Neuroimaging techniques have provided powerful tools to assess various neurological disorders, provid-ing both structural and functional information. In the brain, traditional X-ray, computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET), magnetoencephalography (MEG), electroencephalography (EEG), and combinations of these techniques have been used to help neuroscientists and clinicians to better understand the structural and functional pathology[1]. In recent years, cranial ultrasound, diffuse optical imaging (DOI), and near infrared spectroscopy (NIRS) have been developed as new modalities for neuroimaging.
When applying neuroimaging techniques to the spinal cord, the limited accessibility of the spinal cord caused by the inherent anatomy of the spine impedes the ability to understand human spinal cord function and assess the effects of traumatic injuries or diseases. Conventional plain radiography and CT provide high-contrast images of soft tissues and bony structures; hence, these became the primary imaging methods used in routine orthopedics for visualizing spinal structures[2]. Meanwhile, two nuclear tomography imaging techniques, single-photon emission computed tomography (SPECT) and positron emission tomo-graphy (PET), were developed to meet the growing demand for functional evaluations of the human body. Because of the low spatial resolution of the acquired images, 3D imaging with these two techniques is often accomplished with the aid of a CT scan, through co-performance in a combined scanner (SPECT/CT or PET/CT) or through post-processing co-registration to obtain high-resolution CT images[2]. However, these four imaging methods are less sensitive to soft tissue contrasts and all involve radiation exposure and its inherent risks. The advent of magnetic resonance imag-ing (MRI) changed the field of spinal cord imaging, and is the only approach to gain high-resolution structural images and functional information con-cerning the human spinal cord during one scan with a single device.
The spine is considered to be one of the most challenging structures in the body to be imaged by MRI because of the following reasons: magnetic field inhomogeneity created by the interfaces between the surrounding bone, ligaments and other soft tissues, and cerebrospinal fluid (CSF) in the spinal canal; the small cross-sectional dimensions of the cord; and the relatively large physiological motion of the cord and adjacent tissues during cardiac and respiratory cycles. The artifacts caused by metallic implants or devices often exacerbate the difficulty in such imaging, especially for the patients[3].
With the continuous improvement of imaging sequences, conventional MRI techniques (usually referring to spin echo, gradient echo, and inversion recovery sequences, with T1-, T2-, or proton density- weighting) have proven their ability to provide accurate and high-resolution anatomical images of the spinal cord and have been widely used as an important imaging modality for most spinal disorders. However, conventional MRI provides little information about the inherent integrity and the physiological status of the spinal cord tissue because the alterations in signal intensity are usually non-specific and have poor correlation with neurological and functional impair-ment in various spinal cord pathologies. Therefore, they do not provide sufficiently reliable prognostic information at the microstructural and functional levels[4].
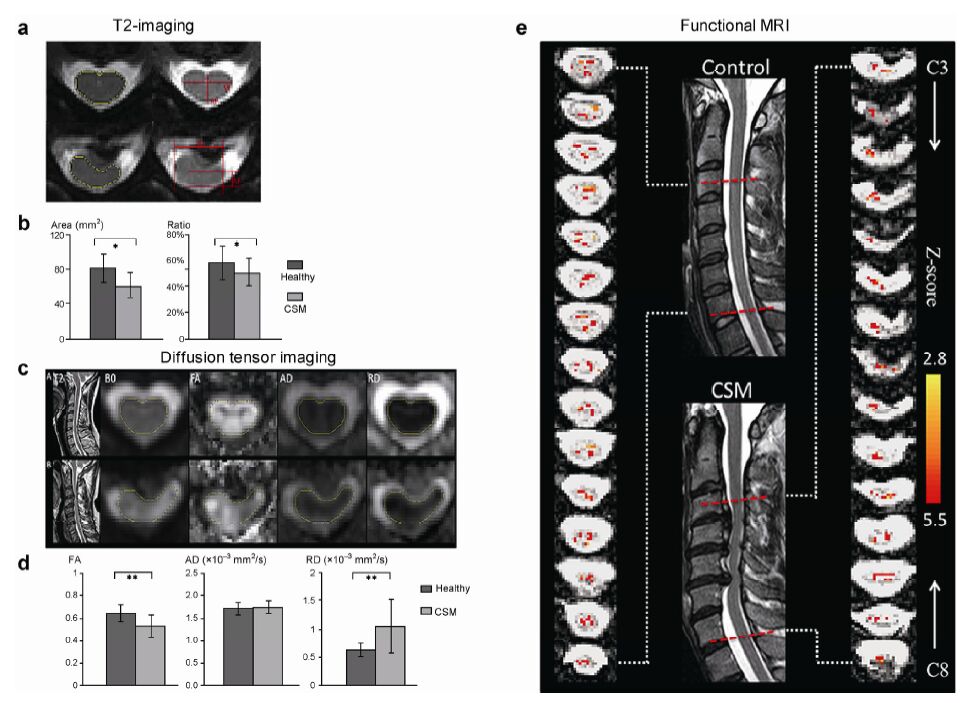
An international meeting sponsored by the Inter-national Spinal Research Trust and the Wings for Life Foundation was held in 2013[3,5]. In this meeting, spinal cord imaging experts identified the current challenges and demands of spinal cord imaging. They delineated five advanced MRI techniques that have the potential to improve the understanding of the human spinal cord in healthy and pathological conditions, and serve as imaging biomarkers to characterize degree of damage, monitor the response to treatment, and predict the outcome of intervention. These five imaging techniques included functional MRI (fMRI)[6], diffusion tensor imaging (DTI)[7], magnetic resonance spectroscopy (MRS)[8], myelin water-fraction imaging (MWF)[9], and magnetization transfer (MT)[10]. The first two key techniques have already been commonly used in clinical research, as shown in Figure 1. Based on the basic concept of the blood oxygen level- dependent (BOLD) signal or signal enhancement by extravascular protons (SEEP), fMRI can characterize neurological function in the resting state or through block or event-related designs, which indirectly reflect the neuronal activity and connectivity occurring within the spinal cord. Diffusion-weighted imaging (DWI) and its extensions, such as DTI and diffusion kurtosis imaging (DKI), measure the directional diffusivity of water. With application to the spinal cord, the derived metrics could provide information concerning axonal integrity, and reveal degenerative and regenera-tive changes[7]. It is notable that all five advanced techniques were initially developed and validated in brain imaging, high-quality spinal cord imaging using these methods has only recently been achieved by using specialized acquisition sequences, cautiously placed shimming, and complicated post-processing for artifact correction.
|
| Figure 1 A combination of magnetic resonance imaging (MRI) techniques. Diffusion tensor imaging (DTI) and corresponding T2-weighted structural measurements provided sufficient structural and functional information in a patient with cervical myelopathy. T2-weighted images showed a morphometric change with stenosis. DTI showed decreased fractional anisotropy (FA) and increased radial diffusivities (RD), indicating demyelination in the white matter. In functional MRI, the blood oxygen level-dependent signal change in a patient with cervical myelopathy was significantly higher than that in a healthy subject (*p<0.05 **p<0.01). |
Significant advances in neuroimaging techniques have been realized in its clinical application to spinal cord disorders in the past decade. However, it should be noted that there are still great challenges in bringing those neuroimaging techniques into clinical use. First, the relatively low spatial resolution is a significant problem, limiting the potential precision and accuracy in the diagnosis of lesions in the spinal cord[3]. Second, partial volume effects with the CSF, the effects of physiological noise, and long scan time still limit the application of these techniques in practical clinical use and require constant endeavor. Further assessments are needed in the following areas: obtaining high resolution images by developing acquisition techni-ques and/or image processing, utilizing multi-modal neuroimaging data fusion from structure to function, and translating neuroimaging findings into pathologi-cal information in clinical studies with a high level of evidence[4]. It is expected that novel applications of spinal cord neuroimaging will arise in clinical use after further research by radiologists, engineers, neuroscientist, clinicians, and surgeons.
| [1] | He B, Coleman T, Genin GM, Glover G, Hu X, Johnson N, Liu T, Makeig S, Sajda P, Ye K. Grand challenges in mapping the human brain: NSF workshop report. IEEE Trans Biomed Eng , 2013, 60 (11) : 2983–2992. DOI:10.1109/TBME.2013.2283970 |
| [2] | Uludağ K, Roebroeck A. General overview on the merits of multimodal neuroimaging data fusion. NeuroImage , 2014, 102 (Pt 1) : 3–10. |
| [3] | Stroman PW, Wheeler-Kingshott C, Bacon M, Schwab JM, Bosma R, Brooks J, Cadotte D, Carlstedt T, Ciccarelli O, Cohen-Adad J, et al. The current state-of-the-art of spinal cord imaging: Methods. NeuroImage , 2014, 84 : 1070–1081. DOI:10.1016/j.neuroimage.2013.04.124 |
| [4] | Martin AR, Aleksanderek I, Cohen-Adad J, Tarmohamed Z, Tetreault L, Smith N, Cadotte DW, Crawley A, Ginsberg H, Mikulis DJ, et al. Translating state-of-the-art spinal cord MRI techniques to clinical use: A systematic review of clinical studies utilizing DTI, MT, MWF, MRS, and fMRI. NeuroImage Clin , 2016, 10 : 192–238. DOI:10.1016/j.nicl.2015.11.019 |
| [5] | Wheeler-Kingshott CA, Stroman PW, Schwab JM, Bacon M, Bosma R, Brooks J, Cadotte DW, Carlstedt T, Ciccarelli O, Cohen-Adad J, et al. The current state-of-the-art of spinal cord imaging: Applications. NeuroImage , 2014, 84 : 1082–1093. DOI:10.1016/j.neuroimage.2013.07.014 |
| [6] | Liu XJ, Zhou FQ, Li X, Qian WS, Cui JL, Zhou IY, Luk KDK, Wu EX, Hu Y. Organization of the intrinsic functional network in the cervical spinal cord: A resting state functional MRI study. Neuroscience , 2016, 336 : 30–38. DOI:10.1016/j.neuroscience.2016.08.042 |
| [7] | Wang SQ, Li X, Cui JL, Li HX, Luk KDK, Hu Y. Prediction of myelopathic level in cervical spondylotic myelopathy using diffusion tensor imaging. J Magn Reson Imaging , 2015, 41 (6) : 1682–1688. DOI:10.1002/jmri.v41.6 |
| [8] | Kowalczyk I, Duggal N, Bartha R. Proton magnetic resonance spectroscopy of the motor cortex in cervical myelopathy. Brain , 2012, 135 (2) : 461–468. DOI:10.1093/brain/awr328 |
| [9] | Laule C, Vavasour IM, Zhao Y, Traboulsee AL, Oger J, Vavasour JD, Mackay AL, Li DKB. Two-year study of cervical cord volume and myelin water in primary progressive multiple sclerosis. Mult Scler , 2010, 16 (6) : 670–677. DOI:10.1177/1352458510365586 |
| [10] | Cohen-Adad J, El Mendili MM, Lehéricy S, Pradat PF, Blancho S, Rossignol S, Benali H. Demyelination and degeneration in the injured human spinal cord detected with diffusion and magnetization transfer MRI. NeuroImage , 2011, 55 (3) : 1024–1033. DOI:10.1016/j.neuroimage.2010.11.089 |



