2. Department of Anaesthesia, Zhongda Hospital, Southeast University, Nanjing 210009, China
Lukui Chen (MD PhD) holds the Professor and Chairman in Department of Neurosurgery at Zhongda Hospital Southeast University (Nanjing, China) and is also the Director of the Minimally Invasive Neurosurgery Center and Functional Neurosurgery Institute at Southeast University. His research interests include spine, brain tumor targeted therapy, stem cell transplantation therapy and nanoneurosurgery. He has edited numerous books and chapters and published over 50 papers in the field of neurosurgery and neuroscience.
Xiaoyuan Guo, MD (2005) from Medical School of Southeast University, Attending surgeon at Department of Neurosurgery, Zhongda Hospital. Research interest in spine, epilepsy and cerebrovascular diseases.
Hong Wang, MD (2012) from Medical School of Southeast University, Resident at Department of Neurosurgery, Zhongda hospital. Research interest in spine, emergency neurosurgery and brain tumor.
Guojian wu, MD (2011) from Medical School of Nanjing University, Resident at Department of Neurosurgery, Zhongda Hospital. Research interest in spine, emergency neurosurgery and functional neurosurgery.
Jun Kong, MD (2016) from Medical School of Southeast University, Resident at Department of Neurosurgery, Zhongda Hospital. Research interest in spine, functional neurosurgery and intraoperative neurophysiological monitoring.
Ning Yin (MD PhD) holds the Professor in Department of Anaesthesiology at Zhongda Hospital Southeast University (Nanjing, China). Her research interests include neuro-anaesthesia and intraoperative neurophysiological monitoring. She has edited four books or chapters and published about 50 papers in the field of anesthesiology.
Most intramedullary spinal tumors are benign, comprising approximately 2%-4% of all central nervous system neoplasms[1]. Ependymoma is the most common tumor type in adults, accounting for 40%-60% of all intramedullary spinal tumors, with a mean age of 35-40 years at presentation. Astrocytoma is the most common type in children, accounting for approximately 60% of all intramedullary spinal tumors in this population, with a mean age at presentation of 5-10 years. Most spinal astrocytomas are low grade (WHO grade Ⅱ) and less aggressive than astrocytomas in the brain, but those malignant also exist. Pain and neurologic defects associated with these tumors are due to invasion of spinal cord parenchyma and nerve fiber stretching.
Intramedullary tumors can arise from any segment of the spinal cord, from the cervicomedullary junction to the filum terminale. The cervical cord is the most common location, probably due to the abundance of neural tissue in this segment as compared to the thoracic or lumbar segments. Irrespective of the histology, surgical resection of tumors in the cervical cord is very risky and complicated. Consequently, surgical removal is only recommended when the tumor borders are clearly demarcated[2-4].
Complete surgical resection without damage to functional tissue is usually not possible because of the infiltrative nature of these tumors. On the other hand, increased overall survival is reported in patients with spinal cord astrocytomas who underwent grosstotal resection (GTR)[5]. Several recent reports have also shown the impact of maximal resection on overall survival and progression-free survival in patients with spinal cord tumors. If GTR is considered safe after evaluating all relevant factors, complete surgical resection is the primary treatment goal[6, 7] in patients with malignant spinal cord tumors (intramedullary)[8, 9].
2 Cases descriptionCase 1
A 46-year-old male presented with a 4-year history of upper and lower limb numbness and weakness (motor strength 4/5 and 2/5 in upper and lower limbs, respectively). Symptoms worsened 30 days before consulting our facility. Magnetic resonance imaging (MRI) of the spinal cord showed an intramedullary mass at C5-C6. Surgical resection under awake anesthesia was recommended; the patient received detailed information regarding the procedure and possible risks, and informed consent was obtained. He remained intubated and his motor speech function was not monitored, because the language center was not involved in surgery. The patient followed commands and performed all upper and lower limb movements as instructed. Sensory functions were not fully assessed; instead, minimal monitoring was performed with sensory stimulation of his hands and feet, with instruction to raise his thumb if he perceived sensation. GTR of the tumor was performed under awake (AAA) anesthesia with neurophysiological monitoring (NPM). Intraoperative frozen section confirmed the diagnosis of diffuse spinal cord astrocytoma. Symptoms completely resolved postoperatively, and no motor deficits were observed; however, there was minimal numbness in the finger tips.
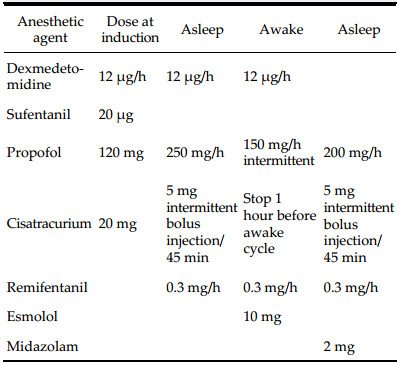
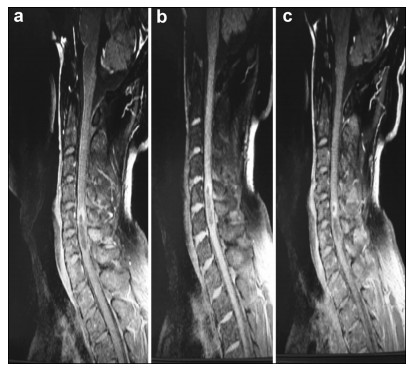
Preoperative and postoperative MR images of Case 1 are shown in Figure 1 and Figure 2, respectively.
|
| Figure 1 a–c. Preoperative MR images of Case 1. |
|
| Figure 2 a–c. Postoperative MR images of Case 1 |
Case 2
A 35-year-old male presented with the chief complaint of pain in the neck and back for 6 months. There was no weakness or numbness in the upper and lower limbs. MRI of the spine revealed an intramedullary mass at C5-T1. We advised resection. Because of cervical cord involvement, he underwent GTR under awake anesthesia (AAA), after providing informed consent. Intraoperatively, he was instructed to move his upper and lower limbs during the awake cycle of AAA. He was also not extubated during the awake cycle. All commands were followed. As the language center was not in the surgical field, no speech assessment was required. Sensory functions were also minimally assessed by lightly touching the upper and lower limbs and then asking the patient to raise his thumb if he perceived sensation. Frozen section confirmed the diagnosis of ependymoma, and GTR was performed successfully. His symptoms were completely relieved, and no motor deficits were observed at 3 and 6 months of follow up.
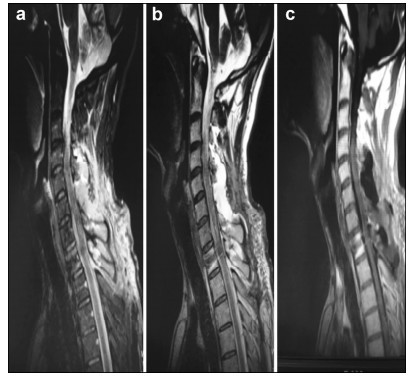
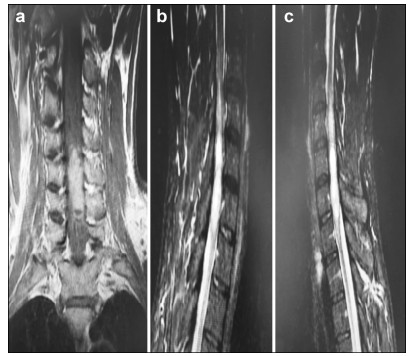
Preoperative and postoperative MR images of Case 2 are shown in Figure 3 and Figure 4, respectively.
|
| Figure 3 a–c. Preoperative MR images of Case 2. |
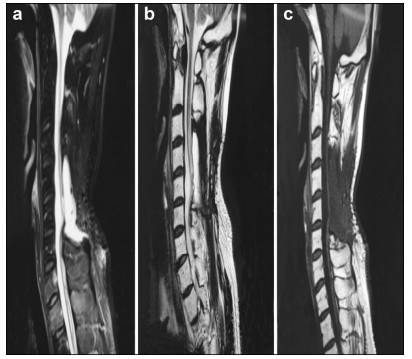
|
| Figure 4 a–c. Postoperative MR images of Case 2. |
Awake resection of spinal cord tumors can be carried out safely using asleep-awake-asleep (AAA) anesthesia technique; however, the surgeon, anesthesiologist, and neurophysiologist must have sufficient training and experience. Care must be taken in choosing anesthetic agents, and patient selection should be made after careful history taking and examination to rule out any contraindication to awake anesthesia. The anesthetics and doses used in our procedures are shown in Table 1.
Awake craniotomies for resection of tumors are often performed to protect the brain's eloquent areas, particularly those responsible for language and motor skills, from intraoperative damage[10-12]. The direct and rapid feedback obtained by talking to the patient and observing both motor and language functions provides more effective monitoring than that obtained using only evoked potentials. Intraoperative brain mapping using bipolar stimulation further enhances feedback, and allows surgeons to definitively identify the eloquent regions[13]. When a language or motor deficit occurs during awake craniotomy, lesion resection can be modified immediately. Additionally, awake craniotomies can potentially reduce resource utilization, and can result in shorter intensive care unit and hospital stays.[14]
Traditionally, intramedullary spinal tumor resection is performed under general anesthesia with NPM; however, intraoperative NPM sometimes does not provide accurate information, and the responses are sometimes delayed[4]. Generally, surgical resection of these tumors is very challenging, even with intensive intraoperative NPM, because of a significant risk of transient or permanent neurological deterioration[15-19]. Studies have reported worsening of preexisting neurological impairment or new neurological deficits, or both, in up to 64% of cases, after surgical resection of intrinsic spinal cord tumors[15, 20-23].
Moreover, aggressive resection of a tumor requires histological diagnosis of a frozen section and the ability to find and maintain a surgical plane. Patients may have cysts or syringomyelia at both ends of an intramedullary spinal tumor, which can provide a clear plane for safe resection. Because of the difficulty in distinguishing many ependymomas from astrocytomas on frozen section, the presence or absence of a clear surgical plane is the key determining factor in defining the surgical goal. After analysis of all available data including intraoperative appearance, frozen section, and imaging features, if the likely diagnosis is ependymoma, complete surgical resection should be performed, if possible. If a diagnosis of astrocytoma is likely, most experts favor limited debulking of only the tissue that is clearly abnormal. Partial or subtotal resection can be performed for soft tumors, while GTR is suggested for solid tumors with clear borders[20, 24].
Safe GTR of an intramedullary tumor without causing neurological deficits is the goal of surgery. GTR will not only relieve the symptoms, but will also reduce the likelihood of recurrence. Surgery on the spinal cord can cause serious postoperative complications. Permanent neurological deficits can occur, even if the spinal cord is mildly compressed or stretched. Other than direct surgical injuries, the complications may include ischemia and edema of the cord. Careful selection of potential candidates for this procedure is vital. Contraindications to awake craniotomy include inability to cooperate, dysphasia, language barriers, emotional lability, cognitive impairment, age < 11 years old, low occipital tumors, and tumors with significant dural attachment. The same criteria can be applied to awake procedures on the spinal cord.
To reduce intraoperative iatrogenic injuries, combined sensory evoked potential (SEP) and motor evoked potential (MEP) monitoring is a safe, sensitive, and reliable method for detection of injury to the spinal cord. Sensory and motor pathways can be independently assessed during surgery, the number of false negatives is reduced to zero, and there is probably a positive influence on the final postoperative outcome. Routine combined use of SEPs and MEPs is advisable during complex spine/spinal cord surgery, and the combined use of epidural and muscle MEPs is probably the best way to assess motor pathways during spinal cord surgery.
Identification of an abnormality on SEP and MEP is critical during surgery involving the spinal cord. A 50% drop in cortical SEP amplitude, regardless of whether it is associated with an increase in latency, is a universally accepted warning sign. Conversely, different warning signs for MEPs have been proposed, ranging from changes in the thresholds that elicit muscle MEPs to the pure presence or absence of responses, amplitude variation, or a combination of changes in threshold and amplitude variation.
Despite the advantages of intraoperative NPM, there are also limitations that can interfere with intraoperative identification of neurological deficits. Although there is no evidence to support the failure of MEPs to detect deficits, delayed responses have been reported, and the accuracy of responses has also been questioned[4]. Furthermore, one study[25] reported that transcranial MEPs recorded from the lower extremities can fail to provide rapid detection of spinal cord ischemia at the upper thoracic level during surgery on the descending thoracic aorta. Spinal cord and aortic surgeries are quite different, but there is strong evidence supporting the possibility of delayed responses with NPM. These delayed and less accurate responses will not alert the surgeon to injury of important neurological structures. Accordingly, we performed resection of intramedullary cervical spinal tumors under awake anesthesia (AAA), in order to obtain more detailed and accurate intraoperative information about general and motor neurological function. Awake craniotomy performed while approaching lesions in the eloquent areas of the brain has resulted in reduced postoperative neurological functional deficits. Similarly, we believe that procedures involving the spinal cord-particularly the resection of intramedullary spinal cord tumors-can also successfully be performed after careful selection of patients. Thus, we suggest that the best strategy for resection of an intramedullary spinal cord tumor will be to combine NPM (MEP and SEP monitoring) with awake anesthesia, to make spinal cord tumor resection even safer, with decreased postoperative motor deficits and reduced morbidity.
Conflict of interests
No contributing authors have a conflict of interests.
| [1] | Klekamp J, Samii M. Surgery of Spinal Tumors. Berlin Heidelberg: Springer, 2007 . |
| [2] | McCormick PC, Stein BM. Intramedullary tumors in adults. Neurosurg Clin N Am, 1990, 1(3): 609–630. |
| [3] | Wang ZY, Sun JJ, Xie JC, Li ZD, Ma CC, Liu B, Chen XD, Liao HI, Yu T, Zhang J. Comparative analysis on the diagnosis and treatments of multisegment intramedullary spinal cord tumors between the different age groups. Neurosurg Rev, 2012, 35(1): 85–93. DOI:10.1007/s10143-011-0345-2 |
| [4] | Kim SH, Bak KH, Kim DW, Kang TH. Primary intramedullary spinal sarcoma: A case report and review of the current literatures. J Korean Neurosurg Soc, 2010, 48(5): 448–451. DOI:10.3340/jkns.2010.48.5.448 |
| [5] | McGirt MJ, Goldstein IM, Chaichana KL, Tobias ME, Kothbauer KF, Jallo GI. Extent of surgical resection of malignant astrocytomas of the spinal cord: Outcome analysis of 35 patients. Neurosurgery, 2008, 63(1): 55–61. DOI:10.1227/01.NEU.0000335070.37943.09 |
| [6] | Beneš Ⅷ, Barsa P, Beneš VJr, Suchomel P. Prognostic factors in intramedullary astrocytomas: A literature review. Eur Spine J, 2009, 18(10): 1397–1422. DOI:10.1007/s00586-009-1076-8 |
| [7] | Garcés-Ambrossi GL, McGirt MJ, Mehta VA, Sciubba DM, Witham TF, Bydon A, Wolinksy JP, Jallo GI, Gokaslan ZL. Factors associated with progression-free survival and longterm neurological outcome after resection of intramedullary spinal cord tumors: Analysis of 101 consecutive cases. J Neurosurg Spine, 2009, 11(5): 591–599. DOI:10.3171/2009.4.SPINE08159 |
| [8] | Chi JH, Parsa AT. Intramedullary spinal cord metastasis: Clinical management and surgical considerations. Neurosurg Clin N Am, 2006, 17(1): 45–50. DOI:10.1016/j.nec.2005.10.003 |
| [9] | Abdel-Wahab M, Etuk B, Palermo J, Shirato H, Kresl J, Yapicier O, Walker G, Scheithauer BW, Shaw E, Lee C, et al. Spinal cord gliomas: A multi-institutional retrospective analysis. Int J Radiat Oncol Biol Phys, 2006, 64(4): 1060–1071. DOI:10.1016/j.ijrobp.2005.09.038 |
| [10] | Blanshard HJ, Chung F, Manninen PH, Taylor MD, Bernstein M. Awake craniotomy for removal of intracranial tumor: Considerations for early discharge. Anesth Analg, 2001, 92(1): 89–94. |
| [11] | Pereira LCM, Oliveira KM, L'Abbate GL, Sugai R, Ferreira JA, da Motta LA. Outcome of fully awake craniotomy for lesions near the eloquent cortex: Analysis of a prospective surgical series of 79 supratentorial primary brain tumors with long follow-up. Acta Neurochir (Wien), 2009, 151(10): 1215–1230. DOI:10.1007/s00701-009-0363-9 |
| [12] | De Benedictis A, Moritz-Gasser S, Duffau H. Awake mapping optimizes the extent of resection for low-grade gliomas in eloquent areas. Neurosurgery, 2010, 66(6): 1074–1084. DOI:10.1227/01.NEU.0000369514.74284.78 |
| [13] | Chacko AG, Thomas SG, Babu KS, Daniel RT, Chacko G, Prabhu K, Cherian V, Korula G. Awake craniotomy and electrophysiological mapping for eloquent area tumours. Clin Neurol Neurosurg, 2013, 115(3): 329–334. DOI:10.1016/j.clineuro.2012.10.022 |
| [14] | de Amorim RLO, de Almeida AN, de Aguiar PHP, Fonoff ET, Itshak S, Fuentes D, Teixeira MJ. Cortical stimulation of language fields under local anesthesia: Optimizing removal of brain lesions adjacent to speech areas. Arq Neuropsiquiatr, 2008, 66(3A): 534–538. DOI:10.1590/S0004-282X2008000400018 |
| [15] | Hoshimaru M, Koyama T, Hashimoto N, Kikuchi H. Results of microsurgical treatment for intramedullary spinal cord ependymomas: Analysis of 36 cases. Neurosurgery, 1999, 44(2): 264–269. DOI:10.1097/00006123-199902000-00012 |
| [16] | Jallo GI, Freed D, Epstein F. Intramedullary spinal cord tumors in children. Childs Nerv Syst, 2003, 19(9): 641–649. DOI:10.1007/s00381-003-0820-3 |
| [17] | Jallo GI, Kothbauer KF, Epstein FJ. Intrinsic spinal cord tumor resection. Neurosurgery, 2001, 49(5): 1124–1128. |
| [18] | Morota N, Deletis V, Constantini S, Kofler M, Cohen H, Epstein FJ. The role of motor evoked potentials during surgery for intramedullary spinal cord tumors. Neurosurgery, 1997, 41(6): 1327–1336. DOI:10.1097/00006123-199712000-00017 |
| [19] | Matsuyama Y, Sakai Y, Katayama Y, Imagama S, Ito Z, Wakao N, Sato K, Kamiya M, Yukawa Y, Kanemura T, et al. Surgical results of intramedullary spinal cord tumor with spinal cord monitoring to guide extent of resection. J Neurosurg Spine, 2009, 10(5): 404–413. DOI:10.3171/2009.2.SPINE08698 |
| [20] | Cristante L, Herrmann HD. Surgical management of intramedullary spinal cord tumors: Functional outcome and sources of morbidity. Neurosurgery, 1994, 35(1): 69–76. DOI:10.1227/00006123-199407000-00011 |
| [21] | Sandalcioglu IE, Hunold A, Müller O, Bassiouni H, Stolke D, Asgari S. Spinal meningiomas: Critical review of 131 surgically treated patients. Eur Spine J, 2008, 17(8): 1035–1041. DOI:10.1007/s00586-008-0685-y |
| [22] | Schwartz TH, McCormick PC. Intramedullary ependymomas: Clinical presentation, surgical treatment strategies and prognosis. J Neuro-Oncol, 2000, 47(3): 211–218. DOI:10.1023/A:1006414405305 |
| [23] | Yang SY, Yang XY, Hong GL. Surgical treatment of one hundred seventy-four intramedullary spinal cord tumors. Spine, 2009, 34(24): 2705–2710. DOI:10.1097/BRS.0b013e3181b43484 |
| [24] | Boström A, Kanther NC, Grote A, Boström J. Management and outcome in adult intramedullary spinal cord tumours: A 20-year single institution experience. BMC Res Notes, 2014, 7: 908. DOI:10.1186/1756-0500-7-908 |