2. The First Clinical Medical College of Northwest University for Nationalities, Yinchuan 750002, China
The process of cardiopulmonary-cerebral resuscitation (CPCR) after cardiac arrest (CA) is essentially the same as that of hypoxic-ischemia reperfusion in the whole body. In this process, the severe inflammatory response in the body is an important reason for brain injury after resuscitation[1,2]. ICAM-1 can make leukocytes adhere to endothelial cells and induce inflammatory response via leukocytes, which can injure neurons in the brain[3]. This may be an important cause of brain injury after resuscitation. S100β is a protein, the excessive expression of which is associated with the severity of brain injury[4]. The clinical drug heparin has an anticoagulationeffect; in addition, it also has anti-inflammatory effects and regulates the immune response[5]. The effect of heparin on serum ICAM-1 and S100β protein in the hippocampus after resuscitation was unknown. In this study, we usedheparin after CA in rabbits and observed the expression of serum ICAM-1 and hippocampal S100β protein after cardio-pulmonary resuscitation (CPR), to explore the impact of heparin on inflammatory response and neuron protection.
2 Materials and methods 2.1 Animal preparation and groupingThirty-two healthy male New Zealand rabbits, weighing 2.0-2.5 kg, were randomly divided into three groups (group I, II, and III). Group I, n = 8: control group; Group II, n = 12: saline group; Group III, n = 12: heparin group. The CA animal model was created according to the method by Ye et al. [6]. Anesthesia was induced by intraperitoneal injection of pentobarbital sodium (45mg/kg). Continuous monitoring of electrocardiogram (ECG), mean arterial pressure (MBP), heart rate (HR), and the end-tidal carbon dioxide partial pressure (PetCO2) was performed. The trachea was intubated and the lungs were mechanically ventilated using a rodent ventilator with a target tidal volume of 10mL/kg and respiratory rate of 30-40 breaths/min to generate a partial pressure of carbon dioxide near 40mmHg, with an endotracheal tube that has an inflatable sac at the end. The right femoral artery was catheterized to measure the MBP and the right femoral vein venipuncture infusion fluids in the form of 0.9% saline were infused at a rate of 2-3 mL/kg/h to prevent volume depletion.
2.2 Asphyxial cardiac arrest (ACA) and stability after 5minTo induce ACA, apnea was induced by clipping the endotracheal tube causing CA for 5 min. The following were the standards for CA: ①ECG waveform disap-peared or showed ventricular fibrillation, electrical mechanical dissociation; ②MBP <10 mmHg; ③Oral mucosa showed severe cyanosis. Animals in group II received heparin (0.5 mg/kg) and in group III did not receive heparin.
2.3 Cardiopulmonary resuscitationResuscitation was initiated by chest compression with a frequency of 200 times/minute and administration of a bolus injection of epinephrine (0.01mg/kg, intravenously) and 5% sodium bicarbonate (1 mol/kg, intravenously), followed by mechanical ventilation using pure oxygen with a target tidal volume of 10mL/kg and respiratory rate of 60 breaths/min. We continued chest compression until the return of spontaneous circulation (ROSC). Following were the standard characteristics for ROSC: ①The QRS wave ofECG appeared normal, ②Heart beat can be touched, ③MBP >60 mmHg for 10 min, ④Oral mucosa, skin color turned back to ruddy. If ROSC was not present within 3 min, we determined the recovery as a failure.
2.4 Sample collectionAfter 24h ROSC, we placed the blood sample from the carotid artery quickly into the centrifuge at 3, 000 r/min for 10 min with no anticoagulation. The collected serum was stored under -20℃. After a blood sample was obtained, the chest was opened to cut off the right auricle and ligature the abdominal aorta. Saline (500mL) was injected from the left ventricle, followed by 800 mL of 4% paraformaldehyde. After the whole body was fixed, the skull was cut open from the foramen magnum to expose the whole cerebrum. The optic nerve was cut and the cerebrum was removed and fixed using 4% paraformaldehyde.
2.5 ObservationEnzyme-linked immunosorbent assay (ELISA) method was used to detect the blood serum levels of ICAM-1, and immunohistochemical staining was used to detect the expression of S100β protein in the hippocampus. Five horizons were randomly selected in each section under a microscope (100x magnification), and the posi-tive and total cells were counted and normalized. Based on the S100β protein positive cells rate, all sections were divided into four levels—Level 1, number of stained cells < 10%; Level 2, staining cells 11%-35%; Level 3, staining cells 36%-55%; Level 4, staining cells > 55%.
2.6 Statistical analysisResults are expressed as a mean ± standard deviation. Statistical analysis was by one-way analysis of variance followed by Tukey's post hoc test or Student's t-test for paired or unpaired samples and rank sum test for ranked data as appropriate with Statistical Package for Social Sciences 15.0 statistical software. A P < 0.05 level of probability was accepted as significant.
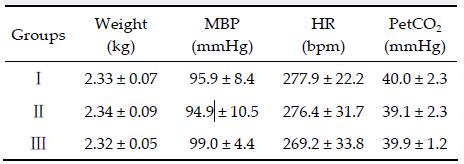
3 Results 3.1 Baseline of vital signs before CPRThere was no statistically significant difference among the three groups in body weight, MBP, HR, and PetCO2 (P > 0.05) (Table 1).
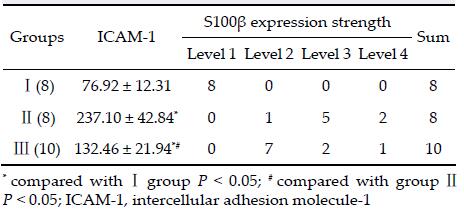
ELISA method was used to detect the serum con-centration of ICAM-1 after recovery. The difference was statistically significant among the three groups (P< 0.05). Immunohistochemical staining method was used to detect S100β protein expression in the hippo-campus. Eight cases in group I were at level 1; 1 case in group II and 7 cases in group III were at level 2; 5cases in group II and 2 cases in group III were at level 3; 2 cases in group II and 1 case in group III were at level 4. The difference of S100β protein expression was statistically significant among the three groups (P < 0.05).
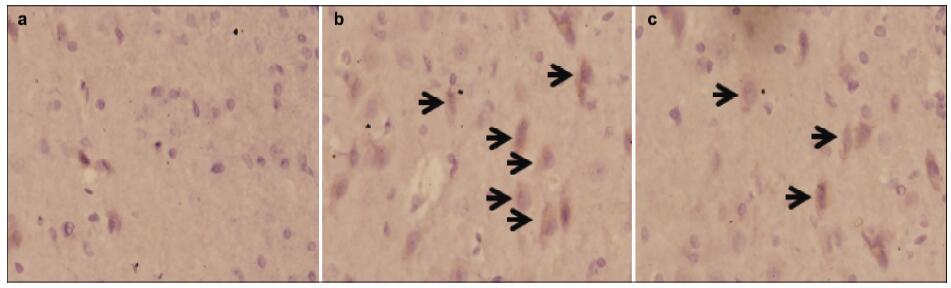
Immunohistochemical staining was used to observe S100β protein in the rabbit hippocampus sections. Negative protein cells displayed transparent light membrane and cytoplasm. Positive protein cells displayed brownish yellow cytoplasm and the color was deeper than the background (Figure 1).
|
| Figure 1 Immunohistochemical analysis of S100 β expression in the different groups. a Sham group. b Saline group. c Heparin group. |
The results of this study showed low levels of expression of ICAM-1 in the normal rabbit's serum, but the quantity increased significantly after CA-CPR. This means that after recovery the body's expression of ICAM-1 may be raised by certain mechanisms, which also enhances the inflammatory reaction mediated by ICAM-1. Our research also showed that serum ICAM-1 expression in the heparin group decreased compared
with that of the normal saline group, but was still lower than normal levels after recovery. This indicates that during CA, heparin therapy can inhibit the high expression of ICAM-1 in the serum after recovery. Belonging to the immunoglobulin superfamily, ICAM-1 is widely distributed in the whole body but is mainly concentrated in vascular endothelial cells, mediating the white blood cells' inflammatory response[7]. Therefore, we can speculate that heparin may somehow inhibit the expression of ICAM-1 in the process of recovery, which could reduce the inflammatory reaction after recovery. Further research is required in this regard.
This study shows that the strength of the rabbit hippocampal S100β protein expression in the normal saline group increased after recovery. The heparin group showed decreased expression of S100β protein compared with the saline group, but the levels did not reach those of the normal control group. The S100β protein level increase in the hippocampus is an early response to ischemia and hypoxia. It is also con-sidered as a regulatory gene protein, which can mediate many pathophysiological reactions in ischemia and hypoxia[8]. High concentration of S100β protein is poisonous to nerve cells and can inhibit neuronal growth, induceapoptosis, and even necrosis. Studies have shown that higher the concentration of S100β protein, the greater injury to the brain[9]. The main reason for the decrease in S100β protein expression after recovery in the heparin group may be that heparin could decrease the expression of ICAM-1 in the serum, which reduced the inflammatory response mediated by ICAM-1 after ischemia and hypoxia. This kind of inflammatory response in the intracranial brain cells may be the most important cause forincreasing S100β protein level.
As discussed above, we suggestthat heparin can reduce the body's ICAM-1 expression through cer-tain pathways after hypoxic-ischemia reperfusion, then reduce the inflammatory response mediated by ICAM-1, and also reduce S100β expression in the brain. The above indirectly illustrates that heparin can alleviate brain damage after recovery by reducing the inflammatory response. However, further studies should be conducted on how heparin influences the body to regulate ICAM-1.
Conflict of interests
All contributing authors have no conflict of interests.
| [1] | Beurskens CJ, Horn J, de Boer AM, Schultz MJ, van Leeuwen EM, Vroom MB, Juffermans NP. Cardiac arrest patients have an impaired immune response, which is not influenced by induced hypothermia. Crit Care , 2014, 18 (4) : R162. DOI:10.1186/cc14002 |
| [2] | Kelm RF, Wagenführer J, Bauer H, Schmidtmann I, Engelhard K, Noppens RR. Effects of levosimendan on hemodynamics, local cerebral blood flow, neuronal injury, and neuroinflammation after asphyctic cardiac arrest in rats. Crit Care Med , 2014, 42 (6) : e410–e419. DOI:10.1097/CCM.0000000000000308 |
| [3] | Chakraborty S, Núñez D, Hu SY, Domingo MP, Pardo J, Karmenyan A, Eva Ma Gálvez, Chiou A. FRET based quantification and screening technology platform for the interactions of leukocyte function-associated antigen-1(LFA-1) with intercellular adhesion molecule-1(ICAM-1). PLoS One , 2014, 9 (7) : e102572. DOI:10.1371/journal.pone.0102572 |
| [4] | Kabadi SV, Stoica BA, Zimmer DB, Afanador L, Duffy KB, Loane DJ, Faden AI. S100B inhibition reduces behavioral and pathologic changes in experimental traumatic brain injury. J Cereb Blood Flow Metab , 2015, 35 (12) : 2010–2020. DOI:10.1038/jcbfm.2015.165 |
| [5] | Mulloy B, Hogwood J, Gray E, Lever R, Page CP. Pharmacology of heparin and related drugs. Pharmacol Rev , 2016, 68 (1) : 76–141. |
| [6] | Ye QS, Wang LL, Wang JK, Shi WZ, Liu H, Han HW, Ma ZF, Hai KR. Evaluation of a rat model of asphyxial cardiac arrest and cardiopulmonary resuscitation. Chin J Anesthesiol , 2008, 28 (8) : 750–752. |
| [7] | Tang HP, Sun LX, Han W. Endothelial cells on the proliferation and expression of intercellular adhesion molecule 1 and interleukin 8 of vascular smooth muscle cells. Genet Mol Res , 2013, 12 (4) : 4363–4370. DOI:10.4238/2013.October.10.2 |
| [8] | Yao B, Zhang LN, Ai YH, Liu ZY, Huang L. Serum S100β is a better biomarker than neuron-specific enolase for sepsis-associated encephalopathy and determining its prognosis: A prospective and observational study. Neurochem Res , 2014, 39 (7) : 1263–1269. DOI:10.1007/s11064-014-1308-0 |
| [9] | Cheong KA, Noh M, Kim CH, Lee AY. S100B as a potential biomarker for the detection of cytotoxicity of melanocytes. Exp Dermatol , 2014, 23 (3) : 165–171. DOI:10.1111/exd.12332 |