A, Rieger J, Hau P, Kortmann R-D, Meyermann R, Schabet M, Bamberg M, Dichgans J, Bogdahn U, et al. Adult medulloblastoma: prognostic factors and response to therapy at diagnosis and at relapse. J Neurol 2005, 252: 291-299. |
| Click to display the text | |
| [15] | Friedrich C, von Bueren AO, von Hoff K, Kwiecien R, Pietsch T, Warmuth-Metz M, Hau P, Deinlein F, Kuehl J, Kortmann RD, et al. Treatment of adult nonmetastatic medulloblastoma patients according to the paediatric HIT 2000 protocol: a prospective observational multicentre study. Eur JCancer 2013, 49(4): 893-903. |
| Click to display the text | |
| [16] | Padovani L, Sunyach M-P, Perol D, Mercier C, Alapetite C, Haie-Meder C, Hoffstetter S, Muracciole X, Kerr C, Wagner J-P, Lagrange J-L, et al. Common strategy for adult and pediatric medulloblastoma: a multicenter series of 253 adults. Int JRadiat OncolBiolPhys 2007, 68(2): 433-440 |
| Click to display the text | |
| [17] | Chan AW, Tarbell NJ, Black PM, Louis DN, Frosch MP, Ancukiewicz M, Chapman P, Loeffler JS. Adult medulloblastoma: prognostic factors and patterns of relapse. Neurosurgery 2000, 4(3)7: 623-631 |
| Click to display the text | |
| [18] | Gibson P, Tong YA, Robinson G, Thompson MC, Currle DS, Eden C, Kranenburg TA, Hogg T, Poppleton H, Martin J, et al. Subtypes of medulloblastoma have distinct developmental origins. Nature 2010, 468(7327): 1095-1099. |
| Click to display the text | |
| [19] | Kunschner LJ, Kuttesch J, Hess K, Yung WK. Survival and recurrence factors in adult medulloblastoma: the M.D. Anderson Cancer Center experience from 1978 to 1998. Neuro-Oncol 2001, 3(3): 167-173. |
| Click to display the text | |
| [20] | Prados MD, Warnick RE, Wara WM, Larson DA, Lamborn K, Wilson CB. Medulloblastoma in adults. Int J Radiat Oncol Biol Phys 1995, 32(4): 1145-1152. |
| Click to display the text | |
| [21] | Massimino M, Antonelli M, Gandola L, Miceli R, Pollo B, Biassoni V, Schiavello E, Buttarelli FR, Spreafico F, Collini P, et al. Histological variants of medulloblastoma are the most powerful clinical prognostic indicators. Pediatric Blood Cancer 2013, 60(2): 210-216. |
| Click to display the text | |
| [22] | Rutkowski S, Gerber NU, von Hoff K, Gnekow A, Bode U, Graf N, Berthold F, Henze G, Wolff JEA, Warmuth-Metz M, et al. Treatment of early childhood medulloblastoma by postoperative chemotherapy and deferred radiotherapy. Neuro-Oncol 2009, 11(2): 201-210. |
| Click to display the text | |
| [23] | Northcott PA, Hielscher T, Dubuc A, Mack S, Shih D, Remke M, Al-Halabi H, Albrecht S, Jabado N, Eberhart CG, et al. Pediatric and adult sonic hedgehog medulloblastomas are clinically and molecularly distinct. Acta Neuropathol 2011, 122(2): 231-240. |
| Click to display the text |