Education background:
September 2000–July 2005 Hebei Medical University bachelor degree in clinical medicine
September 2005–July 2010 Beijing College of Neurosurgery the standardized training of residents in neurosurgery
September 2008–July 2011 Capital Medical University master degree in clinical medicine.
Yuqi Zhang is a Ph.D. Tutor, a well-known expert in neurosurgery, and a leading pediatric neurosurgeon in the Chinese Community. He is the Vice President of Tsinghua University's Medical School and President of Tsinghua University's Second Affiliated Hospital. He studied neurosurgery in the United States as post-doctoral researcher, and was for twelve years the Director of Pediatric Neurosurgery at Beijing Tiantan Hospital. He also served as the Chinese Journal of Neurosurgery's Editorial Director for nine years. He is a council member of the Chinese Physicians' Association, the Vice Chairman of the Neurological Surgeons' Chapter of the Chinese Medical Association, a Chinese member of the International Society for Pediatric Neurosurgery (ISPN), and an editorial board member of several distinguished journals, including the Chinese Journal of Neurosurgery and the Journal of Pediatric Surgery.
Huancong Zuo graduated from Beijing Medical University in 1968, and received a Master's degree in Medicine from Beijing Union Medical College in 1981. He studied microneurosurgery in Japan, and in 1996 co-founded the Tsinghua University Sino-Japanese Friendship Institute of Medicine with the cooperation of the Tsinghua University Biology Department, where he served as Executive Director. In 2002, he moved to the Tsinghua University School of Medicine. He is President of Tsinghua University's Second Affiliated Hospital and Head of its Institute for Clinical Neuroscience. Professor Zuo was the first surgeon in China to perform microvascular decompression surgery for trigeminal neuralgia and hemifacial spasm.
Glioblastoma is the most common intracranial tumor, and the imaging findings of glioblastoma are variable. However, most glioblastoma lesions present with a ring-shaped pronounced mass effect and heterogeneous enhancement with centrally necrotic regions with no enhancement on magnetic resonance imaging (MRI). Typical glioblastoma is easily identified on MRI; however, some non-characteristic images may be misinterpreted as vascular disease. Here, we report a case of glioblastoma with imaging findings mimicking a cerebral cavernous malformation (CCM). The patient with intracranial hemorrhage (ICH) was ultimately diagnosed as having glioblastoma that contained significant tumor-related vasculature. We discuss the challenges in the diagnosis of glioblastoma and its probable relationship with CCM.
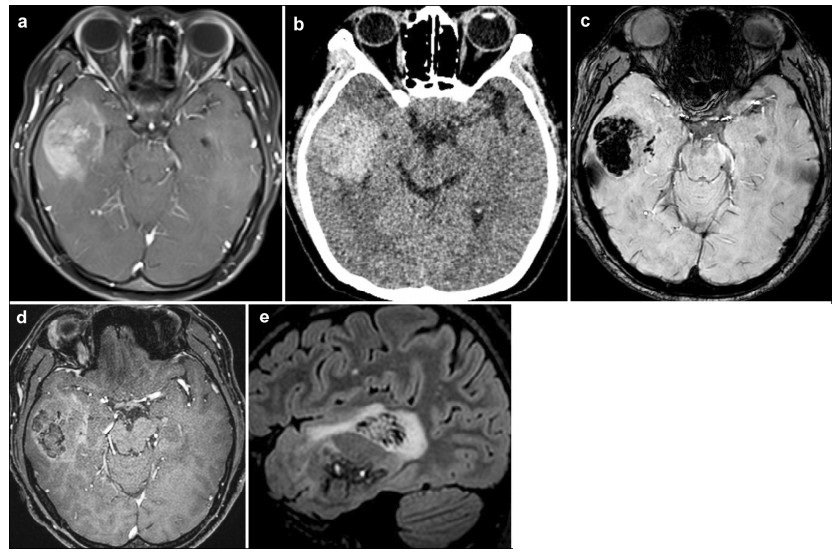
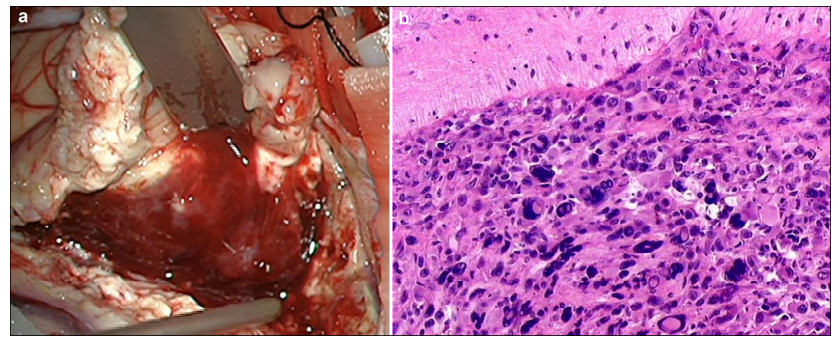
2 Case reportA 34-year-old woman presented with a 9-day history of intermittent dizziness at the Tsinghua University Yuquan Hospital (Beijing, China) in July 2015. No other nervous system deficits were found. Brain MRI (Figure 1a) revealed a 52 mm × 51 mm × 48 mm lesion on the right temporal lobe and showed non-obvious edema and slightly heterogeneous contrast enhancement without centrally necrotic regions. On the other hand, brain computed tomography (CT) (Figure 1b) revealed a hemorrhage on the right temporal lobe. High-resolution susceptibility weighted imaging (SWI) (Figure 1c) showed a hypointensity lesion. Simultaneous noncontrast angiography and intraplaque hemorrhage (SNAP) imaging (Figure 1d) showed that the lesion was rich in vasculature. T2-weighted inversion recovery based sequence for CSF suppression (T2 Flair) imaging also showed that the lesion was rich in small blood vessels (Figure 1e). All these characteristics did not support a diagnosis of glioblastoma. The patient was scheduled to undergo a right frontotemporal craniotomy. During the procedure, the lesion (Figure 2a) had distinct boundaries with reddish brown appearance and was rich in venous vessels. All these results were consistent with the diagnosis of CCM. The lesion was pathologically confirmed as glioblastoma according to the results of histological examination (Figure 2b). The histopathological analysis revealed that the tumor was composed of densely packed cells and showed nuclear and cell pleomorphism. Follow-up MRI performed at 3 months post-surgery showed no evidence of recurrence.
|
| Figure 1 a Brain magnetic resonance imaging (MRI) reveals a 52 mm × 51 mm × 48 mm-lesion on the right temporal lobe and shows non-obvious edema and slightly heterogeneous contrast enhancement without centrally necrotic regions. b Brain computed tomography (CT) reveals a hemorrhage on the right temporal lobe. c High-resolution susceptibility weighted imaging (SWI) shows a hypointensity lesion. d Simultaneous noncontrast angiography and intraplaque hemorrhage (SNAP) imaging shows that the lesion is rich in vasculature. e T2 weighted inversion recovery based sequence for CSF suppression (T2 Flair)-imaging shows that the lesion is rich in small blood vessels. |
|
| Figure 2 a The lesion, as observed during the surgical procedure, shows distinct boundaries with reddish brown appearance and is rich in venous vessels. b Histopathological analysis reveals that the tumor is composed of densely packed cells and shows nuclear and cell pleomorphism. The histopathological diagnosis is glioblastom. (Hematoxylin-eosin stain, ×100) |
Glioblastomas are highly malignant and invasive brain tumors. Glioblastoma is the most common intracranial tumor and the most frequent cause of hemorrhage in brain tumors. ICH caused by glioblastoma is usually located in the cerebral hemispheres, basal ganglia, and corpus callosum[1]. A few hypotheses have been proposed to explain the etiology of tumor-associated hemorrhage. Hemorrhage within the tumor may be caused by the disruption of vessels and tumor erosion of the cerebral vessels[2]. The vasculature of tumors and peritumoral tissue is structurally and functionally abnormal, and these vessels may bleed easily[3-5]. Suppression of the tissue factor-dependent coagulation cascade may cause glioblastoma-associated ICH[6]. Finally, intratumoral arteriovenous fistulae may also be a mechanism for the pathogenesis of tumor-associated hemorrhage[7]. In this case, the hemorrhage could have originated from the numerous small abnormal vessels, which rupture easily.
It is difficult to make a diagnosis when glioblastoma presents as ICH. Nevertheless, some distinctive characteristics of tumor-related ICH on images may aid in diagnosis. Atypical position, irregular shape, heterogeneous signal, and multiple hemorrhage positions suggest that intracranial hematomas may be caused by neoplasms[7, 8]. Most of tumor-related hemorrhages are intraparenchymal and rarely rupture into the subarachnoid space. In this respect, this case was consistent with a tumor-related hematoma. Glioblastoma may present with a ring-shaped enhancement around the tumor on contrast-enhanced CT or MRI with a hypodense area in the center. Peritumoral edema may be an important factor because it is scarce in acute ICH but very common in intracranial tumor. In the case presented here, the hemorrhage was located in the temporal lobe with some peritumoral edema. The appearance of spontaneous intracranial hematoma was homogenous, and there was no peri-lesional ring-like enhancement. All these features did not suggest a tumor presenting with hematoma. Therefore, it is important for us to consider neoplastic etiology when hemorrhage is located in an abnormal position or presents with untypical features.
The exact relationship between brain tumor and CCM is still unclear. One hypothesis is occasional association[9]. The tumor and CCM are common brain lesions and might coexist. We reported a woman with a right temporal lobe glioblastoma mimicking a CCM. This glioblastoma contained many small vessels with nonvisualization on angiography image. In the case presented, SWI and SNAP to investigate the etiology of ICH demonstrated a large region of irregular vasculature. The combination of ICH and abnormal vasculature on SNAP images was highly suggestive of CCM in this patient. These features resemble a CCM. This case together with other reports[10-14] might suggest an oncogenetic feature of glioblastoma. This indicates that some gliomas may develop from CCM.
Recently, many reports[10-17] have indicated that there may be a relationship between CCM and intracranial tumors such as glioma, meningioma, and schwannoma. There also have been some reports of glioblastoma presenting as suspected arteriovenous malformation[1, 10]. There may be two types of this relationship. The first type is a sequential relationship. In some cases, studies reported[9-15] that after radiation or resection of the CCM, glioma or anaplastic oligo-astrocytoma can occur. Due to this phenomenon, some studies indicated that glioma might originate from CCM. The other type is a coexisting relationship. Some studies reported that a brain tumor might coexist with a CCM[13]. This phenomenon also indicates that the tumor and CCM may have some common etiology. In the development of CCM, some genes such as CCM may be involved in the progression of CCM[18, 19]. These genes are also tumor suppressor genes. Mutation in these genes stimulates cell proliferation[18, 19]. These genes may play an important role in the development of gliomas, and they can be initiated by irradiation[15]. This might explain why some gliomas occur after radiotherapy. Under a second attack, especially after radiotherapy, CCM might develop into a tumor.
4 ConclusionsAlong with the reports of sequential or coexisting finding of vascular lesions and brain tumors, it is suggested that intracranial vascular disease may play a role in tumorigenesis. However, certain issues remain unsolved. It is still unclear as to which types of vascular diseases are more likely to transform into a tumor and which vascular disease might be a pretumor lesion. Therefore, when a lesion presents with hemorrhage, and the imaging features are very similar to a vascular disease, a tumor must be considered as a condition in the differential diagnosis.
Conflict of interests
The authors have no financial interest to disclose regarding the article.
| [1] | Khanna A, Venteicher AS, Walcott BP, Kahle KT, Mordes DA, William CM, Ghogawala Z, Ogilvy CS. Glioblastoma mimicking an arteriovenous malformation. Front Neurol, 2013, 4: 144. |
| [2] | Can SM, Aydin Y, Turkmenoglu O, Aydin F, Ziyal I. Giant cell glioblastoma manifesting as traumatic intracerebral hemorrhage—case report. Neurol Med Chir, 2002, 42(12): 568–571. DOI:10.2176/nmc.42.568 |
| [3] | Glass B, Abbott KH. Subarachnoid hemorrhage consequent to intracranial tumors: review of literature and report of seven cases. AMA Arch Neurol Psychiatry, 1955, 73(4): 369–379. DOI:10.1001/archneurpsyc.1955.02330100001001 |
| [4] | Liwnicz BH, Wu SZ, Tew JM. The relationship between the capillary structure and hemorrhage in gliomas. J Neurosurg, 1987, 66(4): 536–541. DOI:10.3171/jns.1987.66.4.0536 |
| [5] | Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science, 2005, 307(5706): 58–62. DOI:10.1126/science.1104819 |
| [6] | Takeshima H, Nishi T, Kuratsu J, Kamikubo Y, Kochi M, Ushio Y. Suppression of the tissue factor-dependent coagulation cascade: a contributing factor for the development of intratumoral hemorrhage in glioblastoma. Int J Mol Med, 2000, 6(3): 271–276. |
| [7] | Schrader B, Barth H, Lang EW, Buhl R, Hugo HH, Biederer J, Mehdorn HM. Spontaneous intracranial haematomas caused by neoplasms. Acta Neurochir (Wien), 2000, 142(9): 979–985. DOI:10.1007/s007010070052 |
| [8] | Maiuri F, D'Andrea F, Gallicchio B, Carandente M. Intracranial hemorrhages in metastatic brain tumors. J Neurosurg Sci, 1985, 29(1): 37–41. |
| [9] | Schreuder T, Te Lintelo M, Kubat B, Koehler P. Anaplastic oligo-astrocytoma occurring after resection of a cerebral cavernous malformation; malignant transformation? Case report and review on etiology. J Neurol, 2010, 257(3): 349–353. DOI:10.1007/s00415-009-5322-0 |
| [10] | Cemil B, Tun K, Polat O, Ozen O, Kaptanoglu E. Glioblastoma multiforme mimicking arteriovenous malformation. Turk Neurosurg, 2009, 19(4): 433–436. |
| [11] | Jadik S, Stan AC, Dietrich U, Pietilä TA, Elsharkawy AE. Intraparenchymal meningioma mimicking cavernous malformation: a case report and review of the literature. J Med Case Rep, 2014, 8: 467. DOI:10.1186/1752-1947-8-467 |
| [12] | Tumialán LM, Brat DJ, Fountain AJ, Barrow DL. An astroblastoma mimicking a cavernous malformation: Case report. Neurosurgery, 2007, 60(3): E569–E570. DOI:10.1227/01.NEU.0000255336.80285.70 |
| [13] | Feiz-Erfan I, Zabramski JM, Herrmann LL, Coons SW. Cavernous malformation within a schwannoma: review of the literature and hypothesis of a common genetic etiology. Acta Neurochir (Wien), 2006, 148(6): 647–652. DOI:10.1007/s00701-005-0716-y |
| [14] | Furuse M, Miyatake SI, Kuroiwa T. Cavernous malformation after radiation therapy for astrocytoma in adult patients: report of 2 cases. Acta Neurochir (Wien), 2005, 147(10): 1097–1101. DOI:10.1007/s00701-005-0579-2 |
| [15] | Zhang JY, Ming ZY, Wu AH. Is cerebral cavernous malformation a pre-glioma lesion. Chin Med J, 2012, 125(24): 4511–4513. |
| [16] | Jin SC, Ahn JS, Kwun BD, Kwon DH. Intraventricular cavernous malformation radiologically mimicking meningioma. J Korean Neurosurg Soc, 2008, 44(5): 345–347. DOI:10.3340/jkns.2008.44.5.345 |
| [17] | Cruz AS, Jeyamohan S, Moisi M, Tubbs RS, Page J, Chamiraju P, Tkachenko L, Rostad S, Newell DW. Duralbased cavernoma of the posterior cranial fossa mimicking a meningioma: A case report. Cureus, 2016, 8(4): e560. |
| [18] | Yang IY, Yum MS, Kim EH, Choi HW, Yoo HW, Ko TS. Two cases of familial cerebral cavernous malformation caused by mutations in the CCM1 gene. Korean J Pediatr, 2016, 59(6): 280–284. DOI:10.3345/kjp.2016.59.6.280 |
| [19] | Hanjani SA. The genetics of cerebrovascular malformations. J Stroke Cerebrovasc Dis, 2002, 11(5): 279–287. DOI:10.1053/jscd.2002.129617 |



