Objective: The conflicting findings of previous morphological studies on intracranial aneurysm rupture may be caused by the different locations of aneurysms. We aimed to determine the independent risk factors of aneurysm rupture by focusing on only posterior communicating artery (PcomA) aneurysms.
Methods: In 89 PcomA aneurysms (58 ruptured, 31 unruptured), clinical and morphological characteristics were compared between the ruptured and unruptured groups. Multivariate logistic regression analysis was performed to determine the independent predictors for the rupture status of PcomA aneurysms.
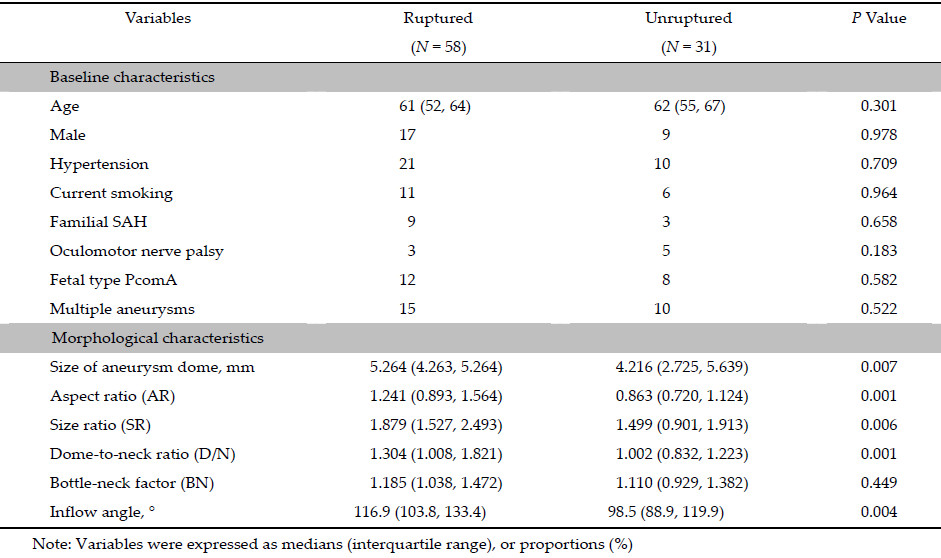
Results: In univariate analyses, the aneurysm dome size, aspect ratio, size ratio, dome-to-neck ratio, and inflow angle were significant parameters. With multivariate analyses, only the aneurysm dome size and inflow angle were significantly associated with the rupture status of PcomA aneurysms.
Conclusions: Morphology was related with rupture of PcomA aneurysms. The aneurysm dome size and inflow angle were found to be the independent parameters characterizing the rupture status of PcomA aneurysms.
1 Introduction Intracranial aneurysms (IAs) affect 7%-8% of the population according to recent studies on their natural histories. Nevertheless,the rupture rate is relatively low at approximately 1% per year[1, 2]. Although the complication rates of microsurgery and endovascular treatment have decreased significantly,clinical decisionmaking for the management of unruptured IAs remains challenging for neurosurgeons. Therefore,the accurate assessment of rupture risk is increasingly demanded. Many studies have been conducted to assess the predictors of IAs rupture. These predictors are usually broken down into three different categories consisting of clinical,morphological,and hemodynamic characteristics[3, 4]. Compared to the clinical and hemodynamic characteristics,morphological parameters are more accessible,convenient,and quantifiable features that are considered ideal for predictors of clinical rupture risk assessment. Although a wide range of morphological parameters were believed to contribute to the characterization of the risk of rupture of IAs,studies on the predictors of aneurysm rupture have conflicting results[5, 6]. This may be due to the fact that most previously published studies included aneurysms from multiple locations despite the variable and distinctive natural histories of aneurysms at different locations[7]. In order to mitigate the biases caused by the inherent differences between IAs at different locations,studies on the IAs in a single location may be more reasonable. Accounting for approximately 15%-25% of IAs cases,posterior communicating artery (PcomA) aneurysms occur frequently, and have a relatively high rupture risk[2, 8, 9]. Therefore, the determination of the morphological predictors of PcomA aneurysms rupture is considered clinically meaningful.
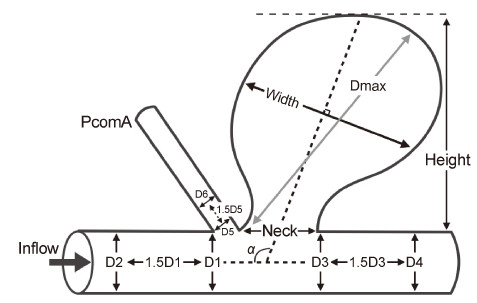
2 Materials and methods The Institution Review Board of the Second Military Medical University affiliated Changhai Hospital approved this retrospective study. The informed consent requirement was waived. 2.1 Patients and baseline characteristics Based on three‐dimensional digital subtraction angiography (3D‐DSA),89 cases of PcomA aneurysms from 81 consecutive patients who were diagnosed in our hospital from January 2012 to December 2013,were included in this study. Fifty‐eight aneurysms ruptured and presented with subarachnoid hemorrhage (SAH) while 31 cases remained unruptured. The rupture statuses of multiple aneurysms in patients with SAH were determined either from the hemorrhage patterns on computerized tomography (CT) scans or from intraoperative findings. The baseline characteristics were collected based on age and gender,as well as the presence of hypertension,current smoking,a history of familial SAH,homolateral oculomotor nerve palsy,fetal PcomA type,and multiple aneurysms. Hypertension was defined as a systolic blood pressure of ≥140 mm Hg,or a diastolic blood pressure of ≥90 mm Hg before the onset of SAH. A fetal‐type of PcomA was defined as the presence of an aneurysm with similar or larger caliber as the P2 segment of the posterior cerebral artery and is associated with an atrophic P1 segment. 2.2 Imaging acquisition and morphological parameters Three‐dimensional DSA was performed using the Artis zee Biplane angiographic system (Siemens,VC14, Germany). We adopted the 5s DSA acquisition protocol and a total of 18 mL of contrast agent was injected through the internal carotid artery at the rate of 3mL/s with a 1 second x‐ray delay. During the 5 seconds of acquisition,a 200‐degree rotation of the C‐arm was performed to obtain 133 frames. The 3D internal carotid artery vessel tree was automatically reconstructed at the syngo X Workplace (Siemens,VB15,Germany) and then exported in the stereolithography (STL) format. The GEOMAGIC STUDIO 9.0 software (Geomagic, Morrisville,North Carolina) was used for preparing the vessel structure for CFD simulation. The neck plane was defined as the location of the outward protrusion of the aneurysmal sac from the parent vessel. The 3D‐vessel was subdivided into the aneurysm,as well as the inlet and outlet planes of the parent artery,and subsequently exported in STL formats. These formats were imported into the Matlab 7.0 (The MathWorks, Inc.,Natick,MA,USA) software for visualization and calculation of the morphological parameters.In this study,seven morphological parameters,including size of the aneurysm dome,aspect ratio (AR), size ratio (SR),dome‐to‐neck ratio (D/N),bottle‐neck factor (BN) and inflow angle,were measured and calculated as previously reported[6, 10] (Figure 1).
|
| Figure 1 Definition of morphological parameters.Size=Dmax; aspect ratio=Height/Neck;size ratio=6Dmax/(D1+D2+D3+D4+ D5+D6);dome‐to‐neck ratio=Dmax/Neck;bottle‐neck factor= Width/Neck;inflow angle=α. |
Accurate assessment of the rupture risk is essential for proper clinical decision‐making in unruptured IAs. Although many studies had previously attempted to determine the predictors of IAs rupture by focusing on the clinical,morphological,or hemodynamic characteristics, some of their findings were controversial[3, 11, 12]. This may be explained by the fact that the rupture risk of IAs depends largely on their anatomical locations. Wiebers et al.[9] reviewed 4,060 patients with IAs and found that the risk of rupture of the posterior circulation and PcomA aneurysms was significantly higher than that of aneurysms in the anterior circulation. The rupture risk differences might result from various factors including the distinct thickness of the parent vessel,and surrounding anatomic structures[7]. However,most of the clinical,morphological, or hemodynamic studies focused on IAs from multiple anatomical locations. Conflicting results were reported when the location of the aneurysms was not considered[11, 12]. Hence,it may be more reasonable to perform the rupture risk analysis only on aneurysms from a single anatomical location[13, 14]. One of the most frequent types of IAs,PcomAs account for approximately 15%-25% of all cases[15]. According to a recent Japanese study on the natural history of unruptured aneurysms,the risk of PcomA aneurysm rupture is relatively higher at 1.72% per year[2]. Therefore,we focused only on PcomA aneurysms to determine the morphological predictors of rupture in this study.
Clinical characteristics were recognized as important factors affecting the risk of rupture in aneurysms[4]. In this study,the clinical characteristics were balanced between the ruptured and unruptured groups,with no significant observed differences. This could be helpful in eliminating patient selection bias during morphological analysis.
An increasing number of morphological parameters have been involved in rupture risk determination in aneurysms[6, 10]. The size of the aneurysm dome was believed to be a significant parameter for predicting the rupture risk of aneurysms. Size was an independent risk factor of PcomA aneurysms rupture in our study. According to the International Study of Unruptured Intracranial Aneurysms (ISUIA),small aneurysms have a relatively low risk of rupture[9]. Nevertheless,in a study by Forget et al.[16],85.6% of ruptured aneurysms were small aneurysms. In a separate study,PcomA aneurysms exceeding 7 mm were not significantly more common in patients with SAH[14]. These conflicting findings might suggest that size alone cannot characterize the rupture risk of IAs.
Therefore,this study also introduced other recently recognized morphological parameters,including AR, SR,D/N,BN,and inflow angle[6]. In the univariate analysis,AR,SR,and D/N were significantly more common in the ruptured group. However,these variables were not retained in the multivariate logistic regression model for rupture risk assessment. This result indicated that these parameters might be closely related to the size of the aneurysm dome.
Unlike the other included parameters (i.e.,AR and SR) that focused on the geometry of the aneurysm dome itself,inflow angle reflected the relationship between the aneurysm dome and parent vessels[5, 17]. Our study found that the inflow angle was an independent predictor of the risk of rupture of PcomA aneurysms,which was consistent with several previous studies[17, 18]. Previous hemodynamic studies revealed that the hemodynamic pattern inside the aneurysm dome is partially determined by the spatial relationships between the aneurysm dome and its parent vessels [17]. More direct flow into the aneurysm dome increases the flow velocity and the size of the shear jet zone,which might be important factors for increased rupture risk. Xiang et al.[13] also obtained several logistic regression models based on the morphological‐hemodynamic analysis of 119 IAs from different locations. Although wall shear stress and oscillatory shear index were also in the hemodynamic model,only SR was retained in the morphological model. Since hemodynamic calculations are a timeconsuming process,they may have limited use in timely clinical decision‐making. Therefore,morphological parameters may be better at rapidly predicting the risk of rupture of IAs in the clinical setting. The stability and efficiency of the logistic regression model in this study will be further tested in future research.
4.1 Limitations The study has several limitations including the small sample size,which is a major limitation in this retrospective study. Additionally,the morphology of PcomA aneurysms might change after rupture and affect the accuracy of measurements. Finally,the logistic regression model was developed only for PcomA aneurysms,and cannot be applied to aneurysms at other anatomic locations. 5 Conclusions Morphological characteristics are involved in the risk of rupture of PcomA aneurysms. The size of the aneurysm dome and inflow angle are independent predictors of the risk rupture of PcomA aneurysms. Acknowledgments This work was supported by the Key Project of Shanghai Science and Technology Committee (No. 13411950300),the National Natural Science Foundation of China (No. 81301004),the Key Project of Shanghai Municipal Health Bureau (No. 2013ZYJB007) and the 1255 Project of Changhai Hospital (No. CH125550400). We gratefully acknowledge the Shanghai Supercomputer Center for providing the mathematical soft wares that were used in this study. Conflict of interests The authors have no financial interest to disclose regarding the article. Author Contributions Conceived and designed the experiments: NL,QH. Performed the experiments: NL,YF,YY. Analyzed the data: JX JL. Wrote the paper: NL,QH.| [1] | Korja M, Lehto H, Juvela S. Lifelong rupture risk of intracranial aneurysms depends on risk factors:A prospective Finnish cohort study. Stroke , 2014, 45(7):1958-1963. |
| Click to display the text | |
| [2] | Morita A, Kirino T, Hashi K, Aoki N, Fukuhara S, Hashimoto N, Takeo N, Sakai M, Teramoto A, Tominari S, Yoshimoto T. The natural course of unruptured cerebral aneurysms in a Japanese cohort. N Engl J Med , 2012, 366(26):2474-2482. |
| Click to display the text | |
| [3] | Bor ASE, Tiel Groenestege AT, terBrugge KG, Agid R, Velthuis BK, Rinkel GJE, Wermer MJH. Clinical, radiological, and flow-related risk factors for growth of untreated, unruptured intracranial aneurysms. Stroke , 2015, 46(1):42-48. |
| Click to display the text | |
| [4] | Greving JP, Wermer MJH, Brown RDJr, Morita A, Juvela S, Yonekura M, Ishibashi T, Torner JC, Nakayama T, Rinkel GJE, Algra A. Development of the PHASES score for prediction of risk of rupture of intracranial aneurysms:A pooled analysis of six prospective cohort studies. Lancet Neurol , 2014, 13(1):59-66. |
| Click to display the text | |
| [5] | Ujiie H, Tamano Y, Sasaki K, Hori T. Is the aspect ratio a reliable index for predicting the rupture of a saccular aneurysm? Neurosurgery , 2001, 48(3):495-502. |
| Click to display the text | |
| [6] | Zanaty M, Chalouhi N, Tjoumakaris SI, Fernando GL, Rosenwasser RH, Jabbour PM. Aneurysm geometry in predicting the risk of rupture. A review of the literature. Neurol Res , 2014, 36(4):308-313. |
| Click to display the text | |
| [7] | San Millán Ruíz D, Yilmaz H, Dehdashti AR, Alimenti A, de Tribolet N, Rüfenacht DA. The perianeurysmal environment:Influence on saccular aneurysm shape and rupture. AJNR Am J Neuroradiol , 2006, 27(3):504-512. |
| [8] | Golshani K, Ferrell A, Zomorodi A, Smith TP, Britz GW. A review of the management of posterior communicating artery aneurysms in the modern era. Surg Neurol Int , 2010, 1:88. |
| Click to display the text | |
| [9] | Wiebers DO, Whisnant JP, Huston J 3rd, Meissner I, Brown RD Jr, Piepgras DG, Forbes GS, Thielen K, Nichols D, O'Fallon WM, Peacock J, Jaeger L, Kassell NF, Kongable Beckman GL, Torner JC; International Study of Unruptured Intracranial Aneurysms gators. Unruptured intracranial aneurysms:Natural history, clinical outcome, and risks of surgical and endovascular treatment. Lancet , 2003, 362(9378):103-110. |
| Click to display the text | |
| [10] | Dhar S, Tremmel M, Mocco J, Kim M, Yamamoto J, Siddiqui AH, Hopkins LN, Meng H. Morphology parameters for intracranial aneurysm rupture risk assessment. Neurosurgery , 2008, 63(2):185-196. |
| Click to display the text | |
| [11] | Cebral JR, Mut F, Weir J, Putman C. Quantitative characterization of the hemodynamic environment in ruptured and unruptured brain aneurysms. AJNR Am J Neuroradiol , 2011, 32(1):145-151. |
| Click to display the text | |
| [12] | Xiang JP, Natarajan SK, Tremmel M, Ma D, Mocco J, Hopkins LN, Siddiqui AH, Levy EI, Meng H. Hemodynamicmorphologic discriminants for intracranial aneurysm rupture. Stroke , 2011, 42(1):144-152. |
| Click to display the text | |
| [13] | Miura Y, Ishida F, Umeda Y, Tanemura H, Suzuki H, Matsushima S, Shimosaka S, Taki W. Low wall shear stress is independently associated with the rupture status of middle cerebral artery aneurysms. Stroke , 2013, 44(2):519-521. |
| Click to display the text | |
| [14] | Matsukawa H, Fujii M, Akaike G, Uemura A, Takahashi O, Niimi Y, Shinoda M. Morphological and clinical risk factors for posterior communicating artery aneurysm rupture. J Neurosurg , 2014, 120(1):104-110. |
| Click to display the text | |
| [15] | Vlak MHM, Algra A, Brandenburg R, Rinkel GJE. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period:A systematic review and meta-analysis. Lancet Neurol , 2011, 10(7):626-636. |
| Click to display the text | |
| [16] | Forget TRJr, Benitez R, Veznedaroglu E, Sharan A, Mitchell W, Silva M, Rosenwasser RH. A review of size and location of ruptured intracranial aneurysms. Neurosurgery , 2001, 49(6):1322-1325. |
| Click to display the text | |
| [17] | Baharoglu MI, Schirmer CM, Hoit DA, Gao BL, Malek AM. Aneurysm inflow-angle as a discriminant for rupture in sidewall cerebral aneurysms:Morphometric and computational fluid dynamic analysis. Stroke , 2010, 41(7):1423-1430. |
| Click to display the text | |
| [18] | Tykocki T, Nauman P, Dow enko A. Morphometric predictors of posterior circulation aneurysms risk rupture. Neurol Res , 2014, 36(8):733-738. |
| Click to display the text |