|
The difference between the electroencephalographic rhythms in awake and sleeping humans was initially described by Berger[1]. It became a breaking point in the development of sleep science or somnology. The appearance of the EEG method initiated a series of discoveries in sleep studies. In 1937, Frederic Bremer in Brussels performed his famous experiments on the isolated brain. He transected the feline brain at the level of the upper and lower brain stem and found the following: with the lower level of transection (when the connections between the most of the brain stem and the hemispheres remain unaffected), the alternation of the EEG patterns of wakefulness and sleep persists; with the upper level transection, the brain assumes the state of "deep sleep" (coma, in modern terms). These studies were an important landmark in understanding the waking mechanisms though they were erroneously interpreted: it was suggested initially that the brain falls asleep due to a decrease in the inflow of the sensory impulses[2].
The reason of the Bremer's cats falling into a "deep sleep" was not revealed until 1949 when Moruzzi and Magoun performed their experiments. Initially, they confirmed that the transition from sleep to wakefulness appears as EEG desynchronization, that is, the alter-nation of the patterns of cerebral electrical activity from the high amplitude low-frequency rhythms characteristic of deep sleep to the low amplitude high- frequency rhythms of wakefulness. Next, they found that the desynchronization is induced by a specific structure located inside the brain stem, the so-called reticular (cancellated) formation[3]. The following studies by the above and other authors have demon-strated that though the structure concerned is activated by the afferent stimuli (from the sensory organs), its injuries result in an uninterrupted deep sleep (coma) as had been demonstrated previously in Bremer's experiments. Thus, the cerebral ascending reticular activating system (ARAS) particularly responsible for waking maintenance was discovered. Initially, it was considered to be diffused; however, modern neuro-morphology has distinguished about ten separate clusters of nerve cell bodies releasing various chemical mediators, the so-called "waking centers" that are located "inside" the ARAS at all the levels of the cerebral axis[2, 4-6] (Figure 1a).
|
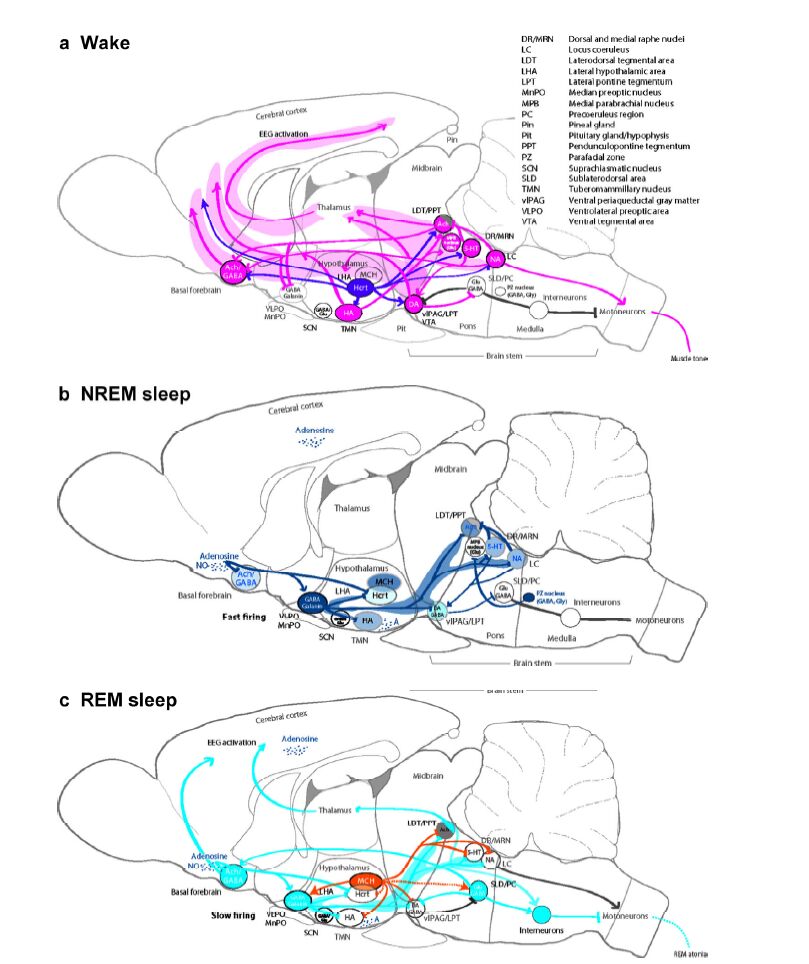
| Figure 1 Sleep-wake regulation. (a) Wake: The brain stem arousal nuclei (pink) containing ACh, DA, 5-HT, or NA activate the thalamus, hypothalamus, spinal cord motor neurons, and the basal forebrain, and inhibit the VLPA (GABA, galanin); the hypothalamic arousal centers [pink: HA; dark purple: Hcrt] activate the cortex and arousal-related regions in the basal forebrain and brain stem; the thalamus activates the cortex. The glutamatergic MPB in the dorsal pontine tegmentum regulates arousal[28]. (b) NREM sleep: The hypothalamic preoptic area nuclei (dark blue), containing GABA and galanin, inhibit the brain stem and hypothalamic arousal nuclei; the endogenous sleep regulatory substances [adenosine and NO] inhibit basal forebrain arousal nuclei, hypocretin neurons, and TMN neurons; and adenosine activates VLPO neurons. NREM-REM switch: During NREM sleep, serotonergic DR and noradrenergic LC neurons inhibit LDT/PPT neurons. During NREM sleep, vlPAG/LPT neurons inhibit SLD/PC neurons. The GABAergic PZ in the pontomedullary junction promotes sleep[36]. (c) REM sleep: The REM-active brain stem nuclei, including LDT/PPT/SLD/PC and containing ACh, Glu, or GABA, promote activity in the basal forebrain and cortex and induce muscle atonia and rapid eye movements; hypothalamic neurons, containing MCH, promote REM sleep by suppressing REM-inhibitory brain centers, including vlPAG/LPT/DR/LC. NREM-REM switch: During REM sleep, DR/LC neurons become silent, enabling the cholinergic LDT/PPT neurons to generate the hallm arks of REM sleep, including rapid eye movements, EEG activation, and muscle atonia. This reciprocal activity between REM-on (LDT/PPT) and REM-off (DR/LC) neurons drives the cycling between REM and NREM sleep episodes. Additionally, GABAergic neurons participate in the mutual inhibition of REM-activating and REM-suppressing neurons. During REM sleep, SLD/PC neurons use ascending and descending projections to activate the cortex and promote muscle atonia. |
After the studies by Moruzzi and Magoun, it became clear that the normal functioning of the thalamocortical system of the brain, which provides the entire range of human conscious activity during wakefulness, is possible only if a strong tonic activating inflow from the definite subcortical structures is available[7]. Direct studies of the neurons involved in the sleep- wakefulness regulation, which was performed during the second half of the last century, revealed membrane depolarization of the majority of cortical neurons to 61-63 mV (the so-called "UP" state[8-11]). This membrane depolarization is due to the aforementioned ARAS effects during wakefulness. These neurons process and respond to the incoming signals from other nerve cells, both receptor and intracerebral, only in a state of tonic depolarization. Neocortical neurons require ascending tonic depolarization. Similarly, the spinal motoneurons require descending depolarization for the maintenance of the muscle tone. The latter is critical for position maintenance and voluntary movement performance. A general description of the ascending activating system as provided at the end of the first decade of the 21st century is the topic of several recent important revi ews[2, 4-6].
Electrographically cortical activation appears as suppression of all the slow rhythms (δ, θ, α, and σ bands) in EEG, increase in a power of the β-band rhythms (15-30 Hz), and synchronization of high- frequency rhythms in γ-band (30-60 Hz). Subsequently, the muscles tone increases, and sympathicotonia emerges. Psychologically, the state of alertness, that is, the readiness of the organism for action, appears. There are some data available, though contradictory and imperfect, on the specificity of the contribution of each of the activating subsystems to waking main-tenance. Thus, generally accepted is the relation of ACh-ergic and Glu-ergic subsystems mostly to electro-graphic and behavioral manifestations of arousal. Furthermore, the NA-ergic is responsible for changes in muscle tone and position responses, Ser-ergic for the transition from waking to sleep, Hist-ergic for the general regulation of behavior and memory, and DA-ergic for strong emotions and stress. Thus, it was proposed that complexity and even apparent redundancy in organization of the cerebral activating system ensure safety, on one hand, and reflect all the complexity of behavioral tasks, which the mammalian brain should resolve during the state of wakefulness, on the other[2, 4-6].
It was revealed that the activity of the "mediators of wakefulness" (Glu, ACh, NA, Ser, Hist, DA, and Orx) is modulated by numerous peptides localized inside the same vesicles[12]. In accordance with the neurological data available for humans (and, presum-ably, other primates), any disorder in the activity of each of these subsystems could not be compensated at the expense of the other. The most critical are the activating subsystems localized at the level of the rostral part of the midbrain, LHA, and BF areas. Lesions in these particular parts of the brain of humans and primates are inconsistent with the presence of consciousness and result in a coma[5, 13, 14].
In accordance with the "classical" theory completed at the end of the first decade of the millennium, besides the cholinergic systems of PPT/LDT and BF, important roles in the hierarchy of the "waking centers" are played by the Orx-ergic system and the Hist-ergic system, closely connected with each other both anatomically and functionally. Figuratively speaking, in this well-coordinated orchestra of activating systems, the Orx-ergic system plays the role of a conductor and the Hist-ergic system of a concertmaster (the first violin). The structure and function of the Orx-ergic and melatonergic (MCH) reciprocal to it as well as the Hist-ergic and DA-ergic systems were considered in several important reviews[2, 4-6, 15-17] (Figure 1a).
2 Revision of the ARAS conceptsHowever, the concepts of well-coordinated interrela-tionships of the hierarchically organized "centers of wakefulness" became gradually disrupted during the first half of the second decade of the 21st century with the incorporation of novel experimental methods. It was revealed that behavioral abnormalities in laboratory rodents induced by selective lesion of aminergic and cholinergic ARAS cell bodies were not as striking as was anticipated. Shiromani, Gerashchenko, and their collaborators performed experiments on big and strong Sprague-Dawley rats (aged up to 6 months and weighing up to 620 g)[18]. They revealed that the local intracerebral injection of specific saporin-containing neurotoxins to these animals, allowing targeted lesions of chemically recognized neuronal bodies of ARAS, fails to induce any significant disorders of the sleep-waking cycle. These lesions affected up to 75% of the Hist/TMN neurons as well as 90% NA/LC and ACh/BF neurons without affecting the surrounding cells (as far as it could be deduced from the results of morphocontrol). It was found that simultaneous lesions of one, two, and even three systems in the same animals after 20 days resulted only in a few minor changes in the sleep-wakefulness cycle, probably due to the activation of compensatory mechanisms. The most important was a two-fold decrease in the waking percentage during the transition from the light to the dark pha se of nychthemeron as well as a two-fold decrease in REM sleep percentage during the light phase.
The effect was very si milar to the characteristics of the histidine-decarboxylase (HDC)-knockout mice (without histamine). Several earlier studies in both rats and cats have shown that the selective toxic destruc-tion of the aminergic and cholinergic ARAS cell bodies also induced only very limited disorders in EEG and waking behavior[19]. That gave Michel Jouveta reason to announce "mort de la théorieréticulaire" (death of the reticular theory)[20]. It might be suggested that the negligible effect of such "chronic" lesions could be attributed, at least in part, to a rather prolonged recovery period (with respect to the rodent lifespan): within 3 weeks after the lesions were made, a few im-portant recovery processes could occur in the rat brain. However, novel optogenetic methods make it possible to selectively, reversibly, and "acutely" (for a short time) switch on and off some of the neuronal groups in free-moving laboratory mice without anesthesia. Such exper iments were performed in the laboratory of Louis de Lecea, and again they demonstrated very limited effects: moderate decrease in waking percentage and an increase in the slow-wave sleep during the dark phase of the nychthemeron after a reversible selective inhibition of noradrenergic neurons of LC for 1 h in free-moving mice[21]. Selective activation of the orexin/hypocretin neurons with the same experiments increased waking and c-Fos expression оf NA/LC and Hist/TMN neurons; however, it could not "counteract" the sleep deprivation effects. The same behavioral effect was observed in HDC-knockout mice, that is, an increase in waking percentage in the complete absence of histamine.
Cliff Saper wrote in 2010: "The presence of such a large number of cell groups that are thought to pro-mote arousal raises the question of how they interact in this process." It is interesting that the drugs that block transmission for one or another of these pathways (e.g., muscarinic antagonists, H1-histamine antagonists, or α2-adrenergic agonists) cause acute sleepiness; however, chronic ablation of the basal forebrain cholinergic neurons[22], tuberomammillary histaminergic neurons[23], or the LC and pontine cholinergic neurons[24-26], or combinations of these structures[18] have minimal effects on the duration of wakefulness. One possible reason for this puzzling result is that the arousal system may contain sufficient redundancy (stressed by me, KV) that remaining wake-promoting systems may be able to compensate for the chronic (but perhaps not acute) loss of one or even a few components, e.g., by increasing activity or receptor sensitivity in intact arousal systems[27].
The minor effect of the irreversible subtotal destruc-tion of the three "key" activating subsystems (including Hist-ergic), whose role in waking maintenance seemed to be undoubtedly proved by Bremer, Magoun and Moruzzi, Lindsley, Llinas, Steriade, and dozens of other researchers based on numerous neuroanatomical, neurophysiological, neuropharmacological, neuro-chemical, neurogenetic, and clinico-neurological data, leads us to accept the "classical" theory of ascending activating effects with caution. The question arises: Could the activation of certain neuronal systems that was recently thought to be the cause of neocortical tonic depolarization be instead its consequence, and the actual cause is the activation of other unknown systems[16]?
This hypothesis was confirmed by the following studies by Saper and his collaborators[28]. They demon-strated that closely to the Glu-ergic PC/PB midbrain complex, the redundancy disappeared completely, and the enlarged but strictly selective lesion of this area (as well as the BF area) results in a coma!
Special attention was paid to the mesopontine area of reticular formation of the brainstem. Lesions in this particular area, not lower or higher along the cerebral axis, resulted in a comatose-like condition in experi-mental animals as well as neurological patients[13]. This is the area immediately below the free edge of the tentorium in the primate brain, and injury to this area due to edema, hemorrhage, tumor, or other mechanical or physiological factors result in a variety of comatose- like conditions. Surprisingly, until recently this area has remained almost unexplored. The comparative contribution of the dorsal (thalamic) and ventral (hypothalamic) ascending streams to the neocortical activation also remains unclear.
The authors used the same Sprague-Dawley rats as model animals, very strong and "clever" animals, much more suitable than the albino Wistar rats for studying behavioral and EEG consequences of the experimental neurotoxic actions. Initially, they made "targeted" neurochemical lesions within the thalamic area and BF. It has been revealed that almost com-plete destruction of the neurons of all the thalamic nuclei (geniculate bodies remained unaffected), using local injections of ibotenic acid, failed to induce any appreciable changes either in EEG and EMG patterns or SWS and REM sleep percentages during the light and dark phases of nychthemeron. Spectral charac-teristics of EEG did not differ from the control groups either except for sleep spindles, which, as anticipated, completely disappeared from EEG. These data contradict the classical studies by Villablanca and Steriade that showed the participation of the thalamus in the EEG generation in cats[29, 30]. The same was found in rats[5].
The authors did not study the complicated forms of behavior but only certain simple forms (activity in the open field, reactivity to novel objects and external stimuli, food search, jumping from an open cage, etc.), which did not differ from the controls. The c-Fos expression in different neocortical areas and the "waking centers" (Hist/TMN, Orx/LHA, and NA/LC) after prolonged periods of wakefulness were similar in both control and athalamic rats.
BF area consists of large corticipetal cholinergic and non-cholinergic (mostly GABAergic) neurons located in the following regions of the rat brain: the medial septum/diagonal band of Broca, medial part of globus-pallidus, magnocellular basal and preoptic nuclei, and substancia innominata. The authors performed an almost complete destruction of all the neurons of these structures using injections of high doses of orexin-saporin, "killing" all the cells that bear orexin receptors on their surface. At the same time, retrograde lesions of the afferent neurons in the areas of Orx/LHA, Hist/TMN, and NA/LC were absent. About 10 days after the injections, all the animals with enlarged bilateral lesions showed a comatose-like condition: they did not respond to sounds and touches though the brain stem reflexes (for instance, the righting reflex) remained, whose disappearance is considered to be a clear indication of deep sleep, coma, or narcosis. The animals lost their ability to eat and drink even if food and water were supplied directly to them; hence, they required daily saccharose administration for the maintenance of living condition. There were no signs of the exit from the coma during the 1-week period of attendance. Permanent high voltage slow waves were observed in EEG though the EMG signal level varied greatly during the nychthemeron. Consequently, normal EEG spectrum changed drastically. The frequencies over 4 Hz disappear ed almost completely, and the frequencies at the band 1-4 Hz were greatly inhibited. Only spectral components of 0.5-1.0 Hz persisted. Sometimes, during the dark period of nychthemeron, a few weak attempts of behavioral activation could be seen when the animal rose and tried to take 1 or 2 steps; however, the EEG remained unchanged, and no signs of arousal reaction were present. Any goal-oriented behavior was completely absent. Histological studies revealed a practically complete disappearance of non-cholinergic and 90% decrease in the number of cholinergic neurons.
To study behavioral reactivity, the animals were placed in cages without covers for 2 h. The control rats continued running without interruption the entire time; however, the comatose ones needed continuous pushing. During these pushing periods, the EMG muscle tone demonstrated adequate changes; however, the EEG showed monotonous synchrony. The c-Fos expression revealed the minimal activity of the neocortical (cingulate) cells similar to that during deep narcosis. At the same time, the neurons in the "waking centers" in the hypothalamus and brain stem (Hist/TMN, Orex/LHA, and NA/LC) demonstrated the level of expression as high as that in the control animals. Thus, it can be concluded that the BF neurons are critical for the processing of these activating stimuli to the neocortex, as suggested by the authors. However, it is known that the activation of Hist/TMN, Orex/LHA, and NA/LC is also necessary for the exit from coma. Probably, all activation system work synergistically to maintain cortical excitability.
These results appear quite unexpected because it is known from the literature that more limited lesions of the same area did not result in such dramatic changes in EEG and behavior. Subsequently, the authors decided to distinguish the cells to which the above- described effects could be attributed. For this purpose, they administrated immunoglobulin-192-saposin to selectively destroy up to 95% cholinergicneurons in the lateral ventricle, with no effect either on the structure of sleep-waking cycle or spectral EEG characteristics and c-Fos expression in the neocortex, hypothalamus, and brain stem (His/TMN, Orex/LHA, and NA/LC) during forced wakefulness. Then, the authors destroyed the non-cholinergic neurons selectively using a lower dose of the orexin-saporin "killing" practically all these cells but only 20% cholinergic units. Strikingly, the result was the same—no changes! How can the state of wakefulness be maintained by the BF cholinergic cells in the absence of the GABAergic ones? Even more surprisingly, how can the state of wakefulness be maintained by the GABAergic neurons in the absence of the cholinergic BF cells? Moreover, why does only the lack of both cell types result in a comatose-like state in rats? Alternatively, is the chemistry of the destroyed cells not significant, and only the number of cells is important? These questions remain without an answer.
Next, the authors decided to identify the afferents of the BF neurons. For this purpose, they used the known retrograde tracer, the fragment of the cholera toxin, which was administered to the area of substanciain-nominata. The authors confirmed the data from the literature about the connections of this area mainly with the "waking centers" DA/VTA, NA/LC, and Ser/ DR. It was already known that their local destruction does not affect the EEG and the sleep-waking cycle markedly. Unexpectedly, the authors found con-siderable space-organized projections to the BF neurons from the side of the glutamatergic system of the rostral brain stem areas. This pathway begins at the rostro- lateral part of the pons from preceruleus nucleus (PC) and captures medial (MPB) and lateral (LPB) parabrachial nuclei. Interestingly, the medial parts of the pons project to the medial parts of the mag-nocellular basal nucleus, and the lateral parts, to the lateral parts of the same nucleus.
Then, the next question arises: what will happen if this pathway is destroyed? To answer the question, the authors administrated orexin-saporin targeting PC, MPB, or LPB, or unselectively the entire PB/PC complex. They found a 1.5-fold increase in the SWS percentage and 2-fold increase in the REM sleep percentage during the dark phase of nychthemeron after the destruction of LPB. The total sleep time of 24 h increased by 13%. Destruction of MPB resulted in a 2-fold increase in SWS and a 3-fold increase in REM sleep percentage during the dark phase; consequently, the total sleep time of 24 h increased by 30%. No changes in EEG spectral characteristics were revealed. Local lesions at certain points of PC selectively eliminated the hippocampal theta rhythm in EEG during REM sleep but did not change the structure of sleep-wakefulness cycle. Bilateral lesions in the adjacent areas of the pontinetegmentum, including locus ceruleus and paramedian reticular formation, did not affect the structure of sleep-wakefulness cycle either.
The results of the destruction of the entire PC/PB complex were in sharp contrast with the aforemen-tioned ones. About 10 days after the local injection of orexin-saporin, all animals unexpectedly showed a comatose-like condition similar to that after BF destruction. During the next 5-7 days, the life of the subjects was supported exclusively by glucose injec-tions. A possible reversibility of this coma by more prolonged overexposure remains unknown. EEG was the same as that after the complete destruction of BF neurons, that is, a continuous, uninterrupted stream of delta waves (<1 Hz) was recorded. Only muscle twitches were infrequently recorded during behavior evaluation; however, as in a case of BF nuclei destruc-tion, the righting reflex remained unchanged.
The authors also tested the safety of cholinergic (ACh/PPT/LDT) and NA-ergic (LC) cells in the animals. They found a few moderate lesions in PPT and LC areas only, which did not affect more than 50% of all the cells, as well as a very weak damage in LDT area (5%-10%).
Two-hour "soft" tactile stimulation revealed very weak c-Fos expression in the neocortex, as it was the case after the lesions in the BF area. However, one very important difference was observed: the expression in the hypothalamic "waking centers" His/TMN and Orex/LHA decreased by 5 times as compared to the control. Expression of the immediate early genes in the thalamus was the same as the control animals during natural sleep. At the same time, Fos expression in the midbrain "waking centers"remained at the same high level as the actively awake control animals.
Finally, the authors came to the conclusion of the presence of two ascending activating subsystems in the brain of the model animals: (1) preceruleus→medialseptum→hippocampus (PC→MS→Hipp, activa-tion of archipaleocortex, hippocampal theta rhythm); (2) parabrachial nuclei/preceruleus→basal forebrain area→neocortex (PB/PC→BF→NC, neocortical activa-tion, EEG desynchronization). It is these two parallel ventral pathways that form the critically important ascending activation system leading from the me-sopontine tegmentum and responsible for behavioral and EEG arousal reaction as well as the maintenance of the state of wakefulness, on the one hand, and the activation of neo- and archipaleocortex in REM sleep, on the other. Further results suggested that glutamatergic neurons in the LPB are necessary for hypercapnic arousals, while MPB glutamatergic ne urons play an im-portant role in promoting spontaneous waking[31, 32].
The Glu/PC/PB neurons contain the combination of REM-on and REM-waking-on cells projecting onto BF. Obviously, this system, in general, coincides with the ascending part of the Saper-Luppi's model of REM sleep regulation[4]. However, an intriguing question arises: what is the need for numerous cholinergic and aminergic cerebral systems if they are "external" to the waking mechanisms, and could their activation may be the consequence of arousal and behavioral activation rather than its cause?
Of course, the brain of humans and apparently other primates is far more complicated and sensitive to lesions than the brain of model animals such as mice, rats, and cats. It is sufficient to recall such genetic illnesses as fatal familiar insomnia[4, 33], caused by the complete destruction of reticular and other thalamic nuclei cells. The reticular thalamic nucleus forms one of those "sleep centers" whose destruction failed to induce any serious behavioral disorders in rats in the aforementioned experiments of Saper et al. However, the presence of more complicated and vulnerable systems of wakefulness regulation in humans does not disregard its evolution and the role in phylo- and ontogenesis.
The anterior hypothalamic "sleep center" was described in detail in several previous works[4, 5, 34] (Figure 1b). This "center" was firstly discovered seemingly by the famous Viennese neuroanatomist Konstantin von Economo in 1916-1917 but finally identified and localized as ventrolateral and median preoptic areas (VLPO/MnPO) using sophisticated histochemical, neurophysiological, and neurophar-macological methods after 70 years, at the end of 1980s-beginning of 1990s. The common feature of all the neurons of this center is the release of the same chemical mediator, γ-aminobutyric acid (GABA), the main inhibitory substance of the brain. In the very "sleep center" of VLPO/MnPO, GABA colocalizes with the peptide galanin (Gal), which intensifies its activity. The VLPO consists of a dense central part (core) and a more diffuse peripheral one (shell). Specific destruc-tion of these neurons in experimental conditions results in more than 2-fold decrease in the daily amount of SWS and REM sleep, although total sleep did not disappear completely. After the lesions of the core, which has important projections to the Hist/ TMN area of the posterior hypothalamus, mainly SWS is decreased, though after the destruction of the shell innervating mostly Ser/DR and NA/LC neurons, mainly REM sleep is suppressed.
These studies again raised the question of alternative "sleep centers". The presence of the "sleep center" at the level of the medulla/nuclei of the solitary tract was established in the early studies on the reticular formation (the so-called "Batini-Moruzzi bulbar syn-chronizing system"). However, the exact localization and neurochemical identification of these cells remained unknown until the end of the first decade of the 21st century. The joint efforts of the labs of Saper and the successor of Jouvet, Lin, resulted in the final identification of this center in the brain of model animals (mice and rats)[35, 36]. Initially, the authors per-formed retrograde labeling the sleep-active medullar afferents of the aforementioned "waking center" in the MPB region using a fragment of cholera toxin. They found a dense area of such cells located more lateral and dorsal to the facial nerve nucleus and named it the parafacial zone (PZ). Then, they performed selective and targeted destruction of the PZ neuronal cell bodies using antiorexin-B immunoglobulin-saporin andfound a 1.5-fold increase in the wakefulness duration during the light time phase of the day, also significant for the 24 h-period at the expense of a decrease in SWS percentage.
The following use of the novel optogenetic and chemigenetic methods for the activation and inhibi-tion of the neurons of the system parafacial zone→parabrachial zone→magnocellular nucleus of the basal forebrain→prefrontal cortex (PZ→PB→BFmc→PFC) in model animals (mice) led the authors to the following conclusions: (1) there is a dense cluster of inhibitory GABAergic neurons in the PZ area of the rostral part of medulla, which monosynaptically suppress the glutamatergic activating cells of PB; the latter, as it was mentioned above, are the most important part of the ARAS, which also activate the BFmc monosynaptically, the neurons of which in turn project onto the neocortical neurons releasing Glu, ACh, and GABA; (2) the "sleep center" in the PZ is a supplementary to the same in the hypothalamus; after its destruction or reversible switch off, the SWS percentage decrease s by two-fold not due to the shortening of its episodes (and simultaneous increase in their frequency) as it could be seen following the suppression of the "sleep center" in VLPO, but due to a decrease in the fre-quency of the episodes (without a pronounced clear change in their duration).
Optogenetic/chemogenetic stimulation of PZ cells induced some unusually long and deep SWS in mice with enormous delta waves in the EEG. This is similar to the sleep rebound after a prolonged instrumental sleep deprivation. After these sleep periods, the authors could observed the so-called "wake rebound" confirming the assumption of a homeostatic regula-tion not only of the sleep process but the waking state as well. The above assumption has repeatedly been suggested by various authors. Simultaneously, a total and prolonged suppression of REM sleep occurred within at least 9 h. The authors suggested that the medullar "sleep center" responds primarily to the nutritional inflow coming from the neighboring vague nucleus of the solitary tract. Hence, the activation of this "center" may be the reason of, for instance, the so-called "after dinner sleep", etc.
The first description of the ventral part of the feline medulla as the REM sleep generation area was suggested by the Reinoso-Suarez's group in 1994[37]. Recently, the ventro-medullary center of REM sleep, which is an analog of the aforementioned medullary center of SWS, was finally discovered and studied in detail in mice using optogenetic activation and inactivation, optrode registration, and dual retrograde tracing[38] (Figure 1c). These neurons are mostly active during REM sleep and sometimes in waking (during eating and washing). It was revealed that the induction and maintenance of REM sleep are due to the activation of the rostral GABAergic projections of these neurons and mediated (at least in part) by the inhibition of the other REM-off neurons of vlPAG, which are also GABAergic in nature. In other words, one of the main parts of the ponto-medullary system of REM sleep control is the "inhibition of the inhibitory neurons" inside this neuronal network. However, although inhibitory neurons are important in REM sleep control, there are glutamatergic neurons projecting to the LC and thalamus that may contribute to REM sleep generation, especially in the EEG activation that occurs in both waking an d REM sleep[39].
3 ConclusionsThus, it appears that the third stage in the studies on the ARAS of the brain is taking place now. The first stage began with the ARAS discovery by Magoun and Moruzzi and the following investigations using the methods of stimulation and lesion of that time mainly in acute cats. Hence, the hypothesis of a "diffuse" and "unspecific" ARAS of the brain stem was formulated. The second stage was related to using the more precise neurophysiological and histochemical methods mainly in chronically operated free-moving cats and rats. As a result, by 2010, the idea of a hierarchy of the organized system of the cerebral "waking centers" appeared. These "centers" are localized at all the levels of the cerebral axis, from the medulla to PFC, and release all the known neuromediators of low molecular weight (Glu, ACh, cerebral amines, and GABA) together with the most important neuropeptides (orexin/hypocretin, gal anin, MCH, and others). The lateral hypothalamic neurons releasing orexin/hypocretin peptide and the cells of TMN of posterior hypothalamus releasing histamine play the central role in this complicated system.
With the optogenetic and chemigenetic methods as well as other sophisticated skills of the 21st century, the aforementioned hypothesis has been revised. The Glu-activating system has been discovered and described in detail, which passes through the ventral pathway from the rostral pontine area of the brain stem to the BF nuclei and, subsequently, to neo- and archipaleocortex. Presumably, this system induces an arousal reaction and maintains the cortex in the state of tonic depolarization during wakefulness and REM sleep. At the same time, the activity of all other "waking centers" is probably the result of the cortical activation. In this case, the question of specific brain activation in REM sleep arises again: what is its nature if the same ARAS is involved in both cases (waking and REM sleep), and is the "silencing" of aminergic cerebral systems during REM sleep the consequence rather than the cause of the activation?
The peripheral part of VLPO (eVLPO) inhibits GABA- ergic REM-off cells (a type of the "switching off the break", that is, switching on as a result) localized in vlPAG and LPT. The same cells are also inhibited by the medullar "center" of REM sleep. Hence, it suggests that the general sleep-promoting system appears to be not less complicated, if not more, than the ARAS itself. How does the relationship between these numerous medullary and hypothalamic NREM and REM "sleep centers" take place if they have no direct anatomical connection? What is the role of the dorsal ("classical", thalamic) pathway brain stem → cortex in model animals as well as in humans?
The answers to all these intriguing questions will be found by future generations of neuroscientists.
| [1] | Berger H. Üeber das elektroenkephalogramm des menschen: IL Mitteilung. J Psychol Neurol , 1930, 40 : 160–179. |
| [2] | Pelayo R, Dement WC. History of sleep physiology and medicine. In Principles and Practice of Sleep Medicine, 6th ed. Kryger M, Roth T, Dement WC, Eds. Philadelphia, PA: Elsevier, 2016, pp 3-14. |
| [3] | Moruzzi G, Magoun H. Brain stem reticular formation and activation of the EEG. Electroencephalogr Clin Neurophysiol , 1949, 1 (4) : 455–473. |
| [4] | Koval'zon VM. Central mechanisms of the sleep-wakefulness cycle control. Hum Physiol , 2011, 37 (4) : 500–508. DOI:10.1134/S0362119711040116 |
| [5] | Brown RE, Basheer R, McKenna JT, Strecker RE, McCarley RW. Control of sleep and wakefulness. Physiol Rev , 2012, 92 (3) : 1087–1187. DOI:10.1152/physrev.00032.2011 |
| [6] | Lim MM, Szymusiak R. Neurobiology of arousal and sleep: updates and insights into neurological disorders. Curr Sleep Med Rep , 2015, 1 (2) : 91–100. DOI:10.1007/s40675-015-0013-0 |
| [7] | Aston-Jones G, Chen S, Zhu Y, Oshinsky ML. A neural circuit for circadian regulation of arousal. Nat Neurosci , 2001, 4 (7) : 732–738. DOI:10.1038/89522 |
| [8] | Steriade M, Timofeev I, Grenier F. Natural waking and sleep states: a view from inside neocortical neurons. J Neurophysiol , 2001, 85 (5) : 1969–1985. |
| [9] | Steriade M, Timofeev I. Neuronal plasticity in thalamocortical networks during sleep and waking oscillations. Neuron , 2003, 37 (4) : 563–576. DOI:10.1016/S0896-6273(03)00065-5 |
| [10] | Timofeev I, Grenier F, Steriade M. Disfacilitation and active inhibition in the neocortex during the natural sleep-wake cycle: An intracellular study. Proc Natl Acad Sci USA , 2001, 98 (4) : 1924–1929. DOI:10.1073/pnas.98.4.1924 |
| [11] | Timofeev I, Bazhenov M, Seigneur J, Sejnowski T. Neuronal synchronization and thalamocortical rhythms duringsleep, wake, and epilepsy. InJasper's Basic Mechanisms of the Epilepsies. 4th edn. Noebels JL, Avoli M, Rogawski MA, Olsen RW, Delgado-Escueta AV, Eds. New York: Oxford University Press, 2012 : 157 . |
| [12] | Richter C, Woods IG, Schier AF. Neuropeptidergic control of sleep and wakefulness. Annu Rev Neurosci , 2014, 37 : 503–531. DOI:10.1146/annurev-neuro-062111-150447 |
| [13] | Parvizi J, Damasio AR. Neuroanatomical correlates of brainstem coma. Brain , 2003, 126 (7) : 1524–1536. DOI:10.1093/brain/awg166 |
| [14] | Siegel JM. Rapid eye movement sleep. InPrinciples and Practice of Sleep Medicine, 6th ed. Kryger M, Roth T, Dement WC, Eds. Philadelphia PA: Elsevier, 2016, pp 78-95. |
| [15] | Koval'zon VM. The role of histaminergic system of the brain in the regulation of sleep-wakefulness cycle. Hum Physiol , 2013, 39 (6) : 574–583. DOI:10.1134/S0362119713060078 |
| [16] | Koval'zon VM. Brain and sleep: from neurons -to the molecules. Zh Vyssh Nerv Deiat Im I P Pavlova , 2013, 63 (1) : 48–60. |
| [17] | Kovalzon VM, Zavalko IM. The neurochemistry of the sleep-wakefulness cycle and Parkinson's disease. J Neurochem , 2013, 7 (3) : 171–183. DOI:10.1134/S1819712413030069 |
| [18] | Blanco-Centurion C, Gerashchenko D, Shiromani PJ. Effects of saporin-induced lesions of three arousal populations on daily levels of sleep and wake. J Neurosci , 2007, 27 (51) : 14041–14048. DOI:10.1523/JNEUROSCI.3217-07.2007 |
| [19] | Denoyer M, Sallanon M, Buda C, Kitahama K, Jouvet M. Neurotoxic lesion of the mesencephalic reticular formation and/or the posterior hypothalamus does not alter waking in the cat. Brain Res , 1991, 539 (2) : 287–303. DOI:10.1016/0006-8993(91)91633-C |
| [20] | Jouvet M. Le sommeil, la conscience et l'éveil. Paris: Odile Jacob, 2016, p 216. |
| [21] | Carter ME, Yizhar O, Chikahisa S, Nguyen H, Adamantidis A, Nishino S, Deisseroth K, de Lecea L. Tuning arousal with optogenetic modulation of locus coeruleus neurons. Nat Neurosci , 2010, 13 (12) : 1526–1533. DOI:10.1038/nn.2682 |
| [22] | Kaur S, Junek A, Black MA, Semba K. Effects of ibotenate and 192IgG-saporin lesions of the nucleus basalis magnocellularis/ substantia innominata on spontaneous sleep and wake states and on recovery sleep after sleep deprivation in rats. J Neurosci , 2008, 28 (2) : 491–504. DOI:10.1523/JNEUROSCI.1585-07.2008 |
| [23] | Gerashchenko D, Chou TC, Blanco-Centurion CA, Saper CB, Shiromani PJ. Effects of lesions of the histaminergic tuberomammillary nucleus on spontaneous sleep in rats. Sleep , 2004, 27 (7) : 1275–1281. |
| [24] | Lu J, Jhou TC, Saper CB. Identification of wake-active dopaminergic neurons in the ventral periaqueductal gray matter. J Neurosci , 2006, 26 (1) : 193–202. DOI:10.1523/JNEUROSCI.2244-05.2006 |
| [25] | Shouse MN, Siegel JM. Pontine regulation of REM sleep components in cats: integrity of the pedunculopontine tegmentum (PPT) is important for phasic events but unnecessary for atonia during REM sleep. Brain Res , 1992, 571 (1) : 50–63. DOI:10.1016/0006-8993(92)90508-7 |
| [26] | Webster HH, Jones BE. Neurotoxic lesions of the dorsolateralpontomesencephalic tegmentum-cholinergic cell area in the cat. Ⅱ. Effectsupon sleep-waking states. Brain Res , 1988, 458 (2) : 285–302. DOI:10.1016/0006-8993(88)90471-4 |
| [27] | Saper CB, Fuller PM, Pedersen NP, Lu J, Scannell TE. Sleep state switching. Neuron , 2010, 68 (6) : 1023–1042. DOI:10.1016/j.neuron.2010.11.032 |
| [28] | Fuller P, Sherman D, Pedersen NP, Saper CB, Lu J. Reassessment of the structural basis of the ascending arousal system. J Comp Neurol , 2011, 519 (5) : 933–956. DOI:10.1002/cne.v519.5 |
| [29] | Villablanca JR. Counterpointing the functional role of the forebrain and of thebrainstem in the control of the sleep-waking system. J Sleep Res , 2004, 13 (3) : 179–208. DOI:10.1111/j.1365-2869.2004.00412.x |
| [30] | Steriade M, McCarley RW. Brainstem Control of Wakefulness and Sleep. US: Springer, 1990. |
| [31] | Xu M, Chung S, Zhang SY, Zhong P, Ma CY, Chang WC, Weissbourd B, Sakai N, Luo LQ, Nishino S. Basal forebrain circuit for sleep-wake control. Nat Neurosci , 2015, 18 (11) : 1641–1647. DOI:10.1038/nn.4143 |
| [32] | Kaur S, Pedersen NP, Yokota S, Hur EE, Fuller PM, Lazarus M, Chamberlin NL, Saper CB. Glutamatergic signaling from the parabrachial nucleus plays a critical role in hypercapnic arousal. J Neurosci , 2013, 33 (18) : 7627–7640. DOI:10.1523/JNEUROSCI.0173-13.2013 |
| [33] | Peng B, Zhang S, Dong H, Lu Z. Clinical, histopathological and genetic studies in a case of fatal familial insomnia with review of the literature. Int J Clin Exp Pathol , 2015, 8 (9) : 10171–10177. |
| [34] | Haas HL, Lin JS. Waking with the hypothalamus. Pflugers Arch Eur J Physiol , 2012, 463 (1) : 31–42. DOI:10.1007/s00424-011-0996-4 |
| [35] | Anaclet C, Ferrari L, Arrigoni E, Bass CE, Saper CB, Lu J, Fuller PM. The GABAergic parafacial zone is a medullary slow wave sleep-promoting center. Nat Neurosci , 2014, 17 (9) : 1217–1224. DOI:10.1038/nn.3789 |
| [36] | Anaclet C, Lin JS, Vetrivelan R, Krenzer M, Vong L, Fuller PM, Lu J. Identification and characterization of a sleep-active cell group in the rostral medullary brainstem. J Neurosci , 2012, 32 (50) : 17970–17976. DOI:10.1523/JNEUROSCI.0620-12.2012 |
| [37] | Reinoso-Suárez F, De Andrés I, Rodrigo-Angulo LM, Rodríguez-Veiga E. Location and anatomical connections of a paradoxical sleep induction site in the cat ventral pontine tegmentum. Eur J Neurosci , 1994, 6 (12) : 1829–1836. DOI:10.1111/ejn.1994.6.issue-12 |
| [38] | Weber F, Chung S, Beier KT, Xu M, Luo LQ, Dan Y. Control of REM sleep by ventral medulla GABAergic neurons. Nature , 2015, 526 (7573) : 435–438. DOI:10.1038/nature14979 |
| [39] | Luppi PH, Clément O, Fort P. Paradoxical (REM) sleep genesis by the brainstem is under hypothalamic control. Curr Opin Neurobiol , 2013, 23 (5) : 786–792. DOI:10.1016/j.conb.2013.02.006 |



