Objective: Parkinson's disease (PD), which is one of the most common neurodegenerative disorders, is characterized by the loss of dopamine (DA) neurons in the substantia nigra in the midbrain. Experimental and clinical studies have shown that fetal neural stem cells (NSCs) have therapeutic effects in neurological disorders. The aim of this study was to examine whether cells that were differentiated from NSCs had therapeutic effects in a rat model of PD.
Methods: NSCs were isolated from 14-week-old embryos and induced to differentiate into neurons, DA neurons, and glial cells, and these cells were characterized by their expression of the following markers:βⅢ-tubulin and microtubule-associated protein 2 (neurons), tyrosine hydroxylase (DA neurons), and glial fibrillary acidic protein (glial cells). After a 6-hydroxydopamine (6-OHDA)-lesioned rat model of PD was generated, the differentiated cells were transplanted into the striata of the 6-OHDAlesioned PD rats.
Results: The motor behaviors of the PD rats were assessed by the number of apomorphine-induced rotation turns. The results showed that the NSCs differentiated in vitro into neurons and DA neurons with high efficiencies. After transplantation into the striata of the PD rats, the differentiated cells significantly improved the motor deficits of the transplanted PD rats compared to those of the control nontransplanted PD rats by decreasing the apomorphine-induced turn cycles as early as 4 weeks after transplantation. Immunofluorescence analyses showed that the differentiated DA neurons survived more than 16 weeks.
Conclusions: Our results showed that cells that were differentiated from NSCs had therapeutic effects in a rat PD model, which suggests that differentiated cells may be an effective treatment for patients with PD.
1 Introduction Parkinson’s disease (PD),which is one of the most common neurodegenerative diseases,affects about 1% of the population aged 60 years and older and 3%-5% of the population over the age of 85[1, 2]. Most cases of PD are sporadic with unknown etiology (80%-90%),but a small percentage of cases (10%-20%) are familial and can be linked to a particular monogenic mutation or PD‐related genes[3, 4, 5].
Pathologically,PD is characterized by Lewy body formation. Lewy bodies are composed of a number of proteins,including alpha‐synuclein,ubiquitin,and tau,that are misfolded in dopamine (DA) neurons[6]. The spread of Lewy bodies in the brain causes motor dysfunctions that are accompanied by cognitive impairments,including hallucinations,dementia,and speech difficulties[7, 8]. The loss of DA neurons in the midbrain eventually causes both the motor and the nonmotor symptoms of PD. The motor symptoms have typically been used to diagnose PD in the clinic. The main motor symptoms are resting tremors,muscle rigidity,bradykinesia (slowness of movement),and postural instability[9, 10]. The nonmotor symptoms of PD include depression,constipation,pain,genitourinary problems and sleep disorders,and these symptoms may precede the motor dysfunctions[11, 12]. Treatment with levodopa has been shown to improve the motor symptoms of PD,but it cannot stop the disease progression or alleviate nonmotor symptoms[13, 14].
Stem cell transplantation with neural stem cells (NSCs) from human fetal brain tissue,human embryonic stem cells (ESCs),mesenchymal stem cells from adults or fetuses,and induced pluripotent stem cells that were reprogrammed from patient somatic fibroblasts or blood cells have shown great potential for treatments of patients with PD[15, 16, 17]. NSCs that are transplanted into rat brains migrate and differentiate in a site‐specific manner,and these NSCs appear to differentiate preferentially into DA neurons in PD model rats that have depleted DA levels,which suggests that these cells have a sensitivity to environmental factors in the host brains[18, 19]. Andersson et al.[20] selectively expressed homeodomain proteins in DA cell progenitors in the ventral midbrain and found that Lmx1a and Msx1 are involved in the development of DA neurons. Burbach and Smidt[21] have reported that these factors initiate the differentiation of neural progenitor cells in chick embryos into midbrain DA neurons. These findings suggest that Lmx1a and Msx1 play important roles in the development and maturation of DA neurons. The overexpression of the transcription factor ASCL1 has been reported to promote neurogenesis in human neural progenitor cells and produce larger neurons with more neurites[22, 23]. The results of these studies suggest that neurogenesis can be increased by upregulating neuron‐specific transcription factors in NSCs. Papanikolaou et al.[24] differentiated NSCs into DA neurons by adding transcription factors that are specific for DA neurons, including sonic hedgehog (SHH) and fibroblast growth factor (FGF8),and they demonstrated the morphological characteristics of the forebrain DA neurons. However,these differentiated neurons remained morphologically immature and exhibited more cell death in the final differentiation stage of NSCs.
Based on the results of the animal studies,several clinical trials involving the transplantation of human fetal DA neurons into patients with PD have been conducted and have shown significant behavioral and histological improvements[25, 26, 27, 28, 29]. Greater improvements have been reported for younger patients with PD, which implies that the treatment may be limited in efficacy in certain subpopulations. Some studies have reported poor long‐term graft survival. In general, because clinical trials have shown extremely variable functional outcomes,further clinical and imaging evaluations of the therapy need to be conducted in order to improve the outcomes and reduce their variability[30, 31, 32].
Even though some improvements have been reported in animal studies and clinical trials involving fetal NSCs,gliosisis frequently observed in the host brains,which suggests that the differentiation of NSCs in in vitro cultures might differ from in vivo differentiation. With the goals of producing more mature DA neurons in the graft cells and achieving better clinical outcomes,we differentiated fetal NSCs into neurons and astrocytes and investigated the therapeutic effects of these differentiated cells in a rat model of PD.
2 Materials and methods 2.1 Materials and reagents All of the reagents and chemicals were purchased from the following companies. The Dulbecco’s Modified Eagle’s Medium (DMEM),DMEM/Nutrient Mixture F12 (F12),fetal bovine serum,nonessential amino acid (NEAA),glutamine,penicillin‐streptomycin (P/S), mitomycin C,knockout serum replacement,mercaptoethanol, FGF2,TrypLE,Polyprene,and N2 were purchased from Thermo Fisher Scientific Inc. (Waltham, MA,USA). The gelatin,noggin,transforming growth factor,SHH,and glial cell line‐derived neurotrophic factor (GDNF) were purchased from R&D Systems, Inc. (Minneapolis,MN,USA),and the brain‐derived neurotrophic factor (BDNF) and FGF8b were purchased from PeproTech (Rocky Hill,NJ,USA). The SB431542 was purchased from Tocris Bioscience (Bristol,UK). Finally,the poly‐L‐ornithine/laminin, paraformaldehyde (PFA),goat serum,donkey serum, Triton X‐100,Hoechst 33258,sucrose,isoflurane, ascorbic acid,and apomorphine were purchased from Sigma‐Aldrich Co. LLC (St. Louis,MO,USA). 2.2 Isolation and culture of the fetal NSCs All of the research involving human participants was performed with the approval of the Human Research Ethics Committee of our hospital (Liaocheng People’s Hospital/The Affiliated Liaocheng Hospital,Taishan Medical University,Liaocheng,China). All of the subjects or responsible persons completed and signed consent forms before the biopsies were performed. Institutional Review Board approval was obtained for all of the human‐related studies.The brain tissues of human fetuses between 12 and 15 weeks of gestation were obtained from the Department of Gynecology and Obstetrics in Liaocheng People’s Hospital/The Affiliated Liaocheng Hospital. Human neural progenitor/stem cells were prepared from the fetal midbrains and induced to proliferate into neurospheres,as previously described[33, 34]. The freshly dissected fetal brain tissue was dissociated in TrypLE and seeded in 10‐cm dishesata density of 200,000 cells/mL in culture medium containing DMEM/F12,knockout serum replacement,B27,NEAA,glutamine,P/S, basic FGF (20 ng/mL),and epidermal growth factor (20 ng/mL) in order to form neurospheres in 2-3 weeks. Once the neurospheres reached a diameter of 300-500 μm,they were passaged by pipetting the neurospheres into small clumps with diameters of 100-200 μm. They were then cultured in maintenance medium containing DMEM/F12,N2,B27,NEAA, glutamine,and P/S,which was supplemented with basic FGF (20 ng/mL) and epidermal growth factor (20 ng/mL). The neurospheres were passaged every 7-14 days by changing the medium every 3-4 days.
2.3 Neural differentiation Based on previous protocols for neuron and DA neuron differentiation[22, 24],we dissociated the neurospheres into single cells with Accutase. The cells (1,000 cells/μL) were plated on glass coverslips coated with poly‐ L‐ornithine/laminin in 6‐well plates and cultured in differentiation medium containing DMEM/F12,2% B27, and 1% P/S with the addition of BDNF (10 ng/mL), ascorbic acid,GDNF (10 ng/mL),transforming growth factor‐β (1 ng/mL),and cyclic AMP (500 μM,Sigma‐ AldrichCo. LLC) to increase the survival of the differentiated neural cells. The plated cells were continuously maintained and differentiated for 7 and 14 days for the immunohistochemical analyses or collected for the transplantation study. 2.4 6‐OHDA‐induced rat model of PD and cell transplantation All of the animal experiments were performed in accordance with local ethical guidelines and with the approval of the Animal Care and Use Committee of Liaocheng People’s Hospital/The Affiliated Liaocheng‐ Hospital. For the lesioning of the DA neurons in the substantia nigra,male Sprague‐Dawley rats (200-250 g) were anesthetized with subcutaneous injections of 10% trichloroacetaldehyde hydrate. The rats were fixed in a KOPF stereotaxic frame (David Kopf Instruments, Tujunga,CA,USA),and 16 μg of 6‐OHDA (H4381; Sigma‐Aldrich Co. LLC) in 4 μL of phosphate‐buffered solution (PBS) containing 0.2 mg/mL of ascorbic acid was stereotaxically injected at a rate of 1 μL/min into the right substantia nigra of the midbrain of the rats in accordance with a distance of anterior‐posterior (-2.0 mm),lateral (-2.5 mm),and vertical (-7.8 mm) from the bregma. For transplantation,the neural cells that were differentiated from the neurospheres (NSCs) were collected at 7-10 days of differentiation and diluted in PBS to yield a cell concentration of 1 × 105 cells/μL. 5 μL of cell suspension or PBS was transplanted into the striata of the anesthetized PD rats 4 weeks after the 6‐OHDA lesions with a Hamilton syringe (0.3-0.5 mm diameter) over a period of 5-10 min rats in accordance with the distance from the bregma of anterior‐posterior (+0.7 mm),lateral (-3 mm),and vertical (-5 mm). In order to avoid rejection of the transplanted cells by the immune system,the transplanted rats were intraperitoneally injected with cyclosporine A (10 mg/kg; Sigma‐Aldrich Co. LLC) 24 h before transplantation and every day afterwards until the rats were sacrificed. The PBS‐transplanted control rats were administered the same dose of cyclosporine A in order to control for the effects of the immunosuppressive agents on the rat brains. 2.5 Behavior testing In order to determine if the rat model of PD was successfully generated by the 6‐OHDA injection,the number of rotation turns of the rats was recorded 3 min after apomorphine was subcutaneously injected into the 6‐OHDA‐lesioned rats. The rats with over 200 complete asymmetric rotation turns in 30 min in response to apomorphine 4 weeks after the 6‐OHDA injections were considered successful models of PD, and they were used as recipients of the cell transplantation and PBS injections. The ratios of the number of turns after transplantation versus the number of turns before transplantation were calculated and compared between the rats with transplanted cells and the rats with PBS injections 4,8,12,and 16 weeks after transplantation. 2.6 Immunocytochemistry and immunohistochemistry For the immunocytochemical characterization of the cultured cells,undifferentiated neurospheres or attached differentiated cultures were fixed in 4% PFA for 15 min at room temperature,which was followed by three washes in PBS. After incubation in blocking buffer for 45 min,the cells were incubated with primary antibodies for 3-4 h at room temperature and then with secondary antibodies for 1 h at room temperature. Finally,the cells were incubated in Hoechst 33258 (1:10,000) for 2-5 min to stain the cell nuclei. For the immunohistochemistry,the 6‐OHDA‐lesioned rats with NSC‐derived neural grafts were perfused with 4% PFA 16 weeks after transplantation. The whole brains of the rats were removed and fixed in 4% PFA for 24 h. Subsequently,the fixed brains were cryoprotected in 30% sucrose for 24-48 h,embedded in Tissue‐Tek OTC (Sakura Finetek,USA,Inc.,Torrance,CA,USA),and sectioned into 20‐μm thick slices with a cryostat (Leica CM1950,Leica Biosystems Nussloch GmbH,Nussloch, Germany). One in 5 sections was immunostained with the primary antibody at 4 °C overnight and then incubated with a fluorescence‐labeled secondary antibody for 3-4 h at room temperature. Finally,the sections were incubated in Hoechst 33258 (1:10,000) for 2-5 min. All of the images were obtained with a fluorescence microscope (Nikon Ti,Nikon Corporation, Tokyo,Japan) or a laser scanning confocal microscope (Olympus Corporation,Tokyo,Japan). The primary antibodies were used to visualize the following structures: the nucleus (antibodies: hNUC,mouse monoclonal,1:500,EMD Millipore,Billerica,MA,USA; nuclear cell adhesion molecule (NCAM),mouse monoclonal, 1:500,EMD Millipore),neurons (βⅢ‐tubulin (Tuj1),mouse monoclonal,1:500,Sigma‐Aldrich Co. LLC; microtubule‐associated protein 2 (MAP2),mouse monoclonal,1:500,Sigma‐Aldrich Co. LLC),DA neurons (tyrosine hydroxylase (TH),rabbit polyclonal, 1:250,Pel Freez),and glia (glial fibrillary acidic protein (GFAP),rabbit polyclonal,1:250,Dako Denmark A/S,Glostrup,Denmark). The secondary antibodies included the following fluorescence‐labeled secondary antibodies: Alexa Fluor 488‐goat anti‐mouse and Cy3‐goat anti‐rabbit (for both,1:1,000,The Jackson Laboratory,Bar Harbor,ME,USA). 2.7 Statistical analysis All of the behavioral test data were statistically analyzed with Student’s t‐tests (SPSS statistics software,version 13.1,IBM Corporation,Armonk,NY,USA). 3 Results 3.1 In vitro differentiation of the fetal NSCs into DA neurons After the NSCs were isolated and propagated,they were cultured as neurospheres in T25 flasks in maintenance medium (Figure 1). For neuron and DA neuron differentiation,the neurospheres were treated with Accutase,pipetted into single cells,and then plated on coverslips coated with poly‐L‐ornithine/laminin in 6‐well plates. After 7 and 14 days of differentiation,the cells were characterized for the expression of neuronal markers (Tuj1 and MAP2) and the DA neuronal marker TH with immunocytochemistry (Figure 2a). Some of the differentiated cells also expressed GFAP (Figure 2a),which indicated that the differentiated cells contained glial cells. The percentages of the differentiated cells that expressed the different markers of the differentiated neurons,DA neurons,and glial cells were 67.8 ± 7.3% expressing TUJ1,62.1 ± 6.7% expressing MAP2,19.9 ± 1.5% expressing TH,and 19.4 ± 1.6% expressing GFAP after 7 days of differentiation. After 14 days of differentiation,the percentages were 63.5 ± 5.7% expressing TUJ1,56.6 ± 6.0% expressing MAP2,24.3 ± 3.7% expressing TH,and 20.0 ± 4.5% expressing GFAP (Figure 2b). In addition,the differentiation efficiencies between 7 and 14 days of differentiation of the cells expressing TUJ1 (t = 0.108, P = 6.498),MAP2 (t = 0.053,P = 6.575),TH (t = 0.004, P = 3.417),and GFAP (t = 0.374,P = 3.177) did not differ significantly. Overall,about 65.65% and 59.35% of the total cells were positively stained for Tuj1 and MAP‐2,respectively. The DA neurons expressing TH accounted for 33.6% (22.1/65.65) of the total cells. These differentiated neural cells were collected for the transplantation study.
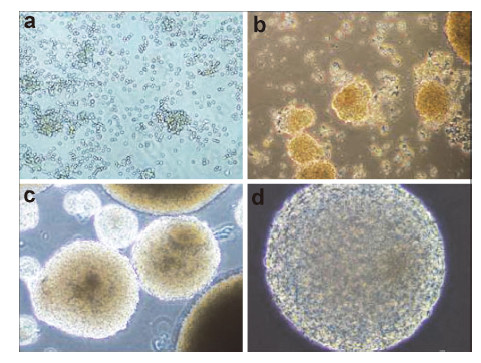
|
| Figure 1 The isolation of the human fetal striatum‐derived neural stem cell (NSC) population at 14 weeks.The suspension of fetal striatal cells was seeded in 10‐cm dishes in order to form adherent clones that started to appear after 24-48 h,as shown in (a).After 10-14 days,floating spheres formed (b).After 2 passages,the neurospheres formed as shown in (c,10×) and (d,20×). |
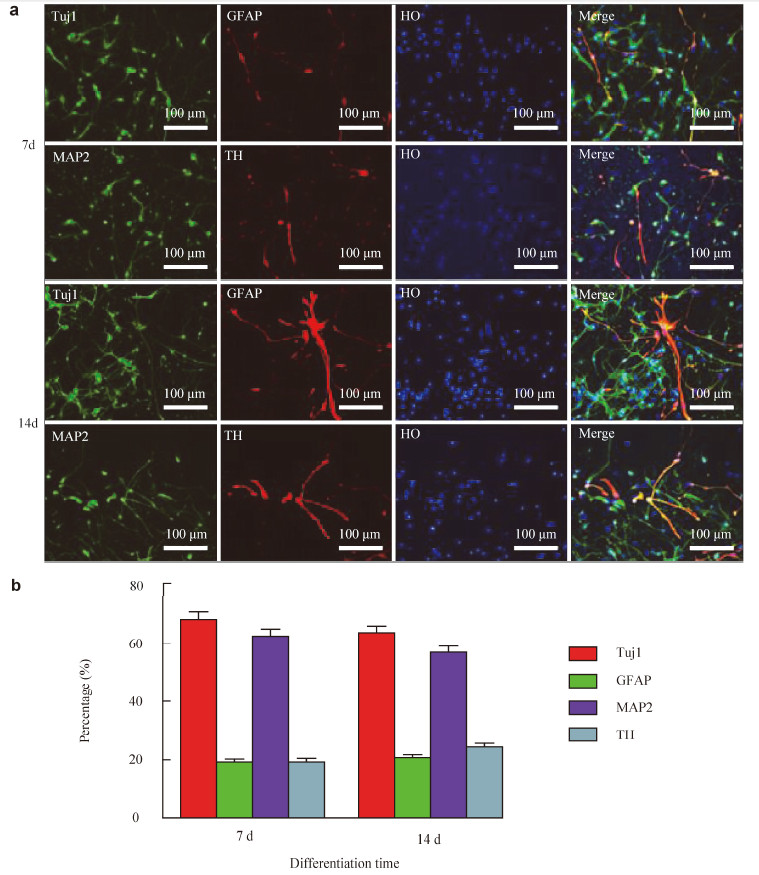
|
| Figure 2 The in vitro neuronal and dopamine (DA) neuron differentiation of fetal NSCs.(a) Characterization of the differentiated neural cells from fetal NSCs by immunocytochemistry after 7 or 14 days of differentiation.βⅢ‐tubulin (Tuj1) and glial fibrillary acidic protein (GFAP) immunoreactivity represent the differentiated neurons and astrocytes,respectively,while microtubule‐associated protein‐2(MAP‐2) and tyrosine hydroxylase (TH) represent the differentiated neurons and DA neurons,respectively.(b) The percentages of neurons, astrocytes,and DA neurons of the total cell population. |
The turn testing was performed on all 16 rats 4,8, 12,and 16 weeks after transplantation,and the data were compared between the cell‐transplanted rats and control rats. A systematic reduction in apomorphineinduced turns was observed in the group of PD rats with transplanted cells (Figure 3). The average turn ratios in 30 min 4,8,12,and 16 weeks after transplantation were reduced to 0.7561 ± 0.0515, 0.6679 ± 0.0492,0.5359 ± 0.0888,and 0.3888 ± 0.0536, respectively,compared with 0.9104 ± 0.0566,0.8290 ± 0.0665,0.8205 ± 0.1031,and 0.8212 ± 0.1217,respectively, in the control group. The turn ratios differed significantly between the PD rats with transplanted cells and the PD rats with PBS injections for the 4th week (t = 5.145,P = 0.03),8th week (t = 5.014,P = 0.0004),12th week (t = 5.351,P = 0.0002),and 16th week (t = 8.531, P < 0.0001). This improvement in motor dysfunction was attributed to the grafting of the differentiated neural cells or the effects of the growth factors that were released from the transplanted cells.
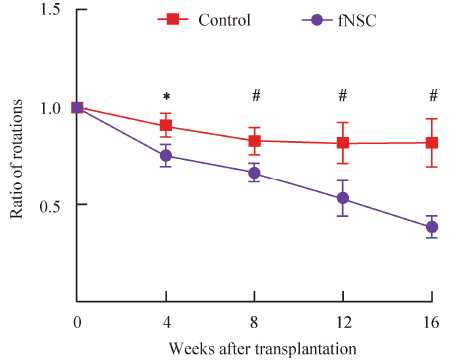
|
| Figure 3 Comparison of the motor behavior assessments of the 6‐hydroxydopamine (6‐OHDA)‐induced rats with grafted NSCs and the control 6‐OHDA‐induced rats treated with phosphatebuffered solution (PBS).Compared to the motor dysfunctions of the rats treated with PBS injections,the motor dysfunctions in the rats with transplanted NSCs were significantly improved on the 4th week (t=5.145,P=0.03),8th week (t=5.014,P=0.0004),12th week (t=5.351,P=0.0002),and 16th week (t=8.531,P < 0.0001)(*indicates P < 0.05;#indicates P < 0.01). |
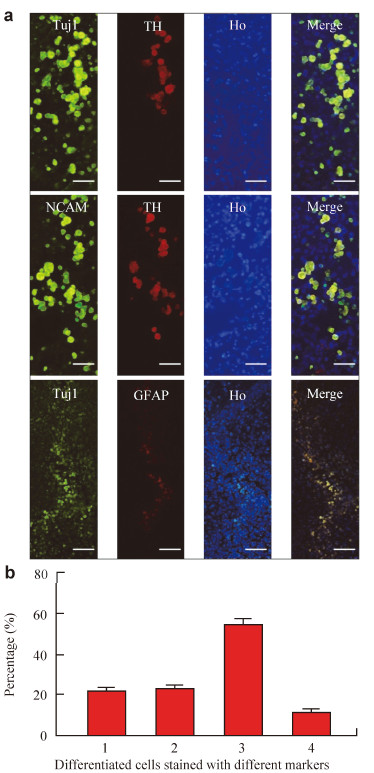
|
| Figure 4 The NSC‐derived neurons and DA neurons survive and integrate into the striata of the 6‐OHDA‐lesioned rats.(a) Upper panel:NSC‐differentiatedcell grafts immunostained for βⅢ‐tubulin (Tuj1,green) and TH (red).Middle panel:Human‐specific nuclear cell adhesion molecular (NCAM) antigen (green) and TH (red).Lower panel:βⅢ‐tubulin (Tuj1,green) and GFAP (red).The cell nucleus was stained with Hoechst 33258(Ho,blue)16 weeks after the transplantation.(b) The percentages of NSC‐differentiated cells in vivo.1:TH/Tuj1(21.33 ± 2.08%);2:TH/NCAM (12.33 ± 2.08%); 3:Tuj1/Ho (59.67 ± 3.79%);4:GFAP/Ho (11.11 ± 2.00%).((a) Upper and Middle panels,magnification:200×,scale bar=50 μm;Lower panel, magnification:100×,scale bar=100 μm). |
Quantification of these observations showed that 59.67% of the transplanted cells were stained with the Tuj1 antibody,whereas the GFAP‐stained cells (glial cells) accounted for 11.11% of the transplanted cells, which suggested that most of the differentiated neurons survived well in the brains of the PD rats. We found that about 21% of the neurons were stained with TH, which indicated that the DA neurons maintained their phenotype after transplantation into the host brains (Figure 4b).
4 Discussion Fetal brain‐derived NSCs have been isolated not only from midbrains but also from other regions of fetal brains or the hippocampus and subventricular zone of mammalian brains[38, 39, 40]. Several studies have shown that fetal brain‐derived NSCs are effective in the treatment of PD animal models and patients with PD[41, 42, 43]. Even though these NSCs appear to differentiate preferentially into DA neurons in PD model rats with depleted host DA neurons[18, 19],most studies have shown that a limited number of neurons and DA neurons differentiate from the grafted NSCs as most of the NSCs become glial cells in vivo. To overcome this problem,we transplanted all of the differentiated cells,including neurons,DA neurons,and astrocytes, into the 6‐OHDA‐lesioned rats and studied their therapeutic effects on PD. We did not transplant DA neurons only because other neurons and astrocytes also support and nourish the DA neurons. The results of our study showed that the in vitro‐differentiated neural cells improved the locomotive deficits of 6‐ OHDA‐lesioned parkinsonian rats. These improvements were seen as early as 4 weeks,and they continued through 16 weeks after the differentiated cells were transplanted into the striata of rats. We found that the neurons and DA neurons grew out from the grafted cells in the rat brains and that less glial cells were stained in the grafted cells,which indicated that transplantation of differentiated NSCs represents an effective approach for the treatment of PD. Papanikolaou et al.[24] differentiated NSCs into DA neurons with a modified protocol that included conditions that were similar to the culture conditions that are used to differentiate ESCs into DA neurons. They demonstrated that the differentiated cells had the morphological characteristics of forebrain DA neurons. We found that NSCs that were derived from the human striatum generated significantly higher numbers of mature neurons that expressed Tuj1 and MAP2,and some of these cells expressed the DA neuron marker of TH, which indicated that the human striatal NSCs have an intrinsic fate specification that is consistent with their region of origin.We observed that two rats in the transplantation group died during the experiments. One rat died in the 5th week,and the other died in the 8th week after the transplantation. The reasons for the deaths were not clear. However,the deaths were not a result of the surgeries because no rats in the PBS group died,and they were subjected to the same surgical conditions. In addition,if the surgeries caused the deaths,then the rats would have most likely died earlier. The deaths could not have been caused by the transplanted cells as we prepared the single cells in suspension or used a limited volume of cell suspension for transplantation by following the standard protocols used in our previous studies and in other published studies. One possibility is that the rats may have had an unidentified infection or other disease. Even though different growth factors may contribute different levels of therapeutic effects to the functional recovery of PD rats,we do not think the growth factors in the cell suspension were a significant factor in the results of this study. Growth factor‐mediated contributions to recovery occur only in the first few weeks and cannot be sustained for 12-16 weeks[37]. The growth factors were included in the cell suspension to increase the survival of the transplanted cells because most of the cells die easily in the first few days in new living tissues (environment).
In order to determine how much time was needed to obtain fully differentiated neurons and DA neurons from NSCs,we plated NSCs and characterized the differentiated NSCs 7 and 14 days after differentiation. About 68% of the plated NSCs differentiated into neurons,and 20% of the plated NSCs differentiated into DA neurons on both differentiation days 7 and 14,which suggested that 7 days was enough time for the NSCs to differentiate into DA neurons. Because the efficiency of DA neuron production was only about 20% in the basic neural differentiation medium,we are considering adding specific factors,such as FGF8 and SHH,in order to increase the DA neuron conversion from NSCs by modifying the differentiation protocol by Papanikolaou et al.[3, 24, 44] Our study showed that Tuj1‐and MAP2‐stained neuronal cells accounted for 65.65% and 59.35%,respectively,of the total differentiated cells. This difference was probably related to the finding that the expression of Tuj1 represents all neurons,whereas the expression of MAP2 mainly appears in only mature neurons. In this study,we found that about 20% of the transplanted neurons were TH‐stained DA neurons in vivo. This percentage was similar to that observed with in vitro differentiation, which suggested that almost all of the differentiated DA neurons survived and functioned in vivo.
Some studies have reported that the stem cell mediated‐functional recovery of PD rats occurs 2-3 months after the transplantation of the ESC‐derived NSCs[45],while we found that the transplantation of differentiated neural cells induced functional improvement as early as 4 weeks after transplantation,which was similar to the findings of a previous study[46]. It is likely that the DA neurons from NSCs can immediately release the DA once they survive in the rat brains. However,one study has reported that the simple secretion of DA is not sufficient and that the integration of DA neurons into the host striatal tissue may be required to achieve a stable functional recovery[47]. More studies are needed to determine the DA levels that are required to obtain the functional recovery of 6‐OHDA‐lesioned rats. A recent high‐performance liquid chromatography study has shown that ESCderived NSCs can differentiate into DA‐like neurons that express the DA neuron marker TH and release DA in vivo after they are transplanted into the striata of PD rats. The DA release was confirmed to come directly from the grafted ESC‐derived NSCs rather than from injured original host cells[48]. In future studies, we will examine DA release and use the patch‐clump technique to examine the in vivo electrophysiological functions of the grafted cells.
In conclusion,we demonstrated that NSCs that were produced from human fetal striatum differentiated into neurons and DA neurons with high efficiencies and improved the symptoms of motor dysfunction of a rat model of 6‐OHDA‐induced PD after the NSCderived differentiated cells were transplanted into the rat brains. However,our study did not explore other effects of the transplanted cells in these rat models. We plan to investigate these mechanisms in order to increase the efficiency and applicability of transplanted neural cells in the treatment of patients with PD.
Acknowledgements We would like to thank all the participants in the study. Funding was supported by the National Natural Science Foundation of China (NSFC,No. 81271251). Conflict of interests The authors have no financial interest to disclose regarding the article.| [1] | Bezard E, Dovero S, Prunier C, Ravenscroft P, Chalon S, Guilloteau D, Crossman AR, Bioulac B, Brotchie JM, Gross CE. Relationship between the appearance of symptoms and the level of nigrostriatal degeneration in a progressive 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridinelesioned macaque model of Parkinson's disease. J Neurosci , 2001, 21(17):6853-6861. |
| Click to display the text | |
| [2] | de Rijk MC, Launer LJ, Berger K, Breteler MMB, Dartigues JF, Baldereschi M, Fratiglioni L, Lobo A, Martinez-Lage J, Trenkwalder C, Hofman A. Prevalence of Parkinson's disease in Europe:A collaborative study of populationbased cohorts. Neurology , 2000, 54(11 Suppl 5):S21-S23. |
| Click to display the text | |
| [3] | Han FB, Wang W, Chen BX, Chen C, Li S, Lu XJ, Duan J, Zhang Y, Zhang YA, Guo WN, Li GY. Human induced pluripotent stem cell-derived neurons improve motor asymmetry in a 6-hydroxydopamine-induced rat model of Parkinson's disease. Cytotherapy , 2015, 17(5):665-679. |
| Click to display the text | |
| [4] | Costello S, Cockburn M, Bronstein J, Zhang X, Ritz B. Parkinson's disease and residential exposure to maneb and paraquat from agricultural applications in the central valley of California. Am J Epidemiol , 2009, 169(8):919-926. |
| Click to display the text | |
| [5] | Ryan BJ, Hoek S, Fon EA, Wade-Martins R. Mitochondrial dysfunction and mitophagy in Parkinson's:From familial to sporadic disease. Trends Biochem Sci , 2015, 40(4):200-210. |
| Click to display the text | |
| [6] | Savitt JM, Dawson VL, Dawson TM. Diagnosis and treatment of Parkinson disease:Molecules to medicine. J Clin Invest , 2006, 116(7):1744-1754. |
| Click to display the text | |
| [7] | Aarsland D, Andersen K, Larsen JP, Lolk A, Nielsen H, Kragh-Sørensen P. Risk of dementia in Parkinson's disease:A community-based, prospective study. Neurology , 2001, 56(6):730-736. |
| Click to display the text | |
| [8] | Lees AJ, Hardy J, Revesz T. Parkinson's disease. Lancet , 2009, 373(9680):2055-2066. |
| Click to display the text | |
| [9] | Obeso JA, Rodriguez-Oroz MC, Rodriguez M, DeLong MR, Olanow CW. Pathophysiology of levodopa-induced dyskinesias in Parkinson's disease:Problems with the current model. Ann Neurol , 2000, 47(4 Suppl 1):S22-S32. |
| Click to display the text | |
| [10] | Cheng HC, Ulane CM, Burke RE. Clinical progression in Parkinson disease and the neurobiology of axons. Ann Neurol , 2010, 67(6):715-725. |
| Click to display the text | |
| [11] | Bergman H, Deuschl G. Pathophysiology of Parkinson's disease:From clinical neurology to basic neuroscience and back. Mov Disord , 2002, 17(Suppl 3):S28-S40. |
| Click to display the text | |
| [12] | Lee HM, Koh SB. Many faces of Parkinson's disease:Non-motor symptoms of Parkinson's disease. J Mov Disord , 2015, 8(2):92-97. |
| Click to display the text | |
| [13] | Fahn S, Oakes D, Shoulson I, Kieburtz K, Rudolph A, Lang A, Olanow CW, Tanner C, Marek K. Levodopa and the progression of Parkinson's disease. N Engl J Med , 2004, 351(24):2498-2508. |
| Click to display the text | |
| [14] | Chaudhuri KR, Healy DG, Schapira AHV. Non-motor symptoms of Parkinson's disease:Diagnosis and management. Lancet Neurol , 2006, 5(3):235-245. |
| Click to display the text | |
| [15] | Meyer AK, Maisel M, Hermann A, Stirl K, Storch A. Restorative approaches in Parkinson's disease:Which cell type wins the race? J Neurol Sci , 2010, 289(1-2):93-103. |
| Click to display the text | |
| [16] | Kriks S, Shim JW, Piao J, Ganat YM, Wakeman DR, Xie Z, Carrillo-Reid L, Auyeung G, Antonacci C, Buch A, Yang LC, Beal MF, Surmeier DJ, Kordower JH, Tabar V, Studer L. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson's disease. Nature , 2011, 480(7378):547-551. |
| Click to display the text | |
| [17] | Han FB. The applications of the induced pluripotent stem cells in studying the neurodegenerative diseases. Chin J Cell Biol , 2012, 34(5):403-414. (in Chinese) |
| Click to display the text | |
| [18] | Fricker RA, Carpenter MK, Winkler C, Greco C, Gates MA, Björklund A. Site-specific migration and neuronal differentiation of human neural progenitor cells after transplantation in the adult rat brain. J Neurosci , 1999, 19(14):5990-6005. |
| Click to display the text | |
| [19] | Nishino H, Hida H, Takei N, Kumazaki M, Nakajima K, Baba H. Mesencephalic neural stem(progenitor) cells develop to dopaminergic neurons more strongly in dopamine-depleted striatum than in intact striatum. Exp Neurol , 2000, 164(1):209-214. |
| Click to display the text | |
| [20] | Andersson E, Tryggvason U, Deng QL, Friling S, Alekseenko Z, Robert B, Perlmann T, Ericson J. Identification of intrinsic determinants of midbrain dopamine neurons. Cell , 2006, 124(2):393-405. |
| Click to display the text | |
| [21] | Burbach JPH, Smidt MP. Molecular programming of stem cells into mesodiencephalic dopaminergic neurons. Trends Neurosci , 2006, 29(11):601-603. |
| Click to display the text | |
| [22] | Kim HJ, McMillan E, Han FB, Svendsen CN. Regionally specified human neural progenitor cells derived from the mesencephalon and forebrain undergo increased neurogenesis following overexpression of ASCL1. Stem Cells , 2009, 27(2):390-398. |
| Click to display the text | |
| [23] | Gonzalez C, Bonilla S, Flores AI, Cano E, Liste I. An update on human stem cell-based therapy in Parkinson's disease. Curr Stem Cell Res Ther, in press, DOI 10.2174/1574888X10666150531172612. |
| Click to display the text | |
| [24] | Papanikolaou T, Lennington JB, Betz A, Figueiredo C, Salamone JD, Conover JC. In vitro generation of dopaminergic neurons from adult subventricular zone neural progenitor cells. Stem Cells Dev , 2008, 17(1):157-172. |
| Click to display the text | |
| [25] | Freed CR, Breeze RE, Rosenberg NL, Schneck SA, Kriek E, Qi JX, Lone T, Zhang YB, Snyder JA, Wells TH, Ramig LO, Thompson L, Mazziotta JC, Huang SC, Grafton ST, Brooks D, Sawle G, Schroter G, Ansari AA. Survival of implanted fetal dopamine cells and neurologic improvement 12 to 46 months after transplantation for Parkinson's disease. N Engl J Med , 1992, 327(22):1549-1555. |
| Click to display the text | |
| [26] | Kordower JH, Freeman TB, Snow BJ, Vingerhoets FJ, Mufson EJ, Sanberg PR, Hauser RA, Smith DA, Nauert GM, Perl DP, Olanow CW. Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with Parkinson's disease. N Engl J Med , 1995, 332(17):1118-1124. |
| Click to display the text | |
| [27] | Lindvall O, Sawle G, Widner H, Rothwell JC, Björklund A, Brooks D, Brundin P, Frackowiak R, Marsden CD, Odin P, Rehncrona S. Evidence for long-term survival and function of dopaminergic grafts in progressive Parkinson's disease. Ann Neurol , 1994, 35(2):172-180. |
| Click to display the text | |
| [28] | Freed CR, Greene PE, Breeze RE, Tsai WY, DuMouchel W, Kao R, Dillon S, Winfield H, Culver S, Trojanowski JQ, Eidelberg D, Fahn S. Transplantation of embryonic dopamine neurons for severe Parkinson's disease. N Engl J Med , 2001, 344(10):710-719. |
| Click to display the text | |
| [29] | Lindvall O, Brundin P, Widner H, Rehncrona S, Gustavii B, Frackowiak R, Leenders KL, Sawle G, Rothwell JC, Marsden CD, Björklund A. Grafts of fetal dopamine neurons survive and improve motor function in Parkinson's disease. Science , 1990, 247(4942):574-577. |
| Click to display the text | |
| [30] | Hagell P, Brundin P. Cell survival and clinical outcome following intrastriatal transplantation in Parkinson disease. J Neuropathol Exp Neurol , 2001, 60(8):741-752. |
| Click to display the text | |
| [31] | Lindvall O, Björklund A. Cell therapy in Parkinson's disease. Neuro Rx , 2004, 1(4):382-393. |
| Click to display the text | |
| [32] | Barker RA, Barrett J, Mason SL, Björklund A. Fetal dopaminergic transplantation trials and the future of neural grafting in Parkinson's disease. Lancet Neurol , 2013, 12(1):84-91. |
| Click to display the text | |
| [33] | Svendsen CN, Caldwell MA, Shen J, ter Borg MG, Rosser AE, Tyers P, Karmiol S, Dunnett SB. Long-term survival of human central nervous system progenitor cells transplanted into a rat model of Parkinson's disease. Exp Neurol , 1997, 148(1):135-146. |
| Click to display the text | |
| [34] | Kallur T, Darsalia V, Lindvall O, Kokaia Z. Human fetal cortical and striatal neural stem cells generate region-specific neurons in vitro and differentiate extensively to neurons after intrastriatal transplantation in neonatal rats. J Neurosci Res , 2006, 84(8):1630-1644. |
| Click to display the text | |
| [35] | Yan YP, Yang DL, Zarnowska ED, Du ZW, Werbel B, Valliere C, Pearce RA, Thomson JA, Zhang SC. Directed differentiation of dopaminergic neuronal subtypes from human embryonic stem cells. Stem Cells , 2005, 23(6):781-790. |
| Click to display the text | |
| [36] | Sundberg M, Bogetofte H, Lawson T, Jansson J, Smith G, Astradsson A, Moore M, Osborn T, Cooper O, Spealman R, Hallett P, Isacson O. Improved cell therapy protocols for Parkinson's disease based on differentiation efficiency and safety of hESC-, hiPSC-, and non-human primate iPSCderived dopaminergic neurons. Stem Cells , 2013, 31(8):1548-1562. |
| Click to display the text | |
| [37] | Lu P, Wang YZ, Graham L, McHale K, Gao MY, Wu D, Brock J, Blesch A, Rosenzweig ES, Havton LA, Zheng BH, Conner JM, Marsala M, Tuszynski MH. Long-distance growth and connectivity of neural stem cells after severe spinal cord injury. Cell , 2012, 150(6):1264-1273. |
| Click to display the text | |
| [38] | Eriksson PS, Perfilieva E, Björk-Eriksson T, Alborn AM, Nordborg C, Peterson DA, Gage FH. Neurogenesis in the adult human hippocampus. Nat Med , 1998, 4(11):1313-1317. |
| Click to display the text | |
| [39] | Doetsch F, Caillé I, Lim DA, García-Verdugo JM, Alvarez-Buylla A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell , 1999, 97(6):703-716. |
| Click to display the text | |
| [40] | Taupin P, Gage FH. Adult neurogenesis and neural stem cells of the central nervous system in mammals. J Neurosci Res , 2002, 69(6):745-749. |
| Click to display the text | |
| [41] | Brundin P, Nilsson OG, Strecker RE, Lindvall O, Åstedt B, Björklund A. Behavioural effects of human fetal dopamine neurons grafted in a rat model of Parkinson's disease. Exp Brain Res , 1986, 65(1):235-240. |
| Click to display the text | |
| [42] | Studer L, Tabar V, McKay RD. Transplantation of expanded mesencephalic precursors leads to recovery in parkinsonian rats. Nat Neurosci , 1998, 1(4):290-295. |
| Click to display the text | |
| [43] | Monni E, Cusulin C, Cavallaro M, Lindvall O, Kokaia Z. Human fetal striatum-derived neural stem (NS) cells differentiate to mature neurons in vitro and in vivo. Curr Stem Cell Res Ther , 2014, 9(4):338-346. |
| Click to display the text | |
| [44] | Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, Studer L. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. NatBiotechnol , 2009, 27(3):275-280. |
| Click to display the text | |
| [45] | Yang DL, Zhang ZJ, Oldenburg M, Ayala M, Zhang SC. Human embryonic stem cell-derived dopaminergic neurons reverse functional deficit in parkinsonian rats. Stem Cells , 2008, 26(1):55-63. |
| Click to display the text | |
| [46] | Roy NS, Cleren C, Singh SK, Yang LC, Beal MF, Goldman SA. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomeraseimmortalized midbrain astrocytes. Nat Med , 2006, 12(11):1259-1268. |
| Click to display the text | |
| [47] | Björklund A, Lindvall O. Cell replacement therapies for central nervous system disorders. Nat Neurosci , 2000, 3(6):537-544. |
| Click to display the text | |
| [48] | Kang XJ, Xu HD, Teng SS, Zhang XY, Deng ZJ, Zhou L, Zuo PL, Liu B, Wu QH, Wang L, Hu MQ, Dou HQ, Liu W, Zhu FP, Li Q, Guo S, Gu JL, Lei Q, Lv J, Mu Y, Jin M, Wang SR, Jiang W, Liu K, Wang CH, Li WL, Zhang K, Zhou Z. Dopamine release from transplanted neural stem cells in Parkinsonian rat striatum in vivo. Proc Natl Acad Sci USA , 2014, 111(44):15804-15809. |
| Click to display the text |



