2. Catholic Neuroscience Institute, College of Medicine, the Catholic University of Korea, Seoul 06591, Republic of Korea;
3. Department of Neurosurgery, Chosun University Hospital, College of Gwangju 61452, Republic of Korea;
4. Department of Neurosurgery, Nowon Eulji Hospital, College of Medicine, Eulji University, Seongnam 826802, Republic of Korea
Deep brain stimulation (DBS) of the somatosensory thalamus and periventricular/periaqueductal gray matter (PVG/PAG) has been used to treat chronic intractable pain syndromes when all other modalities have failed[1-6]. Although the exact mechanism underlying the analgesic effect of thalamic DBS is uncertain[7-9], it probably involves activation of the thalamocortical pathways and changes in cortical activity[7-9]. Despite encouraging reports on the efficacy of MCS for various types of neuropathic pain, its exact analgesic mechanism also remains controversial[10, 11]. However, a number of studies using positron emission tomography have indicated that the thalamus is a key structure involved in mediating the functional effects of MCS[10-13]. Therefore, it appears that MCS and thalamic DBS share a common pathophysiologic analgesic mechanism, i.e., the modulation of thalamic activity[14]. However, the application of MCS and DBS is empirical, and there is no consensus on when MCS or DBS should be used or which one is better[14].
We have performed DBS 32 times and MCS 23 times in selected patients with intractable neuropathic pain since 2000. Among the patients treated for refractory neuropathic pain over the last 16 years, 11 underwent a simultaneous trial of DBS and MCS in an effort to increase the success rate of the initial invasive intervention, and possibly enhance the long-term outcomes by selecting more effective stimulation targets in an individual patient. The results of the simultaneous trial of DBS/MCS in nine initial consecutively recruited patients were reported in 2014[14]. Of the nine patients who were initially implanted with both thalamic ventralis caudalis (Vc) and MCS electrodes, eight (89%) had successful trials, of which six (75%) responded to MCS, and the remaining two responded to Vc DBS. During the long-term follow-up (40 months), the six MCS and two Vc DBS responders reported approximately 37%-38% pain relief in the last follow-up period. Based on these results, we suggested that MCS would be a more reasonable initial approach considering the success rate and the less invasive nature of epidural MCS compared with DBS[14]. A thoughtful criticism against the simultaneous trial of DBS/MCS was raised because of the risks associated with DBS and the lack of a double-blind stimulation[15].
After the first report of the initial combined trial of DBS/MCS[14], we included two more patients in the simultaneous trial. Here, we summarize the longterm results of a simultaneous trial of DBS/MCS for refractory neuropathic pain and summarize the current pathophysiologic mechanisms of invasive neuromodulation. We also raise our concerns about the problem of outcome assessment, defined as >50% long-term pain relief.
2 Patients and methods 2.1 Demographics and inclusion criteriaOf the 32 patients who underwent thalamic DBS and 23 patients who underwent MCS for the treatment of various kinds of chronic neuropathic pain in the last 16 years (2000-2016), a simultaneous trial of DBS and MCS was performed in 11 patients. Patients who were referred for surgery had been refractory to previous maximal medical and physical therapies, i.e., their pain did not respond to maximal medical treatments, including anticonvulsants, antidepressants, and opioids. A trial of spinal cord stimulation was not effective in three of these cases. A simultaneous trial of MCS and DBS was considered for patients with a minimum visual analogue score of 7/10 and those with definite neuropathic pain, both central and peripheral, with prolonged duration (>1 year). Among the patients with refractory neuropathic pain who were candidates for treatment with DBS or MCS, a simultaneous trial of MCS and DBS was proposed for patients who fully understood the additional risks of exploring the multiple intracranial targets associated with the combined use of DBS/MCS to enhance the chances of a successful trial stimulation, and it was performed for those who provided informed consent. Patients were excluded if they had significant psychological or psychosocial overlay and/or secondary gains.
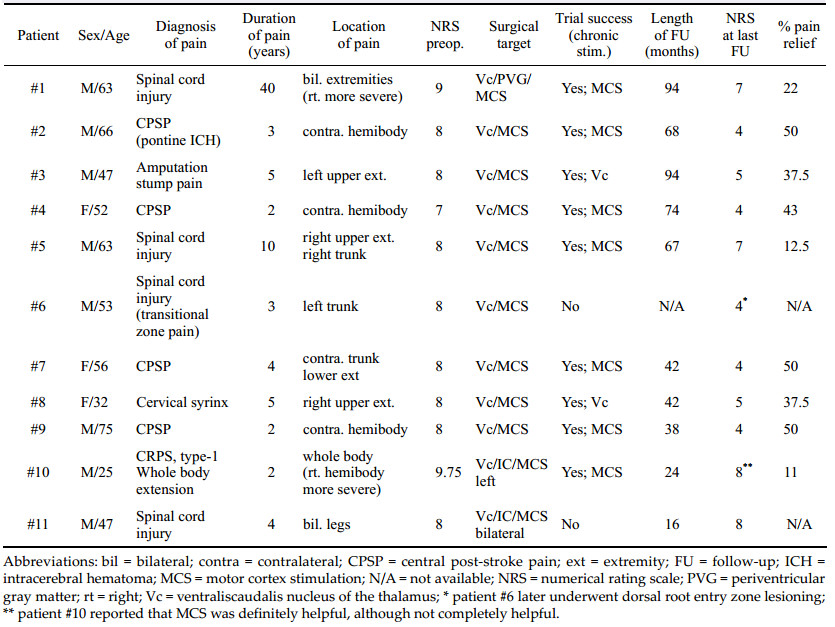
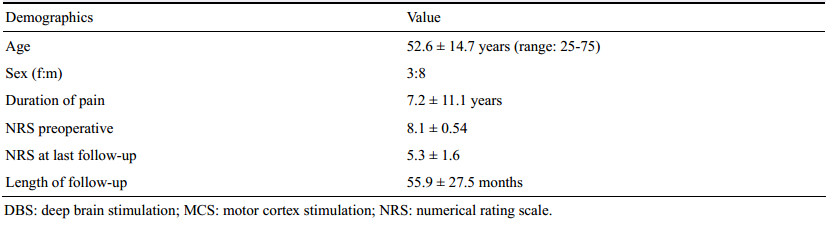
The demographics of the patients included in this trial are summarized in Tables 1, 2 and 3. The mean age of the patients was 52.6 years (range: 25-75; men: women: 8:3), and the mean duration of follow-up was 52.6 ± 14.7 months (range: 24-94 months). The type of pain experienced by the patients included central post-stroke pain (n = 4), spinal cord injury (n = 5), below-level neuropathic pain from C4 cord injury, thoracolumbar shell injury, transitional zone pain, iatrogenic spinal cord transection, central pain of the cervical syrinx, amputation stump pain in the arm, and complex regional pain syndrome type-Ⅰ (CRPS-1) with whole body extension.
The procedures for the simultaneous implantation ofelectrodes for DBS and MCS are described in detail in our previous reports regarding the surgical treatment of refractory neuropathic pain[16-20]. After we took stereotactic magnetic resonance images (MRI-Achieva 1.5T, Phillips, Eindhoven, the Netherlands), we transferred the obtained imaging data to the surgical navigation system, which was used to define the target coordinates of the Vc and the precentral hand knob (Figure 1a). The locations of the central sulcus and the hand knob were also marked on the scalp. The electrodes were implanted into the Vc nucleus of the thalamus and internal capsule contralateral to the side of the most severe pain under local anesthesia, and then electrodes were implanted over the precentral gyrus for MCS under general anesthesia. The tentative anatomical target for the Vc was 2-3 mm anterior to the posterior commissure (PC; y coordinates), with the z coordinates at the level of the anterior commissure (AC)-PC plane. The x coordinates were 14-15 mm for upper extremity pain and 16-17 mm for lower extremity pain.
|
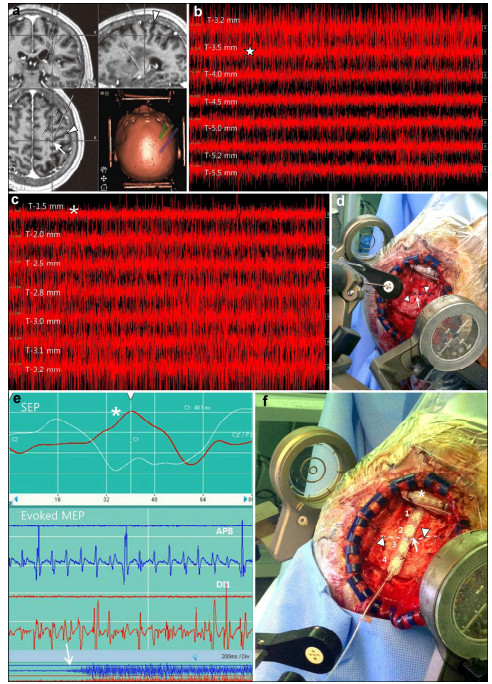
| Figure 1 Combined thalamic ventralis caudalis (Vc) deep brain stimulation (DBS) and motor cortex stimulation (MCS) in patient #4. a An image captured from the stereotactic planning software (Framelink®, Medtronic, Minneapolis, MN) during the determination of the initial target coordinates and trajectory planning for the Vc and the hand knob in the precentral gyrus. A white arrow indicates the location of the hand knob corresponding to the hand somatotopy of the precentral gyrus. An arrowhead indicates the central sulcus. b A typical microelectrode recording during exploration of the thalamic Vc nucleus. In the area T-3.5 mm, increased firing of the thalamic sensory nucleus is observed (star), which indicates entrance into the part of the Vc nucleus corresponding to a cutaneous core of the somatotopy. c A microelectrode recording shows an abrupt decrease in the firing of the Vc nucleus (asterisk). This means that the tip of the microelectroderecording probe is located below the thalamic Vc nucleus and is within the white matter. d An intraoperative photograph showing the localization of the precentral gyrus and the central sulcus. An asterisk denotes the implantation of the Vc DBS electrode before MCS. Arrow heads indicate the marking of the course of the central sulcus over the dura mater and an arrow indicates the location of the target for the precentral gyrus. e An image showing the phase reversal (asterisk) of the somatosensory evoked potential (SEP) in the upper image. The lower image shows a motor evoked potential (MEP) response taken from the abductor policis brevis (APB) and the first dorsal interossoeus muscles (DI1) through macrostimulation of the contacts of the paddle lead over the precentral gyrus. An arrow shows the elicitation of the MEP in the contralateral hand muscles. f An intraoperative photograph showing the placement of paddle lead for MCS. The contact "2" of the paddle lead is placed on the target for the precentral gyrus. A dotted line (arrowheads) denotes the course of the central sulcus and an arrow indicate the location of the predetermined MCS target. |
After a burr hole and dural opening were made, microelectrode recording and stimulation were used to map the Vc targets (Figure 1b). The activity of the individual firing units and their receptive fields (RF), defined as the body sites where tactile stimulation or movement evoked changes in the electrical firing patterns, was recorded along the planned trajectories (mostly three or four trajectories in one session) (Figure 1c). Based on the RF mapping, a DBS electrode (Medtronic model 3387, Medtronic Inc., Minneapolis, MN) was implanted in the target. Macrostimulation was conducted through the DBS leads to verify stimulation-induced paresthesia coverage of the painful area and any adverse effects of the macrostimulation. After the absence of an adverse neurologic deficit was confirmed with DBS, general intravenous anesthesia was administered. An inverted U-shaped scalp incision was made just behind the frontal incision for the DBS, and an approximately 5-cm sized craniotomy was performed along the course of the precentral gyrus and the central sulcus (CS)[6-20] (Figure 1d). The CS was localized with the aid of phase-reversal of the somatosensory evoked potential and evoked EMG response of the muscle (Figure 1e). After the location and course of the CS were confirmed, a paddle electrode (Lamitrode 44®, St. Jude Medical, Plano, Texas, or Resume® TL, model 3986A, Medtronic Inc., Minneapolis, MN) was placed in the epidural space (Figure 1f). Further, the distal ends of the paddle and DBS electrodes were connected to a percutaneous extension cable through the subcutaneous tunnel (Figure 2).
|
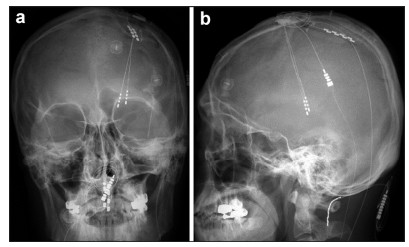
| Figure 2 Anterior-posterior a and lateral b projections of skull x-ray films of a patient (#10) who underwent a simultaneous DBS/ MCS trial for intractable, chronic neuropathic pain. This patient had both thalamic Vc and internal capsule DBS electrodes combined with a left-side MCS electrode. Note the previously implanted C1–C2 paddle lead for treatment-resistant complex regional pain syndrome with whole-body involvement, which was more severe in the right hemibody. In this particular patient, we obtained >50% pain relief with the initial MCS trial. However, thalamic Vc stimulation aggravated the hemibody pain. The prominent analgesic effect of MCS faded postoperatively within six months. However, the patient still today (24 months after MCS) uses chronic MCS for unremitting pain. He judged MCS as being much more effective than spinal cord stimulation. |
An unblined trial was conducted for approximately 14 days after the DBS and MCS electrodes were inserted to assess efficacy. For the Vc and internal capsule DBS, stimulation was performed at 30-130 Hz, 90-120 μs, and voltage intensities of up to 7-8 V, and the four electrode contacts of each DBS lead were systematically explored. In the MCS, various combinations of bipolar stimulation centered at the M1 were performed at 10-180 Hz, 90-500 μs, and voltage intensities of up to 7-10 V. If the insertion effect of the Vc DBS[21] was prominent and it was difficult to assess the effect of the post-operative trial stimulation, we waited a number of days until the patient's preoperative pain recurred. Upon pain recurrence, these patients were re-admitted, and new percutaneous extensions were connected to the electrodes under local anesthesia; a stimulation trial was then conducted.
A postoperative stimulation trial was considered to be successful if a reduction of more than 50% in the visual analogue scale (VAS) was achieved with the stimulation. If the stimulation was successful, the electrodes were connected to a pulse generator (IPG, Soletra®, model 7426, Medtronic Inc., MN, or Eon-mini®, St. Jude Medical, Plano, TX) using a transaxillary, subpectoral approach[22]. Patients who did not have a successful stimulation trial had the DBS and MCS electrodes and the percutaneous extensions removed. Primary outcome variables were examined as follows: (1) whether or not patients were using DBS and/or MCS; (2) the patients' ratings of their worst pain as recorded on the numeric rating scale (NRS, 0-10)[23] in the last follow-up; (3) the percentage of pain relief on the NRS. The baseline NRS was obtained before surgery and postoperatively every two to three months after the procedure. We did not control modifications in the drug regimens over time.
The descriptive statistics are reported as frequencies and percentages. Paired t-test and chi-square analyses were performed to assess the relationships between surgery and outcomes. All statistical analyses were performed with SPSS software, version 12.0 (SPSS Inc., Chicago, IL). The values were considered statistically significant at P < 0.05.
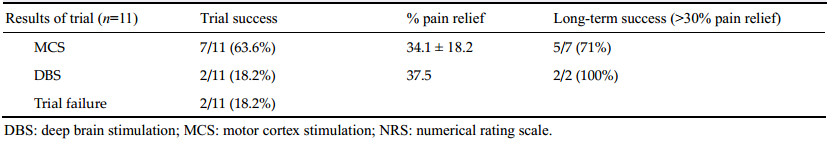
3 Results 3.1 Initial pain relief with simultaneous DBS/MCS trialsThe demographic characteristics and long-term outcomes of the simultaneous trial of DBS/MCS in our series are summarized in Table 1. Of the 11 patients initially implanted with both DBS and MCS electrodes, nine (81.8%) had successful trials. Two of the 11 patients did not obtain >50% pain relief and had the electrodes removed.
Seven of the 11 patients (63.6%) responded to MCS, attaining more than 50% pain relief and two of the 11 (18.2%) responded to Vc DBS. We could not find any synergistic effect of analgesia from combined Vc DBS and MCS stimulation during the two week trial period. A prolonged insertion effect (two weeks), according to implantation of the Vc thalamic DBS[23] was observed in patient #8. A severe squeezing and deep pressurelike intermittent pain was almost completely relieved after the insertion of the electrode in the Vc and progressively reappeared after two weeks; she had a later trial of simultaneous DBS/MCS after re-admission, and Vc DBS was chosen for chronic stimulation.
3.2 Long-term outcomes and complicationsWith a mean follow-up duration of 56 ± 27.5 months (mean ± standard deviation, n = 9, range: 24-96 months), the mean NRS decreased from 8.1 ± 0.54 to 5.3 ± 1.6 (P < 0.05, paired-t test). The percentage pain relief (PPR) values in the chronic MCS (n = 7) and chronic DBS (n = 2) groups were 34.1% ± 18.2% (mean ± standard deviation, range: 11.1%-50%) and 37.5%, respectively. There was no difference in the degree of pain relief between the MCS and DBS groups (chi-square test, P = 0.807). With more than four years of long-term follow-up, five of the seven patients treated with MCS (71%) and both patients treated with DBS were assessed, with long-term success defined as >30% pain relief compared to baseline.
There was no mortality or neurologic morbidity related to the simultaneous DBS and MCS procedures. A superficial wound infection necessitated wound revision with antibiotic chemotherapy in patient #9 with chronic MCS. Patient #11 experienced postoperative pneumonia and respiratory distress syndrome after operation under general anesthesia because his respiratory condition was compromised owing to his preexisting high cervical (C4) spinal cord injury.
4 Discussion 4.1 Exploration of multiple subcortical targets for DBS in painAlthough DBS is associated with an inherent risk of penetration of the brain parenchyme, DBS has a unique advantage for exploring multiple potential subcortical targets related to the pathophysiology of neuropathic pain. Indeed, DBS-lead implantation in more than two targets during a single session has already been reported in studies on DBS for refractory neuropathic pain. Among the traditional subcortical targets for neuropathic pain, various combinations of periaqueductal gray mater (PAG) with the Vc, the PVG with the PAG, and the PAG with the internal capsule (IC) have been the most commonly reported[1-6]. Thus, the concept of exploring multiple subcortical targets for pain relief is not new.
4.2 Traditional pathophysiological concepts in PVG/PAG DBS for painThe traditional consensus about selecting subcortical targets for pain relief is to recommend stimulating the PVG/PAG for intractable nociceptive pain, whereas the sensory thalamus (Vc) or the IC have been advocated as the preferred targets for neuropathic pain[6]. The reason for selecting the PVG/PAG for nociceptive pain was partly based on laboratory and clinical results that indicated that pain relief from PVG/PAG stimulation depended on the release of endogenous opioids[24, 25]. Some studies have reported that the analgesic effect induced by stimulating the central gray matter is reversed by the administration of naloxone[24-26]. In addition to the activation of the descending opioid system, increased medial dorsal nucleus activation in the thalamus, the ascending pathway from the PVG, and an area associated with the limbic system with extensive connections with the amygdala and cingulate cortex were observed during stimulation of the PVG[6, 27]. However, subsequent studies showed the idea that PAG DBS acts by augmenting endogenous opioid release is contentious[28]. It was shown that the opioid metabolite values in the cerebrospinal fluid may be artifacts[29, 30]. No cross-tolerance between DBS and morphine, or similar reversibility between naloxone and a saline placebo, has been shown[25, 28].
4.3 Analgesic mechanism of thalamic Vc DBS for painThe analgesic effect induced by stimulating the sensory thalamus is still not understood. Mazars[27] originally stimulated the ventroposterior thalamus in patients experiencing deafferentation pain, based on the theory[31] that this pain is caused by a lack of proprioceptive stimuli reaching the thalamus. Stimulating the primary somatosensory pathway at this thalamic site was an effort to compensate for the lack of normal sensory input[27]. The gate control theory[32] further championed the idea that stimulating low-threshold somatosensory pathways inhibits pain; thus, direct stimulation of this pathway at the thalamic level would be expected to reduce neuropathic pain, which is characterized by loss of such input after damage occurs to the peripheral or central nervous system[7]. Stimulating the ventral posterior lateral (VPL) thalamus in anesthetized animals inhibited the activity of both spinothalamic nociceptive neurons in a monkey[33] and thalamic parafascicular nociceptive neurons in a rat[34]. A positron emission tomography (PET) study showed that sensory thalamic stimulation activated a thalamocortical pain modulation circuit that also involved thermal pathways such as the insular cortex[7]. Thus, stimulating the thalamic sensory nucleus may be mediated by activation of the inhibitory thalamocorticalcorticofugal pathway, which prevents the pathological spread of painful stimuli.
4.4 Mechanism of action of motor cortex stimulationDespite encouraging reports on the efficacy of MCS in various types of neuropathic pain, its exact analgesic mechanism remains controversial[10, 11]. However, authors of a number of studies have indicated that the thalamus is a key structure involved in mediating the functional effects of MCS[10-13]. Studies on groups of patients with intractable pain of various origins have confirmed the existence of significant cerebral blood flow (CBF) increases during MCS in the lateral and medial thalamus, the anterior cingulate-orbitofrontal cortex (BA32), the anterior insula-medial temporal lobe, and the upper brain stem near the PAG[10]. The thalamic areas that showed the most significant CBF changes were the ventrolateral and ventroanterior nuclei, i.e., the thalamic region directly connected with the motor and premotor cortices. Another regional CBF study[11] that used 18F-fluorodeoxyglucose PET in nine patients with post-stroke pain treated with MCS showed that successful MCS significantly improved glucose use in the thalamus ipsilateral to the MCS, suggesting that MCS activated the corticothalamic connection from the motor cortex. These results provide evidence that descending axons, rather than apical dendrites, are primarily activated by MCS, and they highlight the thalamus as the key structure involved in mediating the functional effects of MCS[10-13, 35].
Meanwhile, the findings from other PET studies[36-38] suggested that MCS could enhance the release of endogenous opioids in various brain structures; this effect correlated with pain relief when the release was observed in the cingulate cortex and PAG[36]. Furthermore, MCS could activate descending pathways, leading to reinforced or restored inhibitory control of nociceptive transmission in the dorsal horns of the spinal cord[35]. Concurrent with these findings, Nguyen et al.[39] suggested that the mechanisms of action of MCS could also involve inhibitory intracortical and interneuronal circuits in the precentral M1 cortex, acting by reinforcing intracortical gamma-aminobytyric acid (GABA)-ergic inhibition. According to many studies regarding the analgesic mechanisms of DBS and MCS, sensory thalamic DBS and MCS appear to share a common analgesic mechanism, i.e., modulation of thalamic activity[39].
4.5 Rationale for a combined trial of DBS/MCS for painThe use of MCS and thalamic DBS is still considered empirical and off-label because reliable outcomes across etiologies are lacking[40, 41]. Furthermore, the indications of DBS and MCS are generally overlapping. Owing to the invasive nature of the intervention and the inherent risks of deep-brain penetration associated with DBS, DBS has been the last resort for patients whose pain has not responded to maximal medical and physical treatment for years and also for even less invasive spinal cord stimulation[40-42]. The unfavorable outcomes associated with traditional DBS in chronic pain, especially central pain syndromes[43], marked the advent in the early 1990s of a novel, and less invasive, alternative, i.e., epidural motor cortex stimulation[43-45]. In a subsequent studies, the efficacy of MCS was witnessed among patients with trigeminal neuropathic pain[46], and MCS has also been reported to be effective in peripheral neuropathic pain[16, 47-52].
Since MCS has, to a large extent, replaced DBS as a last resort for invasive pain treatment, the indications for DBS and MCS in the literature are nearly the same. For example, complex regional pain syndrome type Ⅱ was shown to respond very well to MCS by Rasche et al.[53]. The analgesic effect of MCS in CRPS type Ⅱ with hemibody extension was first reported by Son et al.[16], and the effectiveness of MCS in CRPS was further replicated by others[54, 55]. Similarly, the indications are overlapping between MCS and DBS, and there is no consensus on when MCS or DBS should be applied or which technique is better. We performed a simultaneous trial of thalamic DBS and MCS and investigated which one was more successful in treating chronic intractable neuropathic pain.
In our simultaneous trial, we chose the Vc thalamic nucleus rather than the PVG/PAG. The reason for selecting the Vc thalamus was that the Vc had been regarded as the preferred target for neuropathic pain[3, 4], and the patients included in our study showed typically severe, chronic, and neuropathic pain. Another reason was that we did not observe any significant analgesic effect from PVG electrodes simultaneously implanted in the thalamic Vc in six other patients in our early experience with DBS for various types of neuropathic pain. When we began DBS for chronic neuropathic pain in 2000, we thought that both the PVG and PAG would have the same mechanism of endogenous opioid-induced analgesia and we were reluctant to place the electrode deeper into the PAG owing to our limited experience. Although favorable responses to PVG DBS had been reported in some studies[6, 56, 57], we could not reproduce any positive responses from stimulating the PVG.
No synergistic or additive analgesic effects were observed during the two week trial of stimulating the thalamic Vc and the primary motor cortex. Indeed, some of the patients receiving sensory thalamic stimulation perceived the induced paresthesia of the sensory thalamic stimulation as an unpleasant, dysesthetic sensation. Although the mechanism of action of MCS is known to be closely linked to thalamic modulation[10-13, 35], we did not observe any additive analgesic effect using a combined stimulation. Further neurophysiologic studies addressing a possible link between the subcortical and cortical targets of pain modulation are warranted.
4.6 Outcome assessment: >50% and >30% reduction in pain scoresWe currently define long-term DBS/MCS treatment as successful when the patient achieves a significant mean reduction in the NRS score of at least 30% compared with the baseline[58, 59]. The reason for considering a 30% mean reduction as a successful outcome is the lack of consistency across studies regarding the method used to evaluate outcomes, as highlighted by Fontaine et al.[58] and others[8, 60-63]. For example, a 40% reduction in pain has been considered a good outcome by some authors, whereas others have required an improvement of >50%[21, 58]. In addition, we feel that there is also a problem in assessing outcomes using four-tier grading (poor, fair, good, and excellent).
It was suggested that this kind of variability in the outcome measures used in clinical trials hinders the evaluation of the efficacy and effectiveness of treatments, and the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) guidelines[61]. In these guidelines[61], a 30% reduction in pain is considered a moderately important change and is equal to “much improved†as measured by the Patient Global Impression of Change (PGIC)[61, 62, 64]. It is now commonly used in the subjective evaluation of clinical trials for different kinds of pain treatment[62]. The problem associated with a mere description of “more than 40% to 50% pain relief†in evaluating the efficacy of a specific treatment has already been reported[62-65]. For example, Sears et al.[63] documented that 29.4% of the patients with failed back surgery syndrome reported a 50% or greater reduction in pain after spinal cord stimulation (SCS) with paddle leads at a mean follow-up time-point of 3.8 years. However, 70.6% of the same group of patients reported that they were satisfied with the surgery to the point of undergoing it again for the same outcome. This discrepancy between VAS and NRS outcomes and overall satisfaction has been reported in SCS studies as well as in a study of MCS for chronic pain by Nuti et al.[65].
Indeed, a >30% improvement on the VAS in the long-term follow-up (>42 months) was observed in a recent, large-scale study of DBS for neuropathic pain by Boccard et al.[66]. Among 85 patients treated with DBS for various types of chronic neuropathic pain[66], 74 (87%) responded to an initial stimulation. Among these 74 patients assessed as initial DBS responders, a two-year long-term follow-up was possible for 22 patients (30%), and 15 patients (18.7%) were followedup for more than four years. Interestingly, the degree of pain relief in the long-term follow-up (four years) assessed with the VAS in those regarded as chronic DBS responders was 36% (±39.2, standard deviation)[66]. Concurrent with this, we observed similar degrees of pain relief in 16 patients treated with chronic MCS in the long-term (53 ± 39 months) follow-up study, and defined a successful outcome as >30% pain relief compared with baseline (P < 0.05, χ2 test)[18]. The PPR values in patients with central post-stroke pain and peripheral neuropathic pain were 40.7% ± 8.85% and 34.6% ± 7.08%, respectively, with >4 years of chronic treatment with MCS[18]. Meanwhile, numerous reports have described >80% pain relief (so-called excellent on a four-tier scale) at the one-year follow-up following treatment with invasive neuromodulation using MCS and DBS. Considering our experiences of using DBS and MCS for chronic refractory neuropathic pain, in our opinion, it is difficult to imagine that such a high analgesic effect would last for more than three or four years.
Achieving >30% pain relief with significantly longterm (>4 years) effects in chronic neuropathic pain appears difficult even with SCS, a widely applied neuromodulation technique. There is good consensus that SCS is effective for complex regional pain syndrome, TypeⅠ (CRPS-1)[67-69]. However, it was shown that only 41% of the patients treated with SCS for CPRS-1 experienced at least >30% pain relief with a median follow-up time-point of 5.2 years, and 63% of 84 patients with chronically implanted SCS who had positive outcomes underwent SCS explantation over the 12 years of follow-up[62]. Considering that a population-based cohort study of 102 patients with CRPS indicated that 31% had remained incapable of working two years after the diagnosis[70], the long-term efficacy and the continued use of SCS were regarded as being of major clinical importance[62]. Assuming that the candidates for DBS or MCS are the patients with similarly intractable, chronic neuropathic pain syndromes such as central post-stroke pain or spinal cord injury, >30% pain relief with invasive brain modulation techniques during a long-term follow-up of more than 3 years should be regarded significant.
5 ConclusionsWe achieved a high success rate (81.8%) in a simultaneous trial of DBS and MCS for refractory, chronic, and neuropathic pain. With our selection of possibly more effective chronic stimulation targets between the thalamus and motor cortex from this trial, five out of seven patients undergoing MCS chronically and both patients undergoing DBS chronically had a successful outcome, attaining >30% pain relief.
Conflict of interests
All contributing authors have no conflict of interests.
| [1] | Hosobuchi Y. Subcortical electrical stimulation for control of intractable pain in humans. J Neurosurg, 1986, 64(4): 543–553. DOI:10.3171/jns.1986.64.4.0543 |
| [2] | Young RF, Kroening R, Fulton W, Feldman RA, Chambi I. Electrical stimulation of the brain in treatment of chronic pain. Experience over 5 years. J Neurosurg, 1985, 62(3): 389–396. DOI:10.3171/jns.1985.62.3.0389 |
| [3] | Levy RM, Lamb S, Adams JE. Treatment of chronic pain by deep brain stimulation: Long-term follow-up and review of the literature. Neurosurgery, 1987, 21(6): 885–893. DOI:10.1227/00006123-198712000-00017 |
| [4] | Kumar K, Toth C, Nath RK. Deep brain stimulation for intractable pain: A 15-year experience. Neurosurgery, 1997, 40(4): 736–746. DOI:10.1097/00006123-199704000-00015 |
| [5] | Turnbull IM, Shulman R, Woodhurst WB. Thalamic stimulation for neuropathic pain. J Neurosurg, 1980, 52(4): 486–493. DOI:10.3171/jns.1980.52.4.0486 |
| [6] | Bittar RG, Kar-Purkayastha I, Owen SL, Bear RE, Green A, Wang SY, Aziz TZ. Deep brain stimulation for pain relief: A meta-analysis. J Clin Neurosci, 2005, 12(5): 515–519. DOI:10.1016/j.jocn.2004.10.005 |
| [7] | Duncan GH, Kupers RC, Marchand S, Villemure JG, Gybels JM, Bushnell MC. Stimulation of human thalamus for pain relief: Possible modulatory circuits revealed by positron emission tomography. J Neurophysiol, 1998, 80(6): 3326–3330. |
| [8] | Rezai AR, Lozano AM, Crawley AP, Joy MLG, Davis KD, Kwan CL, Dostrovsky JO, Tasker RR, Mikulis DJ. Thalamic stimulation and functional magnetic resonance imaging: localization of cortical and subcortical activation with implanted electrodes. Technical note. J Neurosurg, 1999, 90(3): 583–590. DOI:10.3171/jns.1999.90.3.0583 |
| [9] | Davis KD, Taub E, Duffner F, Lozano AM, Tasker RR, Houle S, Dostrovsky JO. Activation of the anterior cingulate cortex by thalamic stimulation in patients with chronic pain: A positron emission tomography study. J Neurosurg, 2000, 92(1): 64–69. DOI:10.3171/jns.2000.92.1.0064 |
| [10] | Peyron R, García-Larrea LG, Deiber MP, Cinotti L, Convers P, Sindou M, Mauguière F, Laurent B. Electrical stimulation of precentral cortical area in the treatment of central pain: Electrophysiological and PET study. Pain, 1995, 62(3): 275–286. DOI:10.1016/0304-3959(94)00211-V |
| [11] | García-Larrea LG, Peyron R, Mertens P, Gregoire MC, Lavenne F, Bonnefoi F, Mauguière F, Laurent B, Sindou M. Positron emission tomography during motor cortex stimulation for pain control. Stereotact Funct Neurosurg, 1997, 68(1-4): 141–148. |
| [12] | Peyron R, García-Larrea LG, Grégoire MC, Convers P, Lavenne F, Veyre L, Froment JC, Mauguière F, Michel D, Laurent B. Allodynia after lateral-medullary (Wallenberg) infarct. A PET study. Brain, 1998, 121(2): 345–356. DOI:10.1093/brain/121.2.345 |
| [13] | Ito M, Kuroda S, Shiga T, Tamaki N, Iwasaki Y. Motor cortex stimulation improves local cerebral glucose metabolism in the ipsilateral thalamus in patients with poststroke pain: Case report. Neurosurgery, 2011, 69(2): E462–469. DOI:10.1227/NEU.0b013e318218cfa0 |
| [14] | Son B, Kim D, Kim H, Lee S. Simultaneous trial of deep brain and motor cortex stimulation in chronic intractable neuropathic pain. Stereotact Funct Neurosurg, 2014, 92(4): 218–226. DOI:10.1159/000362933 |
| [15] | Lévêque M, Weil AG, Nguyen JP. Simultaneous deep brain stimulation/motor cortex stimulation trial for neuropathic pain: Fishing with dynamite?. Stereotact Funct Neurosurg, 2015, 93: 219. DOI:10.1159/000368198 |
| [16] | Son B, Kim MC, Moon DE, and Kang JK. Motor cortex stimulation in a patient with intractable complex regional pain syndrome type II with hemibody involvement. J Neurosurg, 2003, 98(1): 175–179. DOI:10.3171/jns.2003.98.1.0175 |
| [17] | Son B. Reply to the letter by Lévêque et al. entitled 'Simultaneous deep brain stimulation/motor cortex stimulation trial for neuropathic pain: Fishing with dynamite?' Stereotact Funct Neurosurg, 2015, 93: 220–221. |
| [18] | Im S, Ha S, Kim D, Son B. Long-term results of motor cortex stimulation in the treatment of chronic, intractable neuropathic pain. Stereotact Funct Neurosurg, 2015, 93: 212–218. DOI:10.1159/000381557 |
| [19] | Son B, Lee SW, Choi ES, Sung JH, Hong JT. Motor cortex stimulation for central pain following a traumatic brain injury. Pain, 2006, 123(1): 210–216. DOI:10.1016/j.pain.2006.02.028 |
| [20] | Son B, Han S, Kim D, Lee S. Motor cortex stimulation for postcordotomy dysesthetic pain. Int J Pain, 2012, 3: 60–66. |
| [21] | Hamani C, Schwalb JM, Rezai AR, Dostrovsky JO, Davis KD, Lozano AM. Deep brain stimulation for chronic neuropathic pain: Long-term outcome and the incidence of insertional effect. Pain, 2006, 125(1): 188–196. DOI:10.1016/j.pain.2006.05.019 |
| [22] | Son B, Han SH, Choi Y, Kim M, Yang S, Lee S. Transaxillary subpectoral implantation of implantable pulse generator for deep brain stimulation. Neuromodulation, 2012, 15(3): 260–266. DOI:10.1111/ner.2012.15.issue-3 |
| [23] | McCaffery M, Beebe A. Pain: Clinical Manual for Nursing Practice. London: Mosby, 1994 . |
| [24] | Boivie J, Meyerson BA. A correlative anatomical and clinical study of pain suppression by deep brain stimulation. Pain, 1982, 13(2): 113–136. DOI:10.1016/0304-3959(82)90022-7 |
| [25] | Young RF, Chambi VI. Pain relief by electrical stimulation of the periaqueductal and periventricular gray matter.Evidence for a non-opioid mechanism. J Neurosurg, 1987, 66(3): 364–371. DOI:10.3171/jns.1987.66.3.0364 |
| [26] | Richardson DE, Akil H. Pain reduction by electrical brain stimulation in man. Part 1: Acute administration in periaqueductal and periventricular sites. J Neurosurg, 1977, 47: 178–183. DOI:10.3171/jns.1977.47.2.0178 |
| [27] | Mazars G, Merienne L, Cioloca C. Treatment of certain types of pain with implantable thalamic stimulators. Neuro-Chirurgie, 1974, 20(2): 117–124. |
| [28] | Pereira EAC, Boccard SG, Aziz TZ. Deep brain stimulation for pain: Distinguishing dorsolateral somesthetic and ventromedial affective targets. Neurosurgery, 2014, 61: 175–181. DOI:10.1227/NEU.0000000000000397 |
| [29] | Fessler RG, Brown FD, Rachlin JR, Mullan S, Fang VS. Elevated beta-endorphin in cerebrospinal fluid after electrical brain stimulation: Artefact of contrast infusion?. Science, 1984, 224(6452): 1017–1019. |
| [30] | Dionne RA, Mueller GP, Young RF, Greenberg RP, Hargreaves KM, Gracely R, Dubner R. Contrast medium causes the apparent increase in β-endorphin levels in human cerebrospinal fluid following brain stimulation. Pain, 1984, 20(4): 313–321. DOI:10.1016/0304-3959(84)90109-X |
| [31] | Head J, Holmes G. Sensory disturbances from cerebral lesions. Brain, 1911, 34(2-3): 102–254. DOI:10.1093/brain/34.2-3.102 |
| [32] | Melzack R, Wall PD. Pain mechanisms: A new theory. Science, 1965, 150(3699): 971–979. DOI:10.1126/science.150.3699.971 |
| [33] | Gerhart KD, Yezierski RP, Fang ZR, Willis WD. Inhibition of primate spinothalamic tract neurons by stimulation in ventral posterior lateral (VPLc) thalamic nucleus: Possible mechanisms. J Neurophysiol, 1983, 49(2): 406–423. |
| [34] | Benabid AL, Henriksen SJ, McGinty JF, Bloom FE. Thalamic nucleus ventro-postero-lateralis inhibits nucleus parafascicularis response to noxious stimuli through a non-opioid pathway. Brain Res, 1983, 280(2): 217–231. DOI:10.1016/0006-8993(83)90052-5 |
| [35] | García-Larrea L, Peyron R, Mertens P, Laurent B, Mauguière F, Sindou M. Functional imaging and neurophysiological assessment of spinal and brain therapeutic modulation in humans. Arch Med Res, 2000, 31(3): 248–257. DOI:10.1016/S0188-4409(00)00083-7 |
| [36] | Maarrawi J, Peyron R, Mertens P, Costes N, Magnin M, Sindou M, Laurent B, García-Larrea L. Motor cortex stimulation for pain control induces changes in the endogenous opioid system. Neurology, 2007, 69(9): 827–834. DOI:10.1212/01.wnl.0000269783.86997.37 |
| [37] | Maarrawi J, Peyron R, Mertens P, Costes N, Magnin M, Sindou M, Laurent B, García-Larrea L. Brain opioid receptor density predicts motor cortex stimulation efficacy for chronic pain. Pain, 2013, 154(11): 2563–2568. DOI:10.1016/j.pain.2013.07.042 |
| [38] | Peyron R, Faillenot I, Mertens P, Laurent B, García-Larrea L. Motor cortex stimulation in neuropathic pain. Correlations between analgesic effect and hemodynamic changes in the brain. A PET study. Neuroimage, 2007, 34(1): 310–321. DOI:10.1016/j.neuroimage.2006.08.037 |
| [39] | Nguyen JP, Nizard J, Keravel Y, Lefaucheur JP. Invasive brain stimulation for the treatment of neuropathic pain. Nat Rev Neurol, 2011, 7: 699–709. DOI:10.1038/nrneurol.2011.138 |
| [40] | Raslan AM. Deep brain stimulation for chronic pain: Can it help?. Pain, 2006, 120(1-2): 1–2. DOI:10.1016/j.pain.2005.10.026 |
| [41] | Plow EB, Pascual-Leone A, Machado A. Brain stimulation in the treatment of chronic neuropathic and non-cancerous pain. J Pain, 2012, 13(5): 411–424. DOI:10.1016/j.jpain.2012.02.001 |
| [42] | Machado AG, Mogilner AY, Rezai AR. Motor cortex stimulation for persistent non-cancer pain. In Textbook of Stereotactic and Functional Neurosurgery. Lozano AM, Gildenberg PL, Tasker RR, Eds. Berlin Heidelberg: Springer, 2009, pp 2239-2249. |
| [43] | Tsubokawa T, Katayama Y, Yamamoto T, Hirayama T, Koyama S. Chronic motor cortex stimulation in patients with thalamic pain. J Neurosurg, 1993, 78(3): 393–401. DOI:10.3171/jns.1993.78.3.0393 |
| [44] | Tsubokawa T, Katayama Y, Yamamoto T, Hirayama T, Koyama S. Chronic motor cortex stimulation for the treatment of central pain. In Advances in Stereotactic and Functional Neurosurgery 9. Hitchcock ER, Broggi G, Burzaco J, Martin-Rodriguez J, Meyerson BA, Tóth S, Eds. Vienna: Springer, 1991, pp 137-139. |
| [45] | Tsubokawa T., Katayama Y, Yamamoto T, Hirayama T, Koyama S. Treatment of thalamic pain by chronic motor cortex stimulation. Pacing Clin Electrophysiol, 1991, 14(1): 131–134. DOI:10.1111/pace.1991.14.issue-1 |
| [46] | Meyerson BA, Lindblom U, Linderoth B, Lind G, Herregodts P. Motor cortex stimulation as treatment of trigeminal neuropathic pain. In Advances in Stereotactic and Functional Neurosurgery 10. Meyerson BA, Broggi G, Martin-Rodriguez J, Ostertag C, Sindou M, Eds. Vienna: Springer, 1993, pp 150-153. |
| [47] | Nguyen JP, Keravel Y, Feve A, Uchiyama T, Cesaro P, Le Guerinel C, and Pollin B. Treatment of deafferentation pain by chronic stimulation of the motor cortex: report of a series of 20 cases. Acta Neurochir Suppl, 1997, 68: 54–60. |
| [48] | Saitoh Y, Shibata M, Hirano S, Hirata M, Mashimo T, Yoshimine T. Motor cortex stimulation for central pain and peripheral deafferentation pain. Report of eight cases. J Neurosurg, 2000, 92(1): 150–155. DOI:10.3171/jns.2000.92.1.0150 |
| [49] | Nguyen JP, Velasco F, Brugières P, Velasco M, Keravel Y, Boleaga B, Brito F, Lefaucheur JP. Treatment of chronic neuropathic pain by motor cortex stimulation: Results of a bicentric controlled crossover trial. Brain Stimul, 2008, 1(2): 89–96. DOI:10.1016/j.brs.2008.03.007 |
| [50] | Velasco F, Argüelles C, Carrillo-Ruiz JD, Castro G, Velasco AL, Jiménez F, Velasco M. Efficacy of motor cortex stimulation in the treatment of neuropathic pain: A randomized double-blind trial. J Neurosurg, 2008, 108(4): 698–706. DOI:10.3171/JNS/2008/108/4/0698 |
| [51] | Lefaucheur JP, Drouot X, Cunin P, Bruckert R, Lepetit H, Créange A, Wolkenstein P, Maison P, Keravel Y, Nguyen JP. Motor cortex stimulation for the treatment of refractory peripheral neuropathic pain. Brain, 2009, 132(6): 463–1471. |
| [52] | Raslan AM, Nasseri M, Bahgat D, Abdu E, Burchiel KJ. Motor cortex stimulation for trigeminal neuropathic or deafferentation pain: An institutional case series experience. Stereotact Funct Neurosurg, 2011, 89(2): 83–88. DOI:10.1159/000323338 |
| [53] | Rasche D, Rinaldi PC, Young RF, Tronnier VM. Deep brain stimulation for the treatment of various chronic pain syndromes. Neurosurg Focus, 2006, 21(6): E1–8. |
| [54] | Velasco F, Carrillo-Ruiz JD, Castro G, Argüelles C, Velasco AL, Kassian A, Guevara U. Motor cortex electrical stimulation applied to patients with complex regional pain syndrome. Pain, 2009, 147(1): 91–98. DOI:10.1016/j.pain.2009.08.024 |
| [55] | Fonoff ET, Hamani C, de Andrade DC, Yeng LT, Marcolin MA, Teixeira MJ. Pain relief and functional recovery in patients with complex regional pain syndrome after motor cortex stimulation. Stereotact Funct Neurosurg, 2011, 89(3): 167–172. DOI:10.1159/000324895 |
| [56] | Nandi D, Smith H, Owen S, Joint C, Stein J, Aziz T. Peri-ventricular grey stimulation versus motor cortex stimulation for post stroke neuropathic pain. J Clin Neurosci, 2002, 9(5): 557–561. DOI:10.1054/jocn.2001.1042 |
| [57] | Owen SLF, Green AL, Nandi D, Bittar RG, Wang SY, Aziz TZ. Deep brain stimulation for neuropathic pain. Neuromodulation, 2006, 9(2): 100–106. DOI:10.1111/ner.2006.9.issue-2 |
| [58] | Fontaine D, Hamani C, Lozano A. Efficacy and safety of motor cortex stimulation for chronic neuropathic pain: Critical review of the literature. J Neurosurg, 2009, 110(2): 251–256. DOI:10.3171/2008.6.17602 |
| [59] | Coffey RJ, Lozano AM. Neurostimulation for chronic noncancer pain: An evaluation of the clinical evidence and recommendations for future trial designs. J Neurosurg, 2006, 105(2): 175–189. DOI:10.3171/jns.2006.105.2.175 |
| [60] | Lefaucheur JP, Ménard-Lefaucheur I, Goukon C, Keravel Y, Nguyen JP. Predictive value of rTMS in the identification of responders to epidural motor cortex stimulation therapy for pain. J Pain, 2011, 12(10): 1102–1111. DOI:10.1016/j.jpain.2011.05.004 |
| [61] | Dworkin RH, Turk DC, Wyrwich KW, Beaton D, Cleeland CS, Farrar JT, Haythornthwaite JA, Jensen MP, Kerns RD, Ader DN, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain, 2008, 9(2): 105–121. DOI:10.1016/j.jpain.2007.09.005 |
| [62] | Geurts JW, Smits H, Kemler MA, Brunner F, Kessels AGH, van Kleef M. Spinal cord stimulation for complex regional pain syndrome type I: A prospective cohort study with longterm follow-up. Neuromodulation, 2013, 16(6): 523–529. DOI:10.1111/ner.12024 |
| [63] | Sears NC, Machado AG, Nagel SJ, Deogaonkar M, Stanton-Hicks M, Rezai AR, Henderson JM. Long-term outcomes of spinal cord stimulation with paddle leads in the treatment of complex regional pain syndrome and failed back surgery syndrome. Neuromodulation, 2011, 14(4): 312–318. DOI:10.1111/ner.2011.14.issue-4 |
| [64] | Forouzanfar T, Weber WEJ, Kemler M, van Kleef M. What is a meaningful pain reduction in patients with complex regional pain syndrome type 1?. Clin J Pain, 2003, 19(5): 81–285. |
| [65] | Nuti C, Peyron R, Garcia-Larrea L, Brunon J, Laurent B, Sindou M, Mertens P. Motor cortex stimulation for refractory neuropathic pain: Fur year outcome and predictors of efficacy. Pain, 2005, 118(1): 43–52. DOI:10.1016/j.pain.2005.07.020 |
| [66] | Boccard SGJ, Pereira EAC, Moir L, Aziz TZ, Green AL. Long-term outcomes of deep brain stimulation for neuropathic pain. Neurosurgery, 2013, 72(2): 221–231. DOI:10.1227/NEU.0b013e31827b97d6 |
| [67] | Kemler MA, Barendese GAM, van Kleef M, de Vet HCW, Rijks CPM, Furnée CA, van den Wildenberg FAJM. Spinal cord stimulation in patients with chronic reflex sympathetic dystrophy. N Eng J Med, 2000, 343(9): 618–624. DOI:10.1056/NEJM200008313430904 |
| [68] | Turner KA, Loeser JD, Deyo RA, Sanders SB. Spinal cord stimulation for patients with failed back surgery syndrome or complex regional pain syndrome: A systematic review of effectivenss and complications. Pain, 2004, 108(2): 137–147. |
| [69] | Kemler MA, de Vet HCW, Barendse GAM, van den Wildenberg FAJM, van Kleef M. Effect of spinal cord stimulation for chronic complex regional pain syndrome Type I: Five-year final follow-up of patients in a randomized controlled trial. J Neurosurg, 2008, 108(2): 292–298. DOI:10.3171/JNS/2008/108/2/0292 |
| [70] | de Mos M, Huyen FJPM, van den Hoeven-Borgman M, Dieleman JP, Ch Stricker BH, Sturkenboom MCJM. Outcome of the complex regional pain syndrome. Clin J Pain, 2009, 25(7): 590–597. DOI:10.1097/AJP.0b013e3181a11623 |