2. Center for Neuroscience and Cell Biology, Universidade de Coimbra, Faculdade de Medicina, Rua Larga, Pólo I, Coimbra 3004-504, Portugal;
3. Centro de Biologia Ambiental, Faculdade de Ciências da Universidade de Lisboa, Edifício C2, Campo Grande, Lisbon 1749-016, Portugal;
4. Department of Human Anatomy, Moscow State Medical University, Moscow 125009, Russia;
5. Centro de Estudos de Doenças Crónicas(CEDOC), Edifício CEDOC II, Rua Câmara Pestana, no6-6 A, Lisboa, Lisbon 1150-082, Portugal;
6. Centro Interdisciplinar de Investigação em Saúde Animal(CIISA), Faculdade de Medicina Veterinária, Alameda da Universidade Técnica, Lisboa, Lisbon 1300-477, Portugal
The development of novel and powerful therapeutics for patients suffering from neuropsychiatric disorders has raised important concerns regarding the efficacy of current preclinical approaches[1-3], many of which have been intensively discussed in the last decade with regard to their limitations in available models[4-7].
The major concern with the use of small rodents as preclinical models has been their translational validity. In the last decade, a growing body of evidence has demonstrated the ability for a vast diversity of experi-mental conditions to induce potential confounds for the practical application of animal models, which is believed to result in the noteworthy variability of results[8-11]. Some of the pivotal sources of such con-founds are considered to include the circadian phase of manipulations[12], housing conditions[13-15], the role of the experimenter[16-18], and the delivery method, formulation, dosing, and duration of treatment[8, 11, 19, 20].
Regardless of their adequate use, various types of invasive treatments in rodents are reported to evoke distress, inflammation, and pain, predominantly when long-lasting dosing schemes are employed[21-23]. Nonetheless, in many cases it is methodologically challenging to avoid prolonged and invasive drug administration, especially when non-water soluble compounds have to be chronically administered and/or an extended period of time is required to observe its therapeutic effect. These experimental challenges are frequent when testing pharmacologically active compounds in rodent models of depression, where the onset of symptoms[24-26] and antidepressant-like effects[9] may take weeks to develop.
In pre-clinical research, antidepressant treatment is often delivered intraperitoneally[27-32] or subcutaneously[33], while other approaches are applied less frequently[34-36]. However, the oral administration of antidepressant-like treatment and other pharmacological drug candidates is also effective for small rodents[7]. Moreover, the negative effects of chronic invasive treatment are circumvented, providing greater translational value by mimicking a clinical setting of drug intake by patients[37].
In this study we used a low dose of imipramine, a reference drug used for translational research in depression, delivered with food pellets or in drinking water, to assess the efficacy of these two dosing methods, against those that have been previously established[7, 38].
2 Methods 2.1 Animals and housingThree-month-old C57BL/6N male mice were supplied by Instituto Gulbenkian de Ciência, Oeiras, Portugal, and housed individually in standard laboratory conditions under a reverse 12 h:12 h cycle (lights on at 21:00). Behavioral tests were conducted from the onset of the dark phase of the light cycle (9:00) in a dark, quiet room during the morning hours. All procedures were in accordance to the European Union’s Directive 2010/63/EU, Portuguese law Law-Decrees DL129/92 (July 6th), DL197/96 (October 16th), and Ordinance Port.131/97 (November 7th). This project was approved by the Ethical Committee of the Science Faculty of the University of Lisbon.
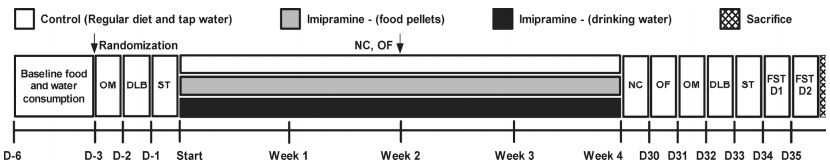
2.2 Study flowPrior to starting treatment, animals were weighed and grouped so that the body weights in each group were balanced. Naïve mice were exposed for 4 weeks to imipramine that were contained in self-made food pellets (Imi-food group) or dissolved in tap water (Imi-drink group). The concentration of imipramine in food pellets was calculated based on the daily food intake of the experimental mice, which constituted 4.07±0.48 g. For animals receiving treatment in drinking water, imipramine was dissolved normally and calculated based on a daily water intake of 4.53±1.56 g. The selection of a desirable dosage of 7 mg/kg/day was based on previous observation of undesirable side effects with higher doses[6]. Control mice received a regular diet and normal drinking water. Before the start and after 4 weeks of dosing, all mice were tested in the sucrose preference test, O-maze test, and in the dark/light box, as previously reported[7, 39, 40]. Locomotor activity of all mice was studied in the novel cage and open field tests, as previously reported[7, 40-42], after 2 and 4 weeks of dosing. At the end of behavioral testing, a 2-day forced swim test with 6-min sessions was performed as previously reported[38, 42] (see Figure 1 for a scheme of the study flow). The number of mice per group is indicated in each Figure legends.
|
| Figure 1 Study flow. Time is expressed in days, relative to treatment period (D). Behavioral tests: OM—O-maze; DLB—Dark/light box; ST—Sucrose test; NC—Novel cage; OF—Open field; FST—Forced swim test. Number of animals: Control, n=7; Imipramine (food), n=8; Imipramine (water), n=8. |
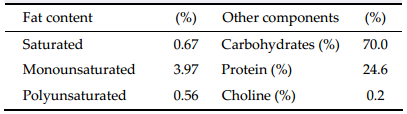
Food pellets were blended to powder and Imipramine Hydrochloride (Sigma-Aldrich, Munich, Germany) was added, adjusted to the dose of mg/kg/day, based on the consumption of normal diet (see Table 1 for composition), which was averaged over 3 days, as previously reported[7]. Small amounts of distilled water were added to allow food pellets to be shaped to a similar size of those available commercially, then dried overnight (16 h) at 60 °C. Fresh pellets were produced twice a week to restock food supply.
Forced swim test. The Porsolt forced swim test has been used as previously reported[38, 42]. Mice were subjected to two 6-min-swimming sessions spaced 24 h apart in a transparent cylinder (17 cm in diameter) filled with water (+23 °C, water height 13 cm, height of cylinder 20 cm, illumination intensity 25 Lux). Floating behavior, defined by the absence of any directed movements of the animal’s head and body, was scored manually by an experienced observer who was blinded to the treatment protocol. Using a previously validated automated scoring method, the latency of the first episode of floating and the duration of floating behavior were recorded during the 6-min-swimming session, on day 1 and day 2 of the test. Latency to begin floating was scored as time between introduction of the animal into the pool and the first moment of complete immobility of the entire body for a duration of > 3 seconds. The total time spent floating and number of floating episodes were scored.
Dark/light box. The dark/light box (Technosmart, Rome, Italy) consisted of 2 plexiglass compartments connected by a tunnel, one of which was black/dark (15 cm × 20 cm × 25 cm) and the other was lit (30 cm × 20 cm × 25 cm). Anxiety-like behavior was assessed by measures that have been previously validated[39, 42]. Mice were placed into the dark compartment, from which they could visit the lit box, which was illuminated by a 25-Lux intensity light. The latency of the first exit to the light compartment, the total duration of time spent in the lit box, and the number of visits to this anxiety-related compartment were scored by visual observation over 5 min.
Elevated O-maze. The apparatus (Technosmart, Rome, Italy), which consisted of a circular path (runway width 5.5 cm, diameter 46 cm), was placed 50 cm above the floor. Two opposing arms were protected by walls (height 10 cm), and the illumination strength was 25 Lux. The apparatus was placed on a dark surface in order to reduce reflection and maintain control over lighting conditions during testing. Anxiety-like behavior was assessed using previously validated parameters[7, 38, 40]. Mice were placed in one of the closed-arm compartments of the apparatus. The latency of the first exit to the anxiety-related open compartments of the maze, the total duration of time spent therein, and the number of exits to the open arms were scored during an observation period of 5 min.
Novel cage test. The novel cage test was performed to assess vertical activity, as previously reported[38]. Mice were introduced into a standard plastic cage (the size of their home cage) filled with small amounts of fresh sawdust. The number of exploratory rearings was counted under red light during a 5-min period.
Open field. The open-field apparatus consisted of 2 square arenas (50 cm × 50 cm × 50 cm), made of wood that were covered by white resopal. Mice were put in the center, and their behavior was recorded on camera for 10 min. The open field was illuminated with white light (25 Lux). The distanced moved, mean instant speed, and number of line crossings were analyzed off-line using the Any-maze software (Stoelting Co, Wood Dale, IL, USA), as previously reported[43].
Sucrose test. Animals were given 8 hours of free choice between 2 bottles of either 1% sucrose or normal drinking water, as previously reported[11]. The bottles were weighed at the beginning and at the end of the period, and consumption calculated. The beginning of the test started with the onset of the dark (active) phase of animals’ cycle. To prevent possible effects of side-preference in drinking behavior, the position of the bottles in the cage was switched halfway through testing. No previous food or water deprivation was applied before the test. The 1% sucrose solution was used in tests performed across the experiment. The percentage preference for sucrose was calculated using the following formula:
| ${\rm{Sucrose Preference = }}\left( {\frac{{{V_{{\rm{Sucrose solution}}}}}}{{{V_{{\rm{Sucrose solution}}}} + {V_{{\rm{Water}}}}}}} \right) \times 100\% $ |
Data were analyzed with GraphPad Prism versions 5.00 for Windows (San Diego, CA, USA). Data were validated for normality, upon which a one-way ANOVA was used followed by a post-hoc Bonferroni test for the comparison of more than two groups with a control. The level of confidence was set at 95% (P < 0.05) and data are shown as mean±standard error of mean (SEM).
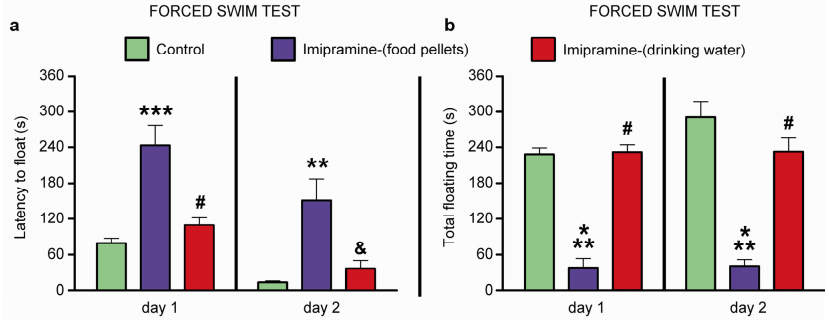
3 Results and discussion 3.1 Antidepressant-like effects of imipramine in the forced swim testIn order to assess the efficacy of an alternative chronic dosing design, we evaluated the effects of the 4-week treatment of imipramine, which were delivered via food pellets and drinking water, in the forced swim test. In comparison to controls, animals subjected to imipramine treatment via food, but not via water, exhibited a significant increase in the latency to float (Day 1: P < 0.0001, F=18.15, t=5.691 and P < 0.001, Imifood vs. Control; Day 2: P=0.0012, F=9.981, t=4.071 and P < 0.01, Imi-food vs. Control, respectively; one-way ANOVA and Bonferroni post-hoc test, Figure 2a), and a decrease in the immobility time (Day 1: P < 0.0001, F=23.53, t=5.181 and P < 0.001, Imi-food vs. Control;
|
| Figure 2 Antidepressant-like effects of imipramine in the forced swim test. In comparison with control group and animals treated with imipramine in drinking water, mice that received imipramine in food pellets exhibit increased latency to float (a) and reduced total duration of floating (b). *P < 0.05 vs control; **P < 0.01 vs control; ***P < 0.001 vs control; & P < 0.01 vs. imi-food or imi-drink; #P < 0.001 vs imi-food or imi-drink; aP < 0.05 vs baseline. Data are represented as mean±standard error of mean (SEM). Number of animals: Control, n=7; Imipramine (food), n=8; Imipramine (water), n=8. |
These results corroborate our recently published work, where the delivery of a low dose of imipramine with food pellets exerted antidepressant-like effects in the forced swim test in C57BL/6N mice[7]. However, when treatment was provided with water, these effects were not significant. As reported by Cline et al.[38], a similar 3-week protocol reduced depressive symptoms such as stress-induced decease in sucrose intake and preference, hyperlocomotion, and elevated aggressive behavior, similar to previous results in a model of elderly depression in 18-month-old C57BL/6N mice[42], or those in the chronic stress depression model with CD1 mice[41]. These differences could potentially be attributed to differential involvement of the sympathetic nervous system in these different conditions. It is proposed that in naïve animals imipramine activates sympathetic tone, which is altered during stress. Besides, since the metabolic rate is enhanced by stress-induced sympathetic activation, the clearance of imipramine in stressed animals is higher, which can also explain the variance of results in the studies discussed above[43].
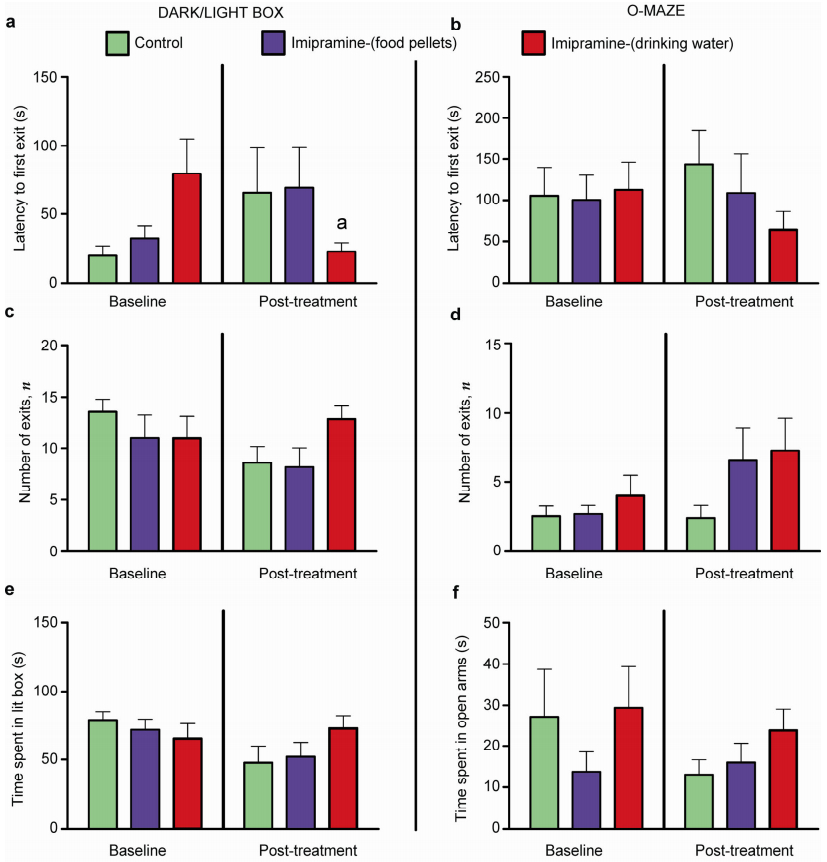
3.2 Effects of imipramine in tests for anxiety-like behaviorIn order to study potential effects of imipramine administration on anxiety-like behaviors, we performed experiments using classical paradigms of anxiety. In both dark/light box (DL) and O-maze (OM) tests, animals receiving treatment in food pellets and water showed no significant differences between groups for latency (DL baseline: P=0.2404, F=2.851; DL posttreatment: P=0.6678, F=0.8076, respectively Figure 3a; OM baseline: P=0.9988, F=0.01177; OM post-treatment: P=0.3396, F=1.144, respectively Figure 3b; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test), number of exits (DL baseline: P=0.5948, F=0.5333; DL post-treatment: P=0.0822, F=2.859, respectively Figure 3c; OM baseline: P=0.5782, F=0.5640; OM posttreatment: P=0.2299, F=1.590, respectively Figure 3d; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test), or time spent in exposed area (DL baseline: P=0.4307, F=0.8831; DL post-treatment, respectively Figure 3e: P=0.2853, F=1.352; OM baseline: P=0.0591, F=3.295; OM post-treatment: P=0.3749, F=1.031, respectively Figure 3f; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test).
|
| Figure 3 Effects of imipramine on anxiety-like behaviour. No significant differences were found in latency (a, b), number of exits (c, d), or time spent in exposed area (e, f) in both dark/light box and O-maze, respectively. aP < 0.05 vs same group, t test. Data are represented as mean±standard error of mean (SEM). Number of animals: Control, n=7; Imipramine (food), n=8; Imipramine (water), n=8. |
Thus, either way of dosing with low dose of imipra-mine did not alter anxiety-like behavior in mice in the present study. While tricyclics are known to reduce anxiety in humans[44] and animals[45, 46], some preclinical studies report a contradiction in anxiety-related behavioral changes in rodents treated by compounds of this class[47-50]. A lack of such effects in current work can be attributed to the low dose of imipramine that was used. Conversely, the use of this dose in models of depression enables the discrimination between anti-anxiety and anti-depressant changes, which frequently overlap.
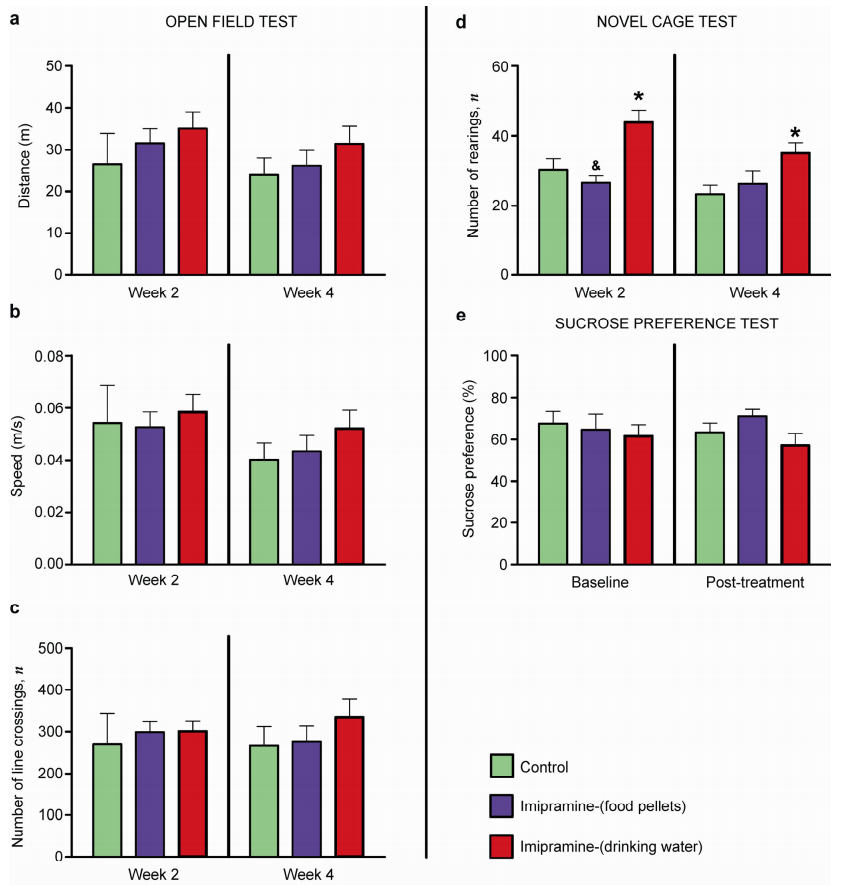
3.3 Effects of imipramine on basal physiological parametersIn order to study the general effects of imipramine administration on basal physiological parameters, the following parameters were evaluated: locomotion, liquid intake, novel cage, O-maze, and sucrose preference. In the open field, no difference between groups was found in distance travelled (week 2: P=0.8313, F=0.1866; week 4: P=0.3993, F=0.9640, respectively Figure 4a; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test), mean instant velocity (week 2: P=0.9036, F=0.1019; week 4: P=3936, F=0.9796, respectively Figure 4b; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test), or number of line crossings in the open field (week 2: P=0.9459, F=0.05156; week 4: P=0.4423, F=0.8517, respectively Figure 4c; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test).
|
| Figure 4 Effects of imipramine on basal parameters of locomotor activity. In the open field test, no differences were found in distance travelled (a), average speed of locomotion (b), or number of line crossings (c) between groups. Increased number of rearings is found in the novel cage test for animals receiving treatment in water, when compared to a control group (d). Sucrose preference is not different between control and imipramine-treated groups (e). Data are represented as mean±standard error of mean (SEM). Number of animals: Control, n=7; Imipramine (food), n=8; Imipramine (water), n=8. |
In the novel cage test, animals treated with imipramine via food exhibited normal vertical activity, whilst those receiving it via water had a significantly increased number of rearings (Week 2: P=0.0013, F=9.575, t=3.214 vs. Control, t=4.126 vs. Imi-food; Week 4: P=0.0348, F=4.027, t=2.695 vs. Control, respectively Figure 4d, ANOVA and Bonferroni post-hoc test). This suggests that the increased exploratory behavior, rather than altered locomotion, in the latter group may indicate anxiolytic-like changes as no differences were observed in an open field arena, as described above.
In a 2-bottle sucrose preference test, there were no significant differences in water intake (baseline: P=0.9444, F=0.05741; post-treatment: P=0.2337, F=1.571; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test, respectively, data not shown), sucrose solution intake (baseline: P=0.3344, F=1.161; post-treatment: P=0.0613, F=3.246; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test, respectively, data not shown), and sucrose preference between the groups (baseline: P=0.7932, F=0.2345; post-treatment: P=0.1485, F=2.111, respectively Figure 4e; P > 0.05 between groups, ANOVA and Bonferroni post-hoc test).
The absence of changes in general locomotion and solution intake or preference found in animals treated with imipramine, via either route of dosing, suggests a lack of basal physiological and behavioral responses, which can be perceived as undesirable side-effects. Together, our observations support the potential usefulness of imipramine in preclinical models of depression, since it avoids potential confounds related to a depressive state.
3.4 Efficacy of Imipramine treatmentAs seen above, our data suggest the distinct effects of a selected dose of imipramine that was selfadministrated via food pellets and drinking water to naïve. It may be argued that animals might have altered their baseline patterns of food or water intake during the course of the experiment, and therefore dosing. However, the daily weighing of pellets and bottles revealed similar intakes on a day-to-day basis (not shown). Moreover, previous reports suggest that food ingestion has no effect on the absolute bioavailability, peak concentration attained after oral dosage, or the time to peak concentration of imipramine[51]. However, it can be claimed that 7 mg/kg/day is a very low dose in naïve animals, requiring the presence of stress for its effects to be seen when delivered with drinking water[39]. This will be addressed in follow-up studies using a stress model of depression.
4 ConclusionsOur data suggest that the administration of phar-macologically active compounds by oral route may be considered an effective method for chronic dosing. The results from the current study are in line with other reports, where antidepressants were delivered via food or dissolved in drinking water[7, 38]. This dosing method could be mostly useful whenever long-lasting drug administration is required, with particular benefit for stressed, operated, or immunodeficient laboratory animals, not only by minimizing suffering and associated risks.
Although drug delivery with voluntary consumed food or liquids is an established method of dosing, its use in pre-clinical research is currently limited. Although in many cases this approach is seen beneficial since it enables the maintenance of a steady blood concentration for the drug, in contrast to bolus drug administration; this method is sometimes regarded as uncontrollable due to its dependence on actual food intake and the variable bioavailability of some compounds depending on their delivery route[52, 53]. Meanwhile, the consummatory behavior of laboratory
animals is usually not very variable and often not modified by the pharmaca. The bioavailability and metabolism of many investigational drugs are well known, and have a good compatibility with the oral dosing route. Thus, their dosing with the voluntary intake of food pellets or drinking water can probably be exploited more frequently.
In experimental paradigms of depression, the overlap between anxiolytic-like effects and antidepressant effects[54-57] may potentially confound the assessment of antidepressant-like effects of investigational drugs in translational research. In this regard, our study offers a possible dosing/testing scheme that may enable dissection of these 2 types of effects by using a low dose of drugs with an antidepressant activity. In addition, our current results suggest that a low dose of imipramine delivered orally with food may preclude overlapping antidepressant and anxiolytic effects, which were shown for other schemes of drug administration with higher doses and other delivery routes. Thus, these data are beneficial for implementation in future pre-clinical experimental design in a field of depression.
Follow-up experiments have been designed to tackle some of the limitations of the present work. We plan to use a stress model of depression to address the effects of voluntary oral intake of imipramine and other pharmacologically active compounds at various dosages on behavioral, molecular, biochemical, and immunological processes.
AcknowledgementsWe would like to thank Dr. Cláudia Oliveira from the CBA and Mrs. Margarida Rama for technical support.
Conflict of interestsThe authors have no financial interest to disclose regarding the article.
| [1] | Munos B. Lessons from 60 years of pharmaceutical innova-tion. Nat Rev Drug Discov , 2009, 8 (12) : 959–968. DOI:10.1038/nrd2961 |
| [2] | Pigott HE, Leventhal AM, Alter GS, Boren JJ. Efficacy and effectiveness of antidepressants:Current status of research. Psychother Psychosom , 2010, 79 (5) : 267–279. DOI:10.1159/000318293 |
| [3] | Araragi N, Lesch KP. Serotonin (5-HT) in the regulation of depression-related emotionality:Insight from 5-HT transporter and tryptophan hydroxylase-2 knockout mouse models. Curr Drug Targets , 2013, 14 (5) : 549–570. DOI:10.2174/1389450111314050005 |
| [4] | Insel TR. From animal models to model animals. Biol Psychiatry , 2007, 62 (12) : 1337–1339. DOI:10.1016/j.biopsych.2007.10.001 |
| [5] | Insel TR, Sahakian BJ, Voon V, Nye J, Brown VJ, Altevogt BM, Bullmore ET, Goodwin GM, Howard RJ, Kupfer DJ, et al. Drug research:A plan for mental illness. Nature , 2012, 483 (7389) : 269. DOI:10.1038/483269a |
| [6] | Strekalova T, Anthony DC, Dolgov O, Anokhin K, Kubatiev A, Steinbusch HMW, Schroeter C. The differential effects of chronic imipramine or citalopram administration on physiological and behavioral outcomes in naïve mice. Behav Brain Res , 2013, 245 : 101–106. DOI:10.1016/j.bbr.2013.02.015 |
| [7] | Costa-Nunes JP, Cline BH, Araújo-Correia M, Valença A, Markova N, Dolgov O, Kubatiev A, Yeritsyan N, Steinbusch HW, Strekalova T. Animal models of depression and drug delivery with food as an effective dosing method:Evidences from studies with Celecoxib and Dicholine succinate. Biomed Res Int , 2015, 2015 : 596126. |
| [8] | Strekalova TV, Cespuglio R, Koval'zon VM. Depressive-like state and sleep in laboratory mice. Zh Vyssh Nerv Deiat Im I P Pavlova , 2008, 58 (6) : 728–737. |
| [9] | Strekalova T, Couch Y, Kholod N, Boyks M, Malin D, Leprince P, Steinbusch HMW. Update in the methodology of the chronic stress paradigm:Internal control matters. Behav Brain Funct , 2011, 7 : 9. DOI:10.1186/1744-9081-7-9 |
| [10] | Strekalova T, Steinbusch H. Factors of reproducibility of anhedonia induction in a chronic stress depression model in mice. In:Mood and Anxiety Related Phenotypes in Mice:Characterization Using Behavioral Tests. Gould TD, Ed. New Jersey:Humana Press, 2009, pp 153-176. |
| [11] | Strekalova T, Steinbusch HWM. Measuring behavior in mice with chronic stress depression paradigm. Prog Neuropsychopharmacol Biol Psychiatry , 2010, 34 (2) : 348–361. DOI:10.1016/j.pnpbp.2009.12.014 |
| [12] | Sato Y, Seo N, Kobahashi E. The dosing-time dependent effects of intravenous hypnotics in mice. Anesth Analg , 2005, 101 (6) : 1706–1708. |
| [13] | Munn E, Bunning M, Prada S, Bohlen M, Crabbe JC, Wahlsten D. Reversed light-dark cycle and cage enrichment effects on ethanol-induced deficits in motor coordination assessed in inbred mouse strains with a compact battery of refined tests. Behav Brain Res , 2011, 224 (2) : 259–271. DOI:10.1016/j.bbr.2011.05.030 |
| [14] | Strekalova T, Spanagel R, Dolgov O, Bartsch D. Stress-induced hyperlocomotion as a confounding factor in anxiety and depression models in mice. Behav Pharmacol , 2005, 16 (3) : 171–180. DOI:10.1097/00008877-200505000-00006 |
| [15] | Ringgold KM, Barf RP, George A, Sutton BC, Opp MR. Prolonged sleep fragmentation of mice exacerbates febrile responses to lipopolysaccharide. J Neurosci Methods , 2013, 219 (1) : 104–112. DOI:10.1016/j.jneumeth.2013.07.008 |
| [16] | Cloutier S, Wahl K, Baker C, Newberry RC. The social buffering effect of playful handling on responses to repeated intraperitoneal injections in laboratory rats. J Am Assoc Lab Anim Sci , 2014, 53 (2) : 168–173. |
| [17] | Bohlen M, Hayes ER, Bohlen B, Bailoo JD, Crabbe JC, Wahlsten D. Experimenter effects on behavioral test scores of eight inbred mouse strains under the influence of ethanol. Behav Brain Res , 2014, 272 : 46–54. DOI:10.1016/j.bbr.2014.06.017 |
| [18] | Sorge RE, Martin LJ, Isbester KA, Sotocinal SG, Rosen S, Tuttle AH, Wieskopf JS, Acland EL, Dokova A, Kadoura B, et al. Olfactory exposure to males, including men, causes stress and related analgesia in rodents. Nat Methods , 2014, 11 (6) : 629–632. DOI:10.1038/nmeth.2935 |
| [19] | Sousa N, Almeida OF, Wotjak CT. A hitchhiker's guide to behavioral analysis in laboratory rodents. Genes Brain Behav , 2006, 5 Suppl 2 : 5–24. |
| [20] | Azar TA, Sharp JL, Lawson DM. Heart rates of male and female Sprague-Dawley and spontaneously hypertensive rats housed singly or in groups. J Am Assoc Lab Anim Sci , 2011, 50 (2) : 175–184. |
| [21] | Gärtner K, Büttner D, Döhler K, Friedel R, Lindena J, Trautschold I. Stress response of rats to handling and experimental procedures. Lab Anim , 1980, 14 (3) : 267–274. DOI:10.1258/002367780780937454 |
| [22] | Cloutier S, Newberry RC. Use of a conditioning technique to reduce stress associated with repeated intra-peritoneal injections in laboratory rats. Appl Anim Behav Sci , 2008, 112 (1-2) : 158–173. DOI:10.1016/j.applanim.2007.07.003 |
| [23] | Thiele TE, Navarro M. "Drinking in the dark" (DID) procedures:A model of binge-like ethanol drinking in non-dependent mice. Alcohol , 2014, 48 (3) : 235–241. DOI:10.1016/j.alcohol.2013.08.005 |
| [24] | Machado-Vieira R, Baumann J, Wheeler-Castillo C, Latov D, Henter ID, Salvadore G, Zarate CA Jr. The timing of antidepressant effects:A comparison of diverse phar-macological and somatic treatments. Pharmaceuticals (Basel) , 2010, 3 (1) : 19–41. DOI:10.3390/ph3010019 |
| [25] | Yan HC, Cao X, Das M, Zhu XH, Gao TM. Behavioral animal models of depression. Neurosci Bull , 2010, 26 (4) : 327–337. DOI:10.1007/s12264-010-0323-7 |
| [26] | Krishnan V, Nestler EJ. Animal models of depression:Molecular perspectives. In:Molecular and Functional Models in Neuropsychiatry. Hagan JJ, Ed. Berlin Heidelberg:Springer, 2011, pp 121-147. |
| [27] | Steru L, Chermat R, Thierry B, Simon P. The tail suspension test:A new method for screening antidepressants in mice. Psychopharmacology (Berl) , 1985, 85 (3) : 367–370. DOI:10.1007/BF00428203 |
| [28] | Steru L, Chermat R, Thierry B, Mico JA, Lenegre A, Steru M, Simon P, Porsolt RD. The automated Tail Suspension Test:A computerized device which differentiates psychotropic drugs. Prog Neuropsychopharmacol Biol Psychiatry , 1987, 11 (6) : 659–671. |
| [29] | Teste JF, Pelsy-Johann I, Decelle T, Boulu RG. Anti-immobility activity of different antidepressant drugs using the tail suspension test in normal or reserpinized mice. Fundam Clin Pharmacol , 1993, 7 (5) : 219–226. DOI:10.1111/fcp.1993.7.issue-5 |
| [30] | Bai FJ, Li X, Clay M, Lindstrom T, Skolnick P. Intra-and interstrain differences in models of "behavioral despair". Pharmacol Biochem Behav , 2001, 70 (2-3) : 187–192. DOI:10.1016/S0091-3057(01)00599-8 |
| [31] | Castagné V, Porsolt RD, Moser P. Early behavioral screening for antidepressants and anxiolytics. Drug Dev Res , 2006, 67 (9) : 729–742. DOI:10.1002/(ISSN)1098-2299 |
| [32] | Castagné V, Porsolt RD, Moser P. Use of latency to immobility improves detection of antidepressant-like activity in the behavioral despair test in the mouse. Eur J Pharmacol , 2009, 616 (1-3) : 128–133. DOI:10.1016/j.ejphar.2009.06.018 |
| [33] | O'Neill MF, Fernández AG, Palacios JM. GR, 127935 blocks the locomotor and antidepressant-like effects of RU, 24969 and the action of antidepressants in the mouse tail suspension test. Pharmacol Biochem Behav , 1996, 53 (3) : 535–539. DOI:10.1016/0091-3057(95)02047-0 |
| [34] | Azima H. Imipramine (Tofranil):A new drug for the depressed. Can Med Assoc J , 1959, 80 (7) : 535–540. |
| [35] | Grecksch G, Zhou D, Franke C, Schröder U, Sabel B, Becker A, Huether G. Influence of olfactory bulbectomy and subsequent imipramine treatment on 5-hydroxytryptaminergic presynapses in the rat frontal cortex:Behavioural correlates. Br J Pharmacol , 1997, 122 (8) : 1725–1731. DOI:10.1038/sj.bjp.0701530 |
| [36] | Winterhoff H, Spengler B, Christoffel V, Butterweck V, Löhning A. Cimicifuga extract BNO, 1055:Reduction of hot flushes and hints on antidepressant activity. Maturitas , 2003, 44 Suppl 1 : S51–S58. |
| [37] | Kaminsky BM, Bostwick JR, Guthrie SK. Alternate routes of administration of antidepressant and antipsychotic medications. Ann Pharmacother , 2015, 49 (7) : 808–817. DOI:10.1177/1060028015583893 |
| [38] | Cline BH, Anthony DC, Lysko A, Dolgov O, Anokhin K, Schroeter C, Malin D, Kubatiev A, Steinbusch HW, Lesch KP, et al. Lasting downregulation of the lipid peroxidation enzymes in the prefrontal cortex of mice susceptible to stress-induced anhedonia. Behav Brain Res , 2015, 276 : 118–129. DOI:10.1016/j.bbr.2014.04.037 |
| [39] | Markova N, Chernopiatko A, Schroeter CA, Malin D, Kubatiev A, Bachurin S, Costa-Nunes J, Steinbusch HM, Strekalova T. Hippocampal gene expression of deiodinases 2 and 3 and effects of 3, 5-diiodo-L-thyronine T2 in mouse depression paradigms. Biomed Res Int , 2013, 2013 : 565218. |
| [40] | Costa-Nunes J, Zubareva O, Araújo-Correia M, Valença A, Schroeter CA, Pawluski JL, Vignisse J, Steinbusch H, Hermes D, Phillipines M, et al. Altered emotionality, hippocampus-dependent performance and expression of NMDA receptor subunit mRNAs in chronically stressed mice. Stress , 2014, 17 (1) : 108–116. DOI:10.3109/10253890.2013.872619 |
| [41] | Cline BH, Steinbusch HWM, Malin D, Revishchin AV, Pavlova GV, Cespuglio R, Strekalova T. The neuronal insulin sensitizer dicholine succinate reduces stress-induced depressive traits and memory deficit:Possible role of insulin-like growth factor 2. BMC Neurosci , 2012, 13 : 110. DOI:10.1186/1471-2202-13-110 |
| [42] | Malatynska E, Steinbusch HWM, Redkozubova O, Bolkunov A, Kubatiev A, Yeritsyan NB, Vignisse J, Bachurin S, Strekalova T. Anhedonic-like traits and lack of affective deficits in 18-month-old C57BL/6 mice:Implications for modeling elderly depression. Exp Gerontol , 2012, 47 (8) : 552–564. DOI:10.1016/j.exger.2012.04.010 |
| [43] | Nunes J. Behavioural effects of chronic administration of imipramine in food and water regarding anxiety and depression paradigms on naïve C57BL/6N male mice. M.Sc. Dissertation, University of Lisbon, Lisbon, Portugal, 2011. |
| [44] | Delini-Stula A, Mikkelsen H, Angst J. Therapeutic efficacy of antidepressants in agitated anxious depression-A meta-analysis of moclobemide studies. J Affect Disord , 1995, 35 (1-2) : 21–30. DOI:10.1016/0165-0327(95)00034-K |
| [45] | Erburu M, Cajaleon L, Guruceaga E, Venzala E, Muñoz-Cobo I, Beltrán E, Puerta E, Tordera RM. Chronic mild stress and imipramine treatment elicit opposite changes in behavior and in gene expression in the mouse prefrontal cortex. Pharmacol Biochem Behav , 2015, 135 : 227–236. DOI:10.1016/j.pbb.2015.06.001 |
| [46] | Ramirez K, Sheridan JF. Antidepressant imipramine diminishes stress-induced inflammation in the periphery and central nervous system and related anxiety-and depressive-like behaviors. Brain Behav Immun, in press, DOI: 10.1016/j.bbi.2016.05.008. |
| [47] | Enginar N, Hatipoğlu I, Firtina M. Evaluation of the acute effects of amitriptyline and fluoxetine on anxiety using grooming analysis algorithm in rats. Pharmacol Biochem Behav , 2008, 89 (3) : 450–455. DOI:10.1016/j.pbb.2008.02.001 |
| [48] | Mogi K, Shimokawa Y, Nagasawa M, Kikusui T. Effects of sex and rearing environment on imipramine response in mice. Psychopharmacology (Berl) , 2012, 224 (1) : 201–208. DOI:10.1007/s00213-012-2821-y |
| [49] | Sorregotti T, Mendes-Gomes J, Rico JL, Rodgers RJ, Nunes-de-Souza RL. Ethopharmacological analysis of the open elevated plus-maze in mice. Behav Brain Res , 2013, 246 : 76–85. DOI:10.1016/j.bbr.2013.02.035 |
| [50] | Baek IS, Park JY, Han PL. Chronic antidepressant treatment in normal mice induces anxiety and impairs stress-coping ability. Exp Neurobiol , 2015, 24 (2) : 156–168. DOI:10.5607/en.2015.24.2.156 |
| [51] | Abernethyl DR, Divoll M, Greenblatt DJ, Harmatz JS, Shader RI. Absolute bioavailability of imipramine:Influence of food. Psychopharmacology (Berl) , 1984, 83 (1) : 104–106. DOI:10.1007/BF00427432 |
| [52] | Pottenger LH, Domoradzki JY, Markham DA, Hansen SC, Cagen SZ, Waechter JM Jr. The relative bioavailability and metabolism of bisphenol A in rats is dependent upon the route of administration. Toxicol Sci , 2000, 54 (1) : 3–18. DOI:10.1093/toxsci/54.1.3 |
| [53] | Volvert ML, Seyen S, Piette M, Evrard B, Gangolf M, Plumier JC, Bettendorf L. Benfotiamine, a synthetic S-acyl thiamine derivative, has different mechanisms of action and a different pharmacological profile than lipid-soluble thiamine disulfide derivatives. BMC Pharmacol , 2008, 8 : 10. |
| [54] | Mombereau C, Gur TL, Onksen J, Blendy JA. Differential effects of acute and repeated citalopram in mouse models of anxiety and depression. Int J Neuropsychopharmacol , 2010, 13 (3) : 321–334. DOI:10.1017/S1461145709990630 |
| [55] | Carr GV, Lucki I. The role of serotonin receptor subtypes in treating depression:A review of animal studies. Psychopharmacology (Berl) , 2011, 213 (2-3) : 265–287. DOI:10.1007/s00213-010-2097-z |
| [56] | Cryan JF, Sweeney FF. The age of anxiety:Role of animal models of anxiolytic action in drug discovery. Br J Pharmacol , 2011, 164 (4) : 1129–1161. DOI:10.1111/bph.2011.164.issue-4 |
| [57] | Ihne JL, Fitzgerald PJ, Hefner KR, Holmes A. Pharmac-ological modulation of stress-induced behavioral changes in the light/dark exploration test in male C57BL/6J mice. Neuropharmacology , 2012, 62 (1) : 464–473. DOI:10.1016/j.neuropharm.2011.08.045 |