Hao Li, male, 1971. 8, chief physician, M.D, P.h.D, Doctoral tutor, Director of neurosurgery in Children's Hospital of Fudan University, Member of Pediatric Neurosurgery Committee of Chinese Medical Association, Member of Pediatric Neurosurgery Expert Committee of Chinese Medical Doctor Association.
Medulloblastoma is a malignant, invasive embryonal tumor that arises in the cerebellum or fourth ventricle and accounts for 12%-25% of central nervous system tumors. It is the most common malignancy in children with an annual incidence of five per 100, 000 children < 15 years of age[1]. Although surgery remains the major treatment for medulloblastoma, there is still controversy regarding the effect of resection on the prognosis of patients with medulloblastoma. Furthermore, in cases of medulloblastoma adherent to the brainstem, complete resection is extremely difficult. In addition, metastasis via the cerebrospinal fluid is common; thus, patients with medulloblastoma often have a poor prognosis and a high mortality[2].
Clinically, the prognosis of patients with medulloblastoma is often determined according to the pathological type, which is also used in the choice of adjunctive therapies, such as radiotherapy and chemotherapy[3, 4]. Currently, the World Health Organization (WHO) classification system for medulloblastoma is based on histomorphology. However, patients with the same pathological type of medulloblastoma still have distinct genetic backgrounds. Therefore, the prognosis of patients with medulloblastoma with the same WHO pathological type may vary. In addition, tumor stage, extent of resection, age, and other factors are used for prognosis determination, but this approach is susceptible to the influence of other clinical factors, leading to poor accuracy. Thus, it is imperative to identify reliable parameters for the assessment of risk and prognosis of patients with medulloblastoma.
Recent studies on medulloblastoma have demonstrated that it is more accurate to stratify risk based on the molecular phenotype, which is also helpful to guide clinical treatment and determine the clinical prognosis. Currently, medulloblastoma is divided into several subtypes according to the molecular phenotypes: WNT, Sonic hedgehog (SHH), and nonSHH/WNT[5, 6]. In addition, Ellison et al.[7] differentiated medulloblastoma subtypes by immunohistochemistry, and they validated their findings using microarray analysis.
Gene expression profiling of medulloblastoma has shown that Yes-associated protein 1 (YAP1), an auxiliary initiator of oncogene transcription that can promote cell proliferation and transformation[7], is highly expressed in the WNT and SHH subtypes of medulloblastoma[8, 9]. In addition to YAP1, GRB2-associated protein 1 (GAB1) has a role in the SHH signaling pathway in medulloblastoma cells. However, few studies have assessed their prognostic value for patients with medulloblastoma.
This retrospective observational study aimed to evaluate the association of molecular subtype, clinical characteristics, and pathological types with the outcomes of medulloblastoma patients. The primary outcome was overall survival (OS). Risk factors associated with survival, disease progression, and recurrence were analyzed by univariate Cox regression analysis, and the identified significant risk factors were further analyzed via Kaplan-Meier survival curves. YAP1 served as a specific marker of the WNT and SHH subtypes and GAB1 was used as a specific marker of the SHH subtype. The primary goal was to identify information pertinent to prognostication and making treatment decisions for children with medulloblastoma.
2 Materials and methods 2.1 Study designIn this retrospective analysis, we reviewed the medical records of 40 children with pathologically proven medulloblastoma who underwent surgical resection at the Affiliated Children's Hospital of Fudan University between January 2004 and June 2014. Medulloblastoma was classified as the classic subtype, desmoplastic/ nodular subtype, extensive nodularity subtype, or large cell/anaplastic subtype according to the 2007 WHO classification system of central nervous system tumors.
The inclusion criteria were the absence of another severe disease diagnosis, complete medical records with follow-up data, and available medulloblastoma tissues that were fixed in 10% neutral formalin and embedded in paraffin following resection. All of the children died of disease progression or recurrence; however, those who died within 3 days after surgery were excluded. This study was approved by the Institutional Review Board of Fudan University.
2.2 Survival analysisThe OS time was defined as the time interval between surgery and death or the last follow-up and was expressed as months. The disease progression-free survival (PFS) time was defined as the time interval between date of surgery and date of progression, last follow-up, or death. The recurrence-free survival time was calculated from the date of surgery to date of diagnosis of recurrence, last follow-up, or death. Censored data were considered if the patient was alive at the last follow-up and was marked in the survival curve.
2.3 ImmunohistochemistryImmunochemistry analysis of YAP1 and GAB1 expression was performed as previously described[7]. After surgical resection, medulloblastoma tissues were fixed in 4% neutral formalin and embedded in paraffin. The paraffin-embedded tissues provided by the Department of Pathology in the Affiliated Children's Hospital of Fudan University were cut into 4 μm sections. After routine processing with xylene, graded ethanol solutions, and 3% H2O2 for 10 min, antigen retrieval was performed in 0.05 M citrate buffer (pH = 6.0) at 100 ℃ for 5-10 min and blocked in goat serum for 10 min. Immunohistochemistry analysis was performed using a EnVision kit with an anti-GAB1 polyclonal antibody (1:50, Abcam; Cambridge, MA, USA) and an anti-YAP1 polyclonal antibody (1:50, Abcam) for 1 h at 37 ℃. After washing, sections were incubated in HRP-conjugated goat anti-rabbit IgG (H + L) (Jackson ImmunoResearch; West Grove, PA, USA) for 1 h at 37 ℃. Visualization was performed with DAB.
The immunohistochemistry results were semiquantified. Five fields were randomly selected from each section at a magnification of 200×, and the positive cells were counted and averaged. Cells positive for YAP1 had brown granules in the nucleus and cytoplasm; GAB1-positive cells had granules in the nucleus. Known strongly positive tissues served as a positive control. At a magnification of 400×, if the proportion of positive cells was ≥30% within a strong positive area, it was scored as positive staining; otherwise, it was scored as negative.
2.4 Statistical analysisAll data were presented as frequency and percentage. A Cox proportional hazard model was performed to identify effectors of poor survival outcome. To quantify the strength of association between a potential risk factor and death, a hazard ratio (HR) and its 95% confidence interval (CI) were estimated and reported. Mutivariable analysis was not implemented due to the limited sample size. A P < 0.05 was defined as statistically significant. All statistical analyses were two-sided and were performed using IBM SPSS Statistics for Windows version 22.0 (IBM Corp., Armonk, NY, USA).
3 Results 3.1 Patient demographicsPatients' characteristics are summarized in Table 1. Among all 40 patients, most were boys (72.5%) and ≥3 years of age. Clinical features of central medulloblastoma (90%), complete tumor resection (85%), and classic subtype (82.5%) were found in the majority of patients. At least 80% of patients had M stage at M0 and T stage at T3 or above (Table 1). Calcification, cerebrospinal fluid fistula, or cerebral herniation were found in 20%-35% of patients. While 57.5% of patients received radiotherapy, only 35% were treated with chemotherapy after surgery. The Karnofsky Performance Scale (KPS) score was ≥80 in 80% of the patients.
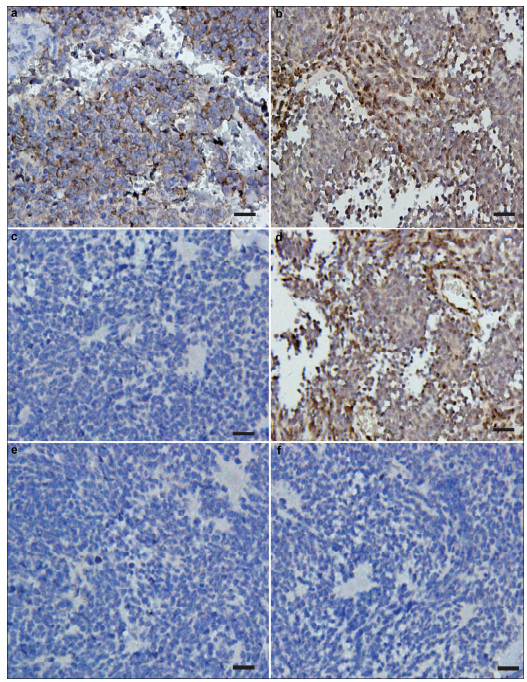
The medulloblastoma subtypes were identified using YAP1 and GAB1 immunohistochemistry analysis. Representative images of the WNT, SHH, and nonSHH/WNT subtypes are shown in Figure 1. As shown in Table 1, 20% of tumors were the WNT subtype, and 32.5% were the SHH subtype. The remaining 47.5% of patients had the non-SHH/WNT subtype of medulloblastoma.
|
| Figure 1 Immunohistochemical analysis of YAP and GAB1 to identify medulloblastoma subtypes. (a, b) The SHH subtype was positive for both (a) YAP1 and (b) GAB1. (c, d) The WNT subtype was positive for YAP1 and negative for GAB1. (e, f) The non-SHH/WNT subtype was negative for both YAP1 and GAB1. Magnification, 400×; scale bar, 50 μm. |
|
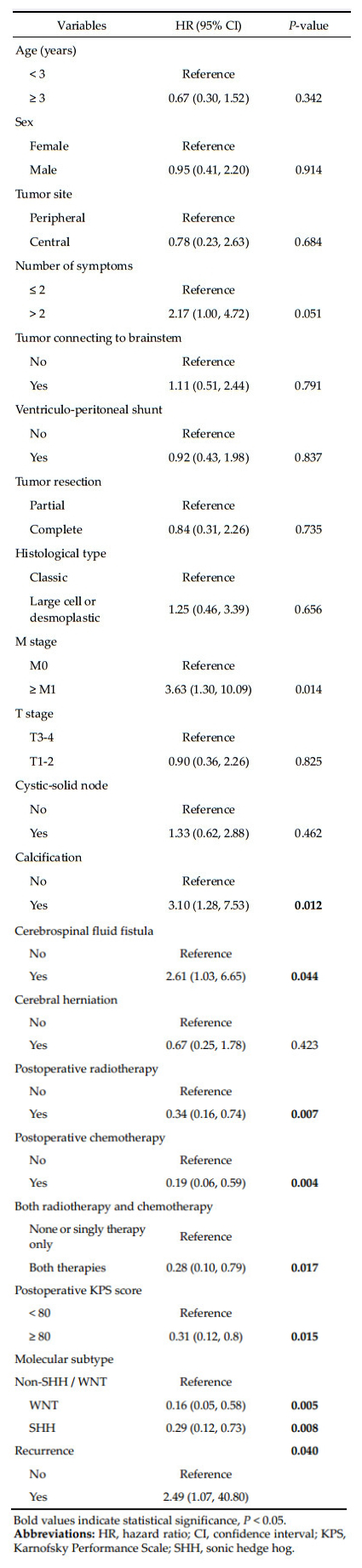
The factors associated with OS of patients with medulloblastoma are shown in Table 2. Univariate analysis indicated that patients with M0 stage, those with M1 stage (HR = 3.63, 95% CI: 1.30-10.09, P = 0.014), calcification (HR = 3.10, 95% CI: 1.28-7.53, P = 0.012), or cerebrospinal fluid fistula (HR = 2.61, 95% CI: 1.03-6.65, P = 0.044) were at significantly greater risk of a shorter survival. Treatment with radiotherapy, chemotherapy, or both following surgical resection positively affected patient survival. The HRs were 0.34 (95% CI: 0.16-0.74, P = 0.007) for radiotherapy, 0.19 (95% CI: 0.06-0.59, P = 0.004) for chemotherapy, and 0.28 (95% CI: 0.10-0.79, P = 0.017) for both therapies. A postoperative KPS score ≥80 was also associated with a lower risk of shorter survival (HR = 0.31, 95% CI: 0.12-0.80, P = 0.015). Compared to patients with non-SHH/WNT tumors, those with WNT (HR = 0.16, 95% CI: 0.05-0.58, P = 0.005) or SHH (HR = 0.29, 95% CI: 0.12-0.73, P = 0.008) subtypes were less likely to have a poor outcome.
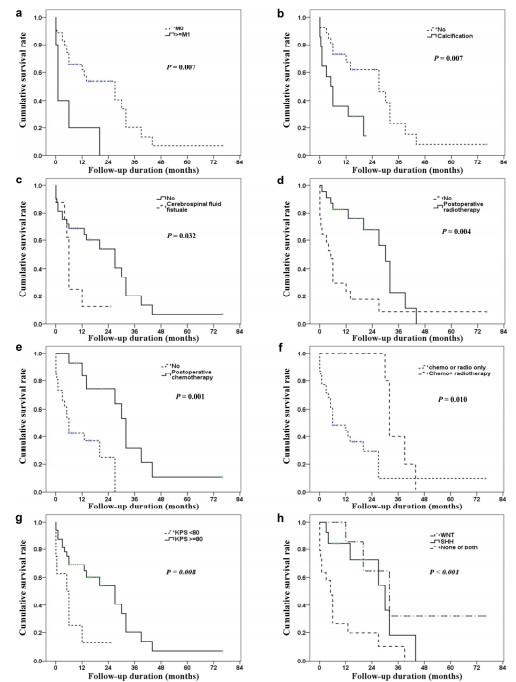
3.4 Kaplan-Meier survival analysisThe Kaplan-Meier curves by factors found to be associated with OS are shown in Figure 2. Four out of five patients with M1 stage or above died within half a year after surgery, with OS rates of 20% at 6 months and 0% at 20 months (Figure 2a). OS rates in patients with M0 stage were 61.8% at 1 year, 54.1% at 2 years, 20.3% at 3 years, and 6.8% after 44 months (Figure 2a). Eleven out of 14 patients with calcification died within 13 months after surgery, with OS rates of 35.7% at 6 months and 14.3% after 20 months (Figure 2b). OS rates in patients without calcification were 67.5% at 1 year, 61.8% at 2 years, 23.2% at 3 years, and 7.7% after 44 months (Figure 2b). The OS rates for patients with cerebrospinal fluid fistula were 25% at 6 months and 12.5% after 12 months (Figure 2c). In patients without cerebrospinal fluid fistula, the OS was 68.8% at 1 year, 54.1% at 2 years, 20.3% at 3 years, and 6.8% after 44 months (Figure 2c).
|
| Figure 2 Kaplan-Meier curves of overall survival according to (a) M stage, (b) calcification, (c) cerebral fluid fistula, (d) postoperative radiotherapy, (e) postoperative chemotherapy, (f) postoperative adjuvant therapy, (g) postoperative KPS score, (h) molecular subtype. The log-rank test was used to test the survival status between groups. |
Whereas the OS rate in patients not receiving radiotherapy was 23.5% at 1 year, it was 67.8% at 2 years (Figure 2d). OS rates in those not treated with chemotherapy were 42.3% at 1 year, 24.7% at 2 years, and 0% at 27 months as compared to 83.6% at 1 year, 74.3% at 2 years, 31.8% at 3 years, and 10.6% after 44 months in those treated with chemotherapy (Figure 2e). The OS rate in patients receiving both chemotherapy and radiotherapy was 100% at 2 years and 40% at 3 years versus 44.4% at 1 year and 29.0% at 2 years in those without chemotherapy or radiotherapy (Figure 2f).
Seven out of 8 patients with postoperative KPS < 80 died within a year of surgery, with OS rates of 50% at 6 months and 12.5% after 12 months. In contrast, those with postoperative KPS scores of ≥80 had OS rates of 68.8% at 1 year, 54.1% at 2 years, 20.3% at 3 years, and 6.8% after 44 months (Figure 2g). The 1-year, 2-year, 3-year, and terminal OS rates were 85.7%, 64.3%, and 32.1% in patients with the WNT subtype, respectively; 84.6%, 72.5%, 18.1%, and 0% in patients with the SHH subtype, respectively; and 26.3%, 19.7%, 9.9%, and 0% in those with non-SHH/ WNT subtypes, respectively (Figure 2h).
3.5 Univariate analysis of potential predictors of disease progressionAs shown in Table 3, the number of symptoms, M stage, and postoperative radiotherapy were associated with a higher risk of disease progression. Patients with ≥2 symptoms had a 2.61-fold higher risk of disease progression (95% CI: 1.10-6.19, P = 0.029). In addition, those with M1 stage or above had a 20.76 times (95% CI: 3.77-114.29, P < 0.001) higher risk of disease progression. Finally, postoperative radiotherapy was protective against disease progression (HR = 0.39; 95% CI: 0.16-0.93, P = 0.033).

|
Univariate analysis indicated that recurrence was associated with a higher risk of death (HR = 2.49, 95% CI: 1.07-40.80, P = 0.040; Table 2); the 1-and 3-year OS rates of patients without recurrence were 70% and 42% compared to 43.7% and 0% in those with recurrence. We next determined factors associated with recurrence by univariate analysis. The risk of recurrence increased with advanced M stage (HR = 30.71; 95% CI: 3.92-240.44, P = 0.001). In contrast, patients receiving both chemotherapy and radiotherapy were less likely to have recurrence as compared to those with only one therapy or without either radiotherapy or chemotherapy (HR = 0.24, 95% CI: 0.05-0.93, P = 0.040; Table 4).
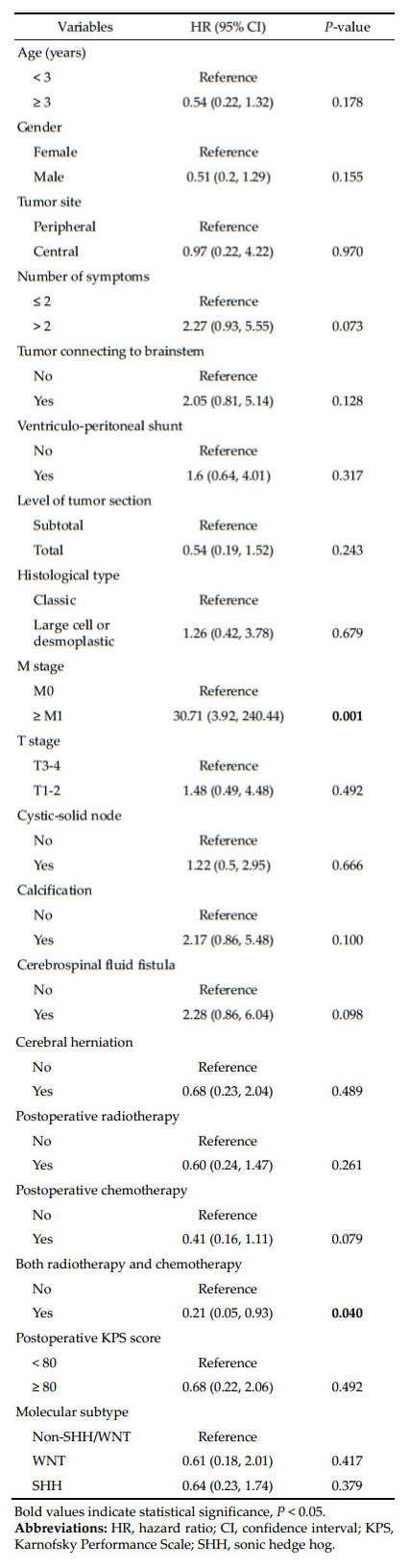
|
Because few studies have investigated factors associated with the prognosis of children with medulloblastoma[20], this retrospective analysis was undertaken to identify clinical characteristics, including molecular subtype, and treatment outcomes of pediatric patients with medulloblastoma in China. M stage, calcification, cerebrospinal fluid fistula, postoperative treatment (radiotherapy, chemotherapy, and both), postoperative KPS score, and molecular subtype were all associated with the OS of patients with medulloblastoma. Factors associated with disease progression included number of symptoms, M stage, and postoperative radiotherapy; M stage and postoperative radiotherapy and chemotherapy were associated with recurrence. Taken together, molecular subtyping of medulloblastoma was more predictive of survival than histopathology in patients undergoing adjuvant therapy.
In the present study, no differences in patient outcomes were detected between the pathological types. Because the prognosis of patients with the same pathological type of medulloblastoma can be drastically different due to varying genetic backgrounds[2], the development of new methods of molecular subtyping of medulloblastoma is necessary. In the present study, molecular subtyping analysis revealed that almost half of the children had the non-SHH/WNT subtype. Furthermore, the prognosis of patients with the WNT subtype was the best followed by the SHH subtype of medulloblastoma. These findings were consistent with those from another study on medulloblastomas in China[10].
Our results further confirmed the prognostic superiority of determining molecular subtypes over pathological types. However, the molecular subtypes as determined by YAP1 and GAB1 were not associated with disease recurrence or progression. Therefore, further studies are required to identify additional markers, including glutamate, a predictive marker for survival of pediatric patients with medulloblastoma[11], for further molecular subtyping of medulloblastoma in children. In addition, a consensus on the optimal method for identifying medulloblastoma subtypes (e.g., immunohistochemistry, CTNNB1 mutation analysis, or quantitative PCR)[12] needs to be reached through additional studies.
In the present study, disease progression was associated with the presence of >2 symptoms, which might be related to the location of medulloblastoma in children. Medulloblastoma is usually present in the midline of the posterior fossa and may cause disordered cerebrospinal fluid circulation, resulting in cerebellar dysfunction characterized by intracranial hypertension and cerebellar tissue destruction[13]. The clinical symptoms mainly include headache, vomiting, ataxia, nystagmus, cranial nerve palsy, increase in head circumference, cerebral hernia, and secondary epilepsy. Nervous system injury caused by the cancer or cerebral hernia due to intracranial hypertension can be life-threatening. Previous studies have confirmed that the time interval between disease onset and surgery may directly affect the prognosis of medulloblastoma, which may be due to greater symptom severity.
The staging for medulloblastoma is mainly based on the Chang staging system, which is based on the pre-operative findings on imaging and intra-operative findings to determine M stage and T stage. M stage is better for assessing the prognosis of medulloblastoma in children as compared to T stage[1, 12], which is consistent with the findings of the present study in which OS as well as disease progression and recurrence were significantly associated with M stage (M0 vs. ≥M1). However, no such associations were observed with T stage (T1-2 vs. T3-4).
Post-operative KPS score has also been used in the determination of post-operative prognosis with a score of ≥80 indicative of the capability of independent living. In the present study, a post-operative KPS score of ≥80 was associated with significantly longer OS. In addition to KPS score, OS was also associated with tumor calcifications that could be visualized on imaging examinations.
Post-operative radiotherapy and chemotherapy are important to improve the prognosis of patients with medulloblastoma[2, 10], which is consistent with the present study. Given the toxicity of radiotherapy and chemotherapy to the nervous system in children[14], in-depth studies examining individualized therapy according to the risk stratification of patients with medulloblastoma are necessary. For example, the dose of radiation or chemotherapeutics may be reduced in children with a low risk for recurrence, which may minimize the associated toxicity without compromising its therapeutic effectiveness.
An age of < 3 years has been identified as a factor associated with a poor prognosis in patients with medulloblastoma[15]. However, no such association was observed in the present study. In addition, previous studies have shown that the extent of resection was a key factor affecting the prognosis of patients with medulloblastoma[16, 17]. Pogorzala et al.[18] reported that incomplete surgical resection was associated with poor outcomes. However, in our study, OS time was similar in children treated with total resection compared to those treated with subtotal resection. Furthermore, OS time was comparable between patients with and without tumors that were adherent to the brainstem. Although these differences may be due to the small sample size in the present study, we speculate that total resection should not be performed if it is difficult to completely remove the cancer, especially given the evidence showing that the residual cancer cells will be cleared by post-operative radiotherapy[19].
This study is limited by its small sample size and that the results are from a single institution. Thus, the results need to be confirmed with larger sample sizes. In addition, the precise types of chemotherapy and radiotherapy and their influence on patient prognosis were not determined. Finally, limitations regarding the immunohistochemistry method utilized for subtype classification did not permit separating groups 3 and 4 molecular subtypes of medulloblastoma, which can be identified via mRNA analysis[20]. Therefore, differences between these subtypes were not identified.
5 ConclusionsMolecular subtypes are better determinants of the prognosis of patients with medulloblastoma as compared to pathological types, and therefore, they may be used to guide the therapy of medulloblastoma. Further studies with a larger sample size are necessary to validate our results and to identify and improve the novel markers of different medulloblastoma subtypes to make this approach more reliable.
Conflict of interests
All contributing authors have no conflict of interests.
| [1] | Bartlett F, Kortmann R, Saran F. Medulloblastoma. Clin Oncol, 2013, 25(1): 36–45. DOI:10.1016/j.clon.2012.09.008 |
| [2] | Gerber NU, Mynarek M, von Hoff K, Friedrich C, Resch A, Rutkowski S. Recent developments and current concepts in medulloblastoma. Cancer Treat Rev, 2014, 40(3): 356–365. DOI:10.1016/j.ctrv.2013.11.010 |
| [3] | Padovani L, Sunyach MP, Perol D, Mercier C, Alapetite C, Haie-Meder C, Hoffstetter S, Muracciole X, Kerr C, Wagner JP, et al. Common strategy for adult and pediatric medulloblastoma: A multicenter series of 253 adults. Int J Radiat Oncol Biol Phys, 2007, 68(2): 433–440. DOI:10.1016/j.ijrobp.2006.12.030 |
| [4] | Rieken S, Mohr A, Habermehl D, Welzel T, Lindel K, Witt O, Kulozik AE, Wick W, Debus J, Combs SE. Outcome and prognostic factors of radiation therapy for medulloblastoma. Int J Radiat Oncol Biol Phys, 2011, 81(3): e7–e13. DOI:10.1016/j.ijrobp.2010.12.042 |
| [5] | Pfister S, Remke M, Benner A, Mendrzyk F, Toedt G, Felsberg J, Wittmann A, Devens F, Gerber NU, Joos S, et al. Outcome prediction in pediatric medulloblastoma based on DNA copy-number aberrations of chromosomes 6q and 17q and the MYC and MYCN loci. J Clin Oncol, 2009, 27(10): 1627–1636. DOI:10.1200/JCO.2008.17.9432 |
| [6] | Pfister SM, Korshunov A, Kool M, Hasselblatt M, Eberhart C, Taylor MD. Molecular diagnostics of CNS embryonal tumors. Acta Neuropathol, 2010, 120(5): 553–566. DOI:10.1007/s00401-010-0751-5 |
| [7] | Ellison DW, Dalton J, Kocak M, Nicholson SL, Fraga C, Neale G, Kenney AM, Brat DJ, Perry A, Yong WH, et al. Medulloblastoma: Clinicopathological correlates of SHH, WNT, and non-SHH/WNT molecular subgroups. Acta Neuropathol, 2011, 121(3): 381–396. DOI:10.1007/s00401-011-0800-8 |
| [8] | Kool M, Korshunov A, Remke M, Jones DTW, Schlanstein M, Northcott PA, Cho YJ, Koster J, Schouten-van Meeteren A, van Vuurden D, et al. Molecular subgroups of medulloblastoma: An international meta-analysis of transcriptome, genetic aberrations, and clinical data of WNT, SHH, Group 3, and Group 4 medulloblastomas. Acta Neuropathol, 2012, 123(4): 473–484. DOI:10.1007/s00401-012-0958-8 |
| [9] | Mamanova L, Coffey AJ, Scott CE, Kozarewa I, Turner EH, Kumar A, Howard E, Shendure J, Turner DJ. Targetenrichment strategies for next-generation sequencing. Nat Methods, 2010, 7(2): 111–118. DOI:10.1038/nmeth.1419 |
| [10] | Zhang ZY, Xu J, Ren Y, Yao Y, Li KKW, Ng HK, Mao Y, Zhou LF, Zhong P. Medulloblastoma in China: Clinicopathologic analyses of SHH, WNT, and non-SHH/WNT molecular subgroups reveal different therapeutic responses to adjuvant chemotherapy. PLoS One, 2014, 9(6): e99490. DOI:10.1371/journal.pone.0099490 |
| [11] | Wilson M, Gill SK, MacPherson L, English M, Arvanitis TN, Peet AC. Noninvasive detection of glutamate predicts survival in pediatric medulloblastoma. Clin Cancer Res, 2014, 20(17): 4532–4539. DOI:10.1158/1078-0432.CCR-13-2320 |
| [12] | Pietsch T, Schmidt R, Remke M, Korshunov A, Hovestadt V, Jones DTW, Felsberg J, Kaulich K, Goschzik T, Kool M, et al. Prognostic significance of clinical, histopathological, and molecular characteristics of medulloblastomas in the prospective HIT2000 multicenter clinical trial cohort. Acta Neuropathol, 2014, 128(1): 137–149. DOI:10.1007/s00401-014-1276-0 |
| [13] | Northcott PA, Korshunov A, Witt H, Hielscher T, Eberhart CG, Mack S, Bouffet E, Clifford SC, Hawkins CE, French P, et al. Medulloblastoma comprises four distinct molecular variants. J Clin Oncol, 2011, 29(11): 1408–1414. DOI:10.1200/JCO.2009.27.4324 |
| [14] | Kiltie AE, Lashford LS, Gattamaneni HR. Survival and late effects in medulloblastoma patients treated with craniospinal irradiation under three years old. Med Pediatr Oncol, 1997, 28(5): 348–354. DOI:10.1002/(ISSN)1096-911X |
| [15] | Rutkowski S, von Hoff K, Emser A, Zwiener I, Pietsch T, Figarella-Branger D, Giangaspero F, Ellison DW, Garre ML, Biassoni V, et al. Survival and prognostic factors of early childhood medulloblastoma: An international meta-analysis. J Clin Oncol, 2010, 28(33): 4961–4968. DOI:10.1200/JCO.2010.30.2299 |
| [16] | Khafaga Y, Kandil AE, Jamshed A, Hassounah M, DeVol E, Gray AJ. Treatment results for 149 medulloblastoma patients from one institution. Int J Radiat Oncol Biol Phys, 1996, 35(3): 501–506. DOI:10.1016/S0360-3016(96)80012-5 |
| [17] | Zeltzer PM, Boyett JM, Finlay JL, Albright AL, Rorke LB, Milstein JM, Allen JC, Stevens KR, Stanley P, Li H, et al. Metastasis stage, adjuvant treatment, and residual tumor are prognostic factors for medulloblastoma in children: Conclusions from the Children's Cancer Group 921 randomized phase Ⅲ study. J Clin Oncol, 1999, 17(3): 832–845. DOI:10.1200/JCO.1999.17.3.832 |
| [18] | Pogorzala M, Styczynski J, Wysocki M. Survival and prognostic factors in children with brain tumors: Long-term follow-up single center study in Poland. Anticancer Res, 2014, 34(1): 323–326. |
| [19] | Modha A, Vassilyadi M, George A, Kuehn S, Hsu E, Ventureyra ECG. Medulloblastoma in children—The Ottawa experience. Childs Nerv Syst, 2000, 16(6): 341–350. DOI:10.1007/s003810050529 |
| [20] | Kaur K, Kakkar A, Kumar A, Mallick S, Julka PK, Gupta D, Suri A, Suri V, Sharma MC, Sarkar C. Integrating molecular subclassification of medulloblastomas into routine clinical practice: A simplified approach. Brain Pathol, 2016, 26(3): 334–343. DOI:10.1111/bpa.2016.26.issue-3 |