2. Department of Neurosurgery, Tsinghua University Yuquan Hospital, Beijing 100040, China;
3. Laboratory of Cerebrovascular Research, Department of Surgical Sciences Anaesthesiology and Intensive Care Medicine, Uppsala University Hospital, Uppsala University, Uppsala Se-75185, Sweden
Objectives: Nerve regeneration after peripheral nerve injury is a slow process with a limited degree of functional recovery, resulting in a high disability rate. Thus, accelerating the rate of nerve regeneration and improving the degree of nerve repair is a clinical challenge. This study aimed to investigate the role of growth factor gel combined with small-gap nerve anastomosis in the regeneration of sciatic nerve injury in rats. This was achieved by injecting nerve growth factor (NGF) and basic fibroblast growth factor (bFGF) gel into a silicon chamber that bridged the transection of the nerve.
Methods: In 27 randomly chosen Sprague Dawley rats, a sharp blade was used to transect the right hind leg sciatic nerve. The rats were divided into 3 groups: in groups A and B, silicon tubes containing NGF and bFGF gel or saline, respectively, were used to bridge the nerve proximal and distal ends (3-mm gap), and in group C, the nerve proximal and distal ends were directly sutured. Eight weeks after surgery, the sciatic nerve function index, neural electrophysiology, and muscle wet weight as well as histological, ultrastructural, and immunohistochemical parameters were evaluated.
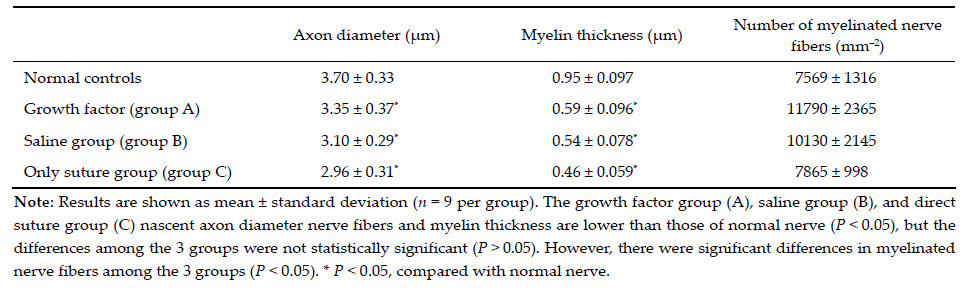
Results: The sciatic nerve function index, nerve conduction velocity, muscle wet weight, density of regenerated nerve fibers, and myelination in group A were better than those in group B or C, but the sciatic nerve function index, muscle wet weight, and thickness of myelination in the 3 groups were not significantly different (P > 0.05). There were no significant differences innerve conduction velocity between groups A and B (P > 0.05), but it was higher in both groups than that of group C (P <0.05). The regenerated nerve fiber density in the 3 groups showed significant differences (P <0.05).
Conclusions: Small-gap nerve anastomosis can provide a good regenerative microenvironment for rat sciatic nerve regeneration, and the combined strategy of growth factor gel with small-gap nerve anastomosis appears to have a superior effect on nerve repair.
In group A,fibrin gel containing heparin (200 U), NGF (2 μg),and bFGF (2 μg) was injected into the silicone tube; in group B,saline was injected into the silicone tube; and in group C,the nerve transection was sutured only. Low‐dose penicillin was added to the incision to prevent infection. Finally,#4 silk was used to close the wound routinely. All animals were housed separately. After 8 weeks,footprint tests and electrophysiological tests were performed and tissue samples were obtained.
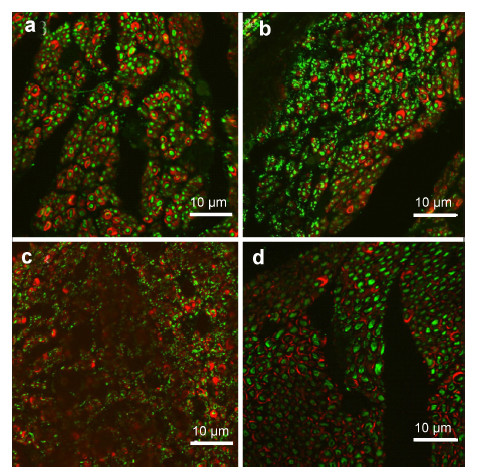
2.4 Sciatic functional index detection The sciatic functional index (SFI) is an important indicator of the degree of recovery of sensory and motor function of the hind limb after sciatic nerve restoration. SFI was measured after 8 weeks with the footprint experiments (walking track analysis)[3]. A SFI score of 0 indicates normal function,and -100 indicates complete loss of sciatic nerve function. 2.5 Nerve electrophysiological test At 8 weeks after surgery,electrophysiological tests were conducted in all animals under chloral hydrate anesthesia. The sciatic nerve in rats was revealed under a microscope and analyzed using the ASB240U biological signal acquisition system. Two bipolar electrodes were respectively placed at the proximal and distal nerve bridges,maintaining a gap of approximately 1.5 cm. Two recording electrodes were inserted into the belly of the gastrocnemius muscle at a 45° angle. After stimulation of the nerve at the proximal and the distal ends of the bridge (single stimulus of 1 mA current),the action potential waveform was traced. The latency difference and distance between the 2 stimulation points were used to calculate the motor nerve conduction velocity. Nerve conduction velocity is calculated as the distance divided by the time required for impulse conduction. 2.6 Muscle wet weight ratio measurement The muscle wet weight ratio is the ratio of the gastrocnemius wet weight on the operative side to that on the normal side. After electrophysiological testing, the gastrocnemius muscles were removed. After the blood was adsorbed on a filter paper,the muscle wet weight was weighed using an analytical balance and the muscle wet weight ratio was calculated. 2.7 Immunofluorescence staining Eight weeks after nerve anastomosis,the nerve segment frozen sections were cut and stained using NF200 and S100 immunofluorescence. Briefly,after 4% paraformaldehyde fixation,the specimens were immersed in 20% (w/v) sucrose solution until they sank. Then, 10‐μm cross sections were cut with a freezing microtome. At room temperature,the slices were incubated in phosphate‐buffered saline (PBS) containing 1% (w/v) goat serum for 1 h before adding NF200 and S100 antibodies (diluted with 1% PBS at a ratio of 1:500) and stored overnight at 4 °C. The sections were washed in 1% PBS 3 times (5 min each) before adding secondary antibody diluted 1:500 with tetramethylrhodamine isothiocyanate (red,to visualize S100) and fluorescein isothiocyanate (green,to visualize NF200) and stored at room temperature for 1 h. After cleaning,the slides were cover slipped and observed under a fluorescence microscope. 2.8 Semi‐thin sections and transmission electron microscopy Eight weeks after nerve anastomosis,segments of nerve tissue were obtained. After 4% paraformaldehyde fixation for 24 h,they were washed in 1% PBS 3 times (5 min each) and then placed in a 1% (w/v) solution of osmium tetroxide. Specimens were embedded in Epon resin,semi‐thin sections were cut (0.5 μm),and ultrathin sections (80 nm) were obtained. The sectionswere stained with 2% (w/v) toluidine blue and observed under a light microscope. The ultrathin sections were counterstained withlead citrate and uranium acetate and examined under a transmission electron microscope (H‐7650B; Hitachi Limited,Japan). 2.9 Statistical analysis The data are expressed as mean ± standard deviation. Groups were compared by analysis of variance using Prism analysis software (GraphPad Software,San Diego, CA,USA). P values < 0.05 were considered statistically significant. 3 Results 3.1 SFI After 8 weeks,the SFI value in group A was -0.71 ± 7.89,which showed the best recovery. The values in groups B and C were -32.4 ± 8.63 and-36.26 ± 8.31, respectively. However,the differences among the 3 groups were not statistically significant (P > 0.05). 3.2 Electrophysiological test Eight weeks after surgery,the sciatic motor nerve conduction velocity in group A was better (33.89 ± 11.12) than that in group B (31.76 ± 13.16) or group C (15.86 ± 12.37). The differences between groups A and C and between groups B and C were statistically significant (P < 0.05),but the difference between groups A and B was not (P > 0.05). 3.3 Muscle wet weight ratio analysis Eight weeks after surgery,the muscles on the involved sides in all 3 groups were atrophied. The muscle wet weight ratio of group A was the highest (0.45 ± 0.13), followed by group B (0.39 ± 0.16) and then group C (0.35 ± 0.12),but the differences among the 3 groups were not statistically significant (P > 0.05). 3.4 Expression of NF‐200 and S100 Immunofluorescence staining of frozen sciatic nerve sections is shown in Figure 1. Green indicates NF‐200, which is an axonal marker protein,and red indicates S100,a Schwann cell myelin marker protein. The results show that the specimens of each group were wrapped in Schwann cells of myelinated nerve fibers, which were,however,smaller than normal nerve fibers (the diameter of normal nerve fibers reaches approximately 20 μm,whereas the diameter of nascent nerve fibers is 5-15 μm). Compared with groups B and C,nerve regeneration density,and myelin thickness in group A were superior,indicating that NGF gel helps regenerate and repair damaged nerves. In group B,the density was also superior to that of group C,which indicates that NGF gel together with a small gap chamber can significantly improve the quality of nerve regeneration. Meanwhile,a small gap around the regeneration chamber can prevent tissue from invading,provide a good microenvironment for nerve regeneration,and avoid the dislocation and derangement of nerve regeneration caused by direct suturing.
|
| Figure 1 NF200 and S100 immunofluorescence straining of frozen sections of distal 2‐mm segments near anastomoses 8 weeks after surgery. (a, b, and c) Groups A, B, and C, respectively. (d) Normal rat sciatic nerve. Green indicates NF‐200; red indicates S100. Scale bar = 10 m. |
|
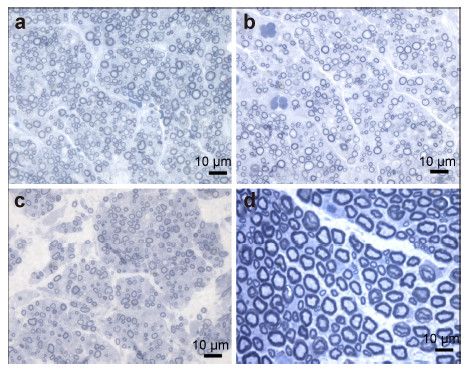
| Figure 2 Light micrographs of myelinated nerve fibersin the distal 1‐mm segments near anastomoses in 8 weeks after surgery (toluidine blue stain). (a, b, and c) Groups A, B, and C, respectively. (d) Normal nerve. Scale bar = 10 μm. |
 |
|
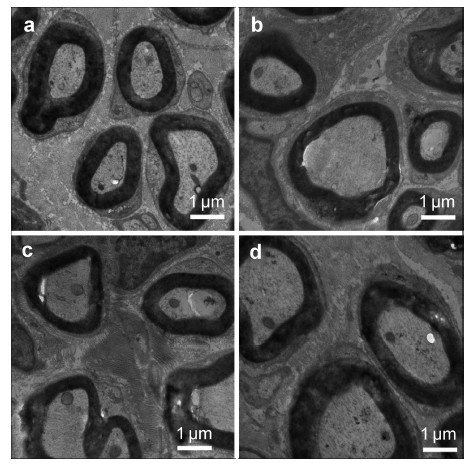
| Figure 3 Transmission electron micrograph of nascent nerve fibers in the distal 1‐mm segments near anastomoses 8 weeks after surgery. (a, b, and c) Groups A, B, and C, respectively. (d) Normal nerve. Scale bar = 1 μm. |
The mechanism of achieving a small‐gap nerve anastomosis is superior to the traditional neural endto‐ end anastomosis according to the nerve chemotaxis theory[4]. Chemotaxis includes nerve tissue specificity, i.e.,nerve growth selectively from the proximal to the distal end,and nerve bundle specificity. Thus,the selective nerve bundle growth from the proximal to the distal nerve bundles and peripheral organ specificity,that is,the selective proximal nerve motor branch,may support growth to the distal nerve,and similar sensory nerve regrowth from the proximal to the distal sensory nerve branch could occur selectively.
For successful regeneration after nerve damage, one of the key issues is to provide a favorable microenvironment for peripheral nerve injury to promote regeneration and functional recovery. The ideal microenvironment of nerve regeneration includes a good blood supply and a chamber that can provide neurotrophic and growth factor regenerative agents[5]. Lundborg and colleagues[6] found that after nerve injury,the liquid secreted by the stump contains some growth and neurotrophic factors,which promote axonal and nerve regeneration. However,the chamber can limit the diffusion of active factors needed to maintain the endogenous NGF concentration for nerve regeneration to create a good microenvironment. A nerve gap also allows a certain degree of deviation of the position, e.g.,to reduce the chance of growth or neuroma formation caused by the tightly stitched nerve segments[7]. Thus,the small‐gap bridging method can accelerate the regeneration of peripheral nerves and improve recovery quality.
Use of exogenous NGF and bFGF can further promote Schwann cell proliferation. The silicone tube regenerative microenvironment provides a reliable tool for the study and application of NGF and bFGF to promote nerve regeneration.
Owing to its excellent biocompatibility,biodegradability, and nontoxicity[8] adhesive fibrin has been widely used as a hemostatic and controlled drug release material. Fibrin glue can be used as a carrier of NGF and bFGF[9, 10]. However,NGF is easily degraded by proteolytic enzymes,whereas heparin,an anticoagulant produced by mast cells,may combine with NGF and bFGF and likely prevent the loss of NGF and bFGF activity[11, 12]. Given the above properties of heparin,we expect that it can increase the stability of NGF in vivo and provide a uniform steady release[12]. The role of heparin as a storage vehicle could enhance the bioavailability of NGF and bFGF.
In conclusion, small‐gap anastomosis created a favorable environment for nerve regeneration chemotaxis, which is conducive to releasing factors in such a way as to continue to induce nerve regeneration. At the same time,direct suturing may cause nerve stump distortion,forced displacement,and other renewable phenomena unsuitable for nerve regeneration. Collectively, the combined strategy of growth factor gel with small‐gap nerve anastomosis appears to play a superior role in peripheral nerve repair.
Acknowledgments This work was supported by the National High Technology Research and Development Program of China (863 Program,No. 2012AA020905),and the Chow Tai Fook Medical Research Special Fund (No. 202836019‐03). Conflict of interests All contributing authors have no conflict of interests.| [1] | Catrina S, Gander B, Madduri S. Nerve conduit scaffolds for discrete delivery of two neurotrophic factors. Eur J Pharm Biopharm 2013, 85(1): 139-142. |
| Click to display the text | |
| [2] | Zhang ZJ, Kou YH, Yin XF, Wang YH, Zhang PX, Jiang BG. The effect of a small gap sleeve suture at the distal anastomosis of a nerve graft on promoting nerve regeneration and functional recovery. Artif Cells Nanomed Biotechnol 2013, 41(4): 282-288. |
| Click to display the text | |
| [3] | Meek MF, Den Dunnen WFA, Schakenraad JM, Robinson PH. Long-term evaluation of functional nerve recovery after reconstruction with a thin-walled biodegradable poly (DLlactide-epsilon-caprolactone) nerve guide, using walking track analysis and electrostimulation tests. Microsurgery 1999, 19(5): 247-253. |
| Click to display the text | |
| [4] | Butí M, Verdú E, Labrador RO, Vilches JJ, Forés J, NavarroX. Influence of physical parameters of nerve chambers on peripheral nerve regeneration and reinnervation. Exp Neurol 1996, 137(1): 26-33. |
| Click to display the text | |
| [5] | Zhao LG, Yao KD. Advances in nerve regeneration through nerve conduit. BME & Clin Med 2003, 7(2): 120-123. (in Chinese) |
| Click to display the text | |
| [6] | Lundborg G. Nerve regenration and repair. A review. Acta Orthop Scand 1987, 58(2): 145-169. |
| Click to display the text | |
| [7] | Song XZ, Gu YD, Xu JG, Li JF. Comparison of suture and conduit nerve repair in different alignment situation. Chin J Hand Surg 1998, 14(4): 250-251. (in Chinese) |
| Click to display the text | |
| [8] | Ahmed TAE, Dare EV, Hincke M. Fibrin: A versatile scaffold for tissue engineering applications. Tissue Eng Part B 2008, 14(2): 199-215. |
| Click to display the text | |
| [9] | Ishii I, Mizuta H, Sei A, Hirose J, Kudo S, Hiraki Y. Healing of full-thickness defects of the articular cartilage in rabbits using fibroblast growth factor-2 and a fibrin sealant. J Bone Joint Surg Br 2007, 89(5): 693-700. |
| Click to display the text | |
| [10] | Jeon O, Ryu SH, Chung JH, Kim SB. Control of basic fibroblast growth factor release from fibrin gel with heparin and concentrations of fibrinogen and thrombin. J Control Release 2005, 105(3): 249-259. |
| Click to display the text | |
| [11] | Ho YC, Mi FL, Sung HW, Kuo PL. Heparin-functionalized chitosan-alginate scaffolds for controlled release of growth factor. Int J Pharm 2009, 376(1-2): 69-75. |
| Click to display the text | |
| [12] | Tanihara M, Suzuki Y, Yamamoto E, Noguchi A, Mizushima Y. Sustained release of basic fibroblast growth factor and angiogenesis in a novel covalently crosslinked gel of heparin and alginate. Biomed Mater Res 2001, 56(2): 216-221. |
| Click to display the text |



