In recent years, both the incidence and morbidity of ischemic cerebrovascular disease have been rising sharply. Intracranial arterial stenosis is not only an important cause of ischemic stroke but also a significant factor for acute stroke recurrence[1]. Therefore, the study of risk factors for intracranial arterial stenosis is of clinical significance for the prevention of ischemic cerebrovascular disease[2]. To measure intracranial blood flow velocity, transcranial doppler ultrosonography (TCD) has been used in clinical studies since it is convenient, flexible, non-invasive, and low-cost[3]. In this study, 486 cases of symptomatic intracranial arterial stenosis were examined using color doppler flow imaging (CDFI) combined TCD technology to analyze the detection rate and distribution of intracranial arterial stenosis, and the correlation between intracranial arterial stenosis and its risk factors.
2 Materials and methods 2.1 SubjectsA total of 486 patients who underwent carotid ultrasound and TCD examination at the neurology department at our hospital from June 2012 to June 2014 were included in this study. Of them, 235 (48.4%) were male patients while 251 (51.6%) were female patients. The mean age was 63 ± 10 (mean ± standard deviation (SD)) years. A total of 317 (65.2%) patients had hypertension, 156 (32.1%) had diabetes, 64 (13.2%) had coronary heart disease, 232 (47.7%) had hyperlipidemia, and 170 (35.0%) were smokers. The following inclusion criteria were used: ① With TIA or ischemic stroke and aged 40 years or more. ② Basic information was complete, and target risk factors were explicit. ③ At least one target artery had stenosis detected using TCD. The following exclusion criteria were used: ① With the poor sound transmission of temporal window and patients were uncooperative. ② With severe stenosis or occlusion of the VA or BA, and with extracranial carotid artery (except patients whose blood flow was restored after the carotid artery stenting or carotid endarterectomy). ③ With cardiac embolism, arterial dissection, arterial inflammation, hypercoagulable state, moyamoya disease, malignant tumor, or other acute and chronic causes of inflammation. ④ Restenosis patients after intracranial or extracranial artery stenting. ⑤ Any other circumstances that researchers considered unsuitable for participation in this study.
2.2 CDFI examinationA Dutch Phillips IU22 bi-functional doppler ultrasonic diagnostic apparatus with a 3.0–9.0 MHz linear probe and a 2.0–5.0 MHz convex array probe was used to observe the carotid artery lumen diameter, plaques, and hemodynamic indicators.
2.3 TCD examinationA TCD ultrasound diagnostic apparatus TC8080 type (Germany) with a 1.6 MHz pulse wave probe was used to detect and record the peak systolic velocity (PSV), end diastolic velocity (EDV), vascular resistance index (RI), and pulsatility index (PI) of the target artery. All patients were evaluated by highly qualified doctors with over 5 years of experience. The detection methods and ultrasound diagnostic criteria of vascular stenosis were based on the literature[4]. A total of 434 patients with stenosis of less than 70% of the innominate artery, extracranial carotid artery, vertebral artery, or subclavian artery were included in group A. In group B, 52 patients with pre-existing 70%–99% stenosis of the innominate artery, extracranial carotid artery, vertebral artery or subclavian artery, whose blood flow had been restored within one month after the artery stenting or endarterectomy, were included. According to the stenosis degrees of the target artery (MCA, ICA, VA, and BA), group A and group B were further divided into the healthy control group A1, group B1 with mild stenosis, group A2 with moderately severe stenosis, and the occlusion group B2. Each target artery was given a different score based on the degree of stenosis: 0 for normal, 1 for mild stenosis, 2 for moderate stenosis, 3 for severe stenosis, and 4 for occlusion. The scores of the 11 target arteries were calculated; the arteries exceeding score 2 were assigned to group A2 or group B2, and the rest were assigned to group A1 or group B1.
2.4 Statistical AnalysisThe SPSS 17.0 software package was used for statistical analysis. The measurement data was expressed as x(-)±s, and t-test and chi-square test were used to evaluate statistically significant differences. Logistic regression analysis was applied to the risk factors associated with moderately severe stenosis, to calculate the relative risk (RR) and 95% confidence interval (CI) values. A P value of < 0.05 was considered statistically significant.
3 Results 3.1 Detection rate and distribution of intracranial arterial stenosisThe detection rate of intracranial arterial stenosis in 486 cases was 32.3% (157/486 cases). There were 37 patients aged 40 years or more with stenosis of the MCA, ICA, BA, or VA, and their detection rates were 62.2% (23/37 cases), 32.4% (12/37 cases), 29.7% (11/37 cases), and 16.2% (6/37 cases), respectively. The detection rate of MCA stenosis was the highest, and the difference was statistically significant (X2 = 904.074, P = 0.000).
Of the 486 cases, there were 115 cases (23.7%) of only MCA stenosis, 22 cases (4.5%) of only ICA stenosis, 20 cases (4.1%) of only anterior cerebral artery (ACA) stenosis, 23 cases (4.7%) of only posterior cerebral artery (PCA) stenosis, 42 cases (8.6%) of only BA stenosis, 27 cases (5.6%) of only VA stenosis, and the remaining 237 cases (48.8%) were of multi-branch arterial stenosis.
3.2 Analysis of risk factorsThe average age of patients in group B was 67.34 ± 7.13 years, higher than 62.32 ± 10.29 years of those in group A, and the difference was statistically significant (P = 0.000). Men accounted for 63.7% in group B, significantly higher than 46.5% in group A (P = 0.037). However, there was no statistically significant difference between the two groups in the risk factors of hypertension, diabetes, coronary heart disease, hyperlipidemia, and smoking history.
The risk factors significantly associated with the distribution of narrow arteries included age (age ≥ 65 years, P = 0.034), and gender (P = 0.044). In patients aged 65 years or more, the stenosis incidence rate of the VA (64.0%) was higher than that of the MCA (31.4%), ICA (40.0%), and BA (39.5%), respectively.
The stenosis incidence rates of the BA were relatively high in patients with hypertension and those with coronary heart disease. The stenosis rate of the VA was relatively high in patients with diabetes. Patients with hyperlipidemia were susceptible to stenosis of the ICA and BA. Smoking patients were susceptible to stenosis of the MCA. However, there were no significant differences (P > 0.05).
Further analysis for the risk factors of the 37 cases showed that there were 21 cases (56.8%) of simple ACA stenosis, 9 cases (24.3%) of simple PCA stenosis, and 7 cases (18.9%) of both ACA and PCA stenosis. Men (P = 0.000) and smokers (P = 0.000) were more susceptible to ACA stenosis. Patients aged 65 years or more were more susceptible to PCA stenosis (P = 0.000). Patients with hypertension (P = 0.014), those with diabetes (P = 0.000), and those with coronary heart disease (P = 0.001) were susceptible to both ACA and PCA stenosis. There was no statistically significant difference in the distribution of arterial stenosis in patients with hyperlipidemia (P = 0.622) as shown in Figure 1.
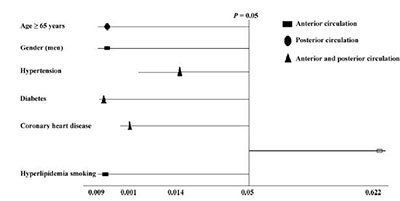
|
| Figure 1 Relationship among risk factors and the distribution of arterial stenosis. |
The multi-factor logistic regression analysis showed that diabetes (odds ratios (OR), 1.672; 95% CI, 1.086-2.574; P = 0.020) was a risk factor for moderately severe intracranial arterial stenosis, while hyperlipidemia (OR, 0.591; 95% CI, 0.393-0.891; P = 0.012) was a protective factor. These results are shown in Figure 2.
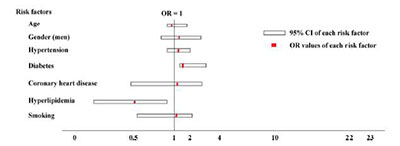
|
| Figure 2 Multi-factor logistic regression analysis of patients with moderately severe intracranial arterial stenosis. CI: Confidence intervals; OR: Odds Ratio. |
Intracranial arterial stenosis or occlusion are important causes for the incidence of ischemic cerebrovascular diseases such as stroke in the elderly[5]. Previous studies found that 80% of the patients who had experienced a first episode of acute cerebral infarction had intracranial or carotid arterial stenosis; patients with TIA who had abnormal TCD examination results were at a high risk of cerebrovascular disease recurrence[6, 7]. Therefore, the early prevention, diagnosis, and treatment of symptomatic intracranial arterial stenosis might reduce the risk of ischemic stroke occurrence. The present study confirmed that TCD results of arterial stenosis are highly consistent with those of digital subtraction angiography (DSA); furthermore, TCD has a higher diagnostic accuracy compared with computed tomography angiography (CTA)[7, 8]. In this study, through the analysis of TCD results, the relationship among intracranial arterial stenosis and its risk factors were further evaluated.
The TCD results of the 486 cases showed that the detection rate of symptomatic intracranial arterial stenosis was 32.3%. Furthermore, among all the stenosis detection rates of target arteries, the rate of MCA stenosis (62.2%) was the highest, which was consistent with previous findings[9]. On the distribution of narrow arteries, MCA stenosis was the most common among single artery stenosis, while most of the patients had complex multi-branch artery stenosis.
In this study, patients were divided into group A and group B according to extracranial artery stenosis. Cases in group B were of patients with pre-existing extracranial artery stenosis whose blood flow had been restored within one month after artery stenting or endarterectomy. There were two advantages of such grouping. First, it avoided the limited detection of intracranial arteries caused by severe stenosis or occlusion of extracranial arteries in previous studies. Second, it reduced the impact on intracranial arteries due to hemodynamic changes, drugs, and other factors caused by recanalization after artery stenting or endarterectomy. The comparative analysis of the risk factors in the two groups showed that patients in group B were older, the proportion of men was higher, and the degrees of stenosis were more serious than that in group A. Besides, studies have found that cerebral blood flow perfusion could be significantly improved in patients who had undergone extracranial-intracranial bypass surgery[10]. It suggests that extracranial stenosis is associated with intracranial stenosis, and patients with carotid arterial stenosis may have an increased risk of the occurrence of intracranial arterial stenosis.
In addition, there were differences in the distribution of cerebral arterial stenosis among patients of different ages and genders, those with hypertension, those with diabetes, those with coronary heart disease, and those who were smokers. The risk factors associated with the distribution of narrow arteries were age (age ≥ 65 years, P = 0.034) and gender (P = 0.044). Age is an independent risk factor for arterial stenosis. An epidemiological study based on the Chinese population found that the incidence of intracranial arterial stenosis in the population over 40 years was 7%[11], and the incidence increased with age[12]. In the current study, the incidence of intracranial vertebral arterial stenosis was high in elderly patients; the incidence of MCA stenosis was high in male patients; patients over 65 years were susceptible to PCA stenosis; patients with hypertension, diabetes, and coronary heart disease respectively were susceptible to both ACA and PCA stenosis. Moreover, a recent study has showed that the recurrent ischemic cerebrovascular events within a few days after an acute ischemic stroke were associated with the location of stenosis[13]. It suggests that the occurrence of cerebral arterial stenosis is closely related to the risk factors, and cerebrovascular events are related to the distribution of stenosis. To prevent the occurrence of ischemic cerebrovascular disease, controlling the risk factors might reduce the incidence and development of cerebral arterial stenosis. According to previous studies, diabetes is associated with the frequency of intracranial large artery disease[14, 15], and it was an important risk factor for the deterioration of intracranial arterial stenosis[16, 17]. The multi-factor logistic regression analysis in the current study showed that diabetes is a major risk factor for moderately severe intracranial arterial stenosis, which was consistent with the study results from related literature domestic and aboard. As diabetes is considered to be a controllable factor, controlling the blood glucose strictly might reduce the incidence rate of moderately severe cerebral arterial stenosis and thus prevent ischemic stroke.
Hyperlipidemia is a major cause for the generation of atherosclerosis[18]. Some studies have suggested that lipid metabolism disorder is closely related to the occurrence of severe cerebral arterial stenosis[19, 20]. Hence, hyperlipidemia might be a control target for the prevention of cerebral arterial stenosis[21]. The level of non-high density lipoprotein cholesterol is an independent predictor for the incidence of asymptomatic intracranial arterial stenosis in male patients[22], especially the increase in low density lipoprotein levels was one of the major risk factors for intracranial arterial stenosis[23]. However, according to the current study, hyperlipidemia might be a protective factor for moderately severe intracranial arterial stenosis. The different results may be due to the following reasons. First, different from previous studies, in this study, patients with at least one mild narrow target artery were included but completely healthy or asymptomatic patients with intracranial arterial stenosis were not. Second, previous studies selected single artery stenosis as research subjects. However, multi-branch arterial stenosis is more common in Chinese population, and comprehensive assessment of arterial stenosis will be more accurate. Hence, in this study, 11 narrow target arteries were included. Third, there are no definite diagnostic criteria for hyperlipidemia. Currently, some studies have shown that statins can reduce the risk of stroke[24, 25]. Therefore, we speculate that in patients with hyperlipidemia, taking statins or other lipid-lowering drugs might delay the progress of intracranial arterial stenosis; however, this should be confirmed further by large sample studies.
In this study, multiple factors related to intracranial arterial stenosis were comprehensively analyzed on the basis of TCD examination results, to provide reliable and useful information for the assessment and control of risk factors of patients with stroke and to reduce the occurrence of ischemic cerebrovascular disease.
Conflict of interests
All contributing authors have no conflict of interests.
| [1] | Ois A, Gomis M, Rodríguez-Campello A, Cuadrado-Godia E, Jiménez-Conde J, Pont-Sunyer C, Cuccurella G, Roquer J. Factors associated with a high risk of recurrence in patients with transient ischemic attack or minor stroke. Stroke, 2008, 39(6): 1717–1721. DOI:10.1161/STROKEAHA.107.505438 |
| [2] | Wang YJ, Zhao XQ, Liu LP, Soo YOY, Pu YH, Pan YS, Wang YL, Zou XY, Leung TWH, Cai YF, et al. Prevalence and outcomes of symptomatic intracranial large artery stenoses and occlusions in China:The Chinese Intracranial Atherosclerosis (CICAS) Study. Stroke, 2014, 45(3): 663–669. DOI:10.1161/STROKEAHA.113.003508 |
| [3] | Accorsi F. Color Doppler of the extracranial and intracranial arteries in the acute phase of cerebral ischemia. J Ultrasound, 2013, 16(4): 187–193. DOI:10.1007/s40477-013-0036-7 |
| [4] | Turan TN, Makki AA, Tsappidi S, Cotsonis G, Lynn MJ, Cloft HJ, Chimowitz MI. the WASID Investigators. Risk factors associated with severity and location of intracranial arterial stenosis. Stroke, 2010, 41(8): 1636–1640. DOI:10.1161/STROKEAHA.110.584672 |
| [5] | Yamada S, Kobayashi M, Watanabe Y, Miyake H, Oshima M. Quantitative measurement of blood flow volume in the major intracranial arteries by using 123i-iodoamphetamine SPECT. Clin Nucl Med, 2014, 39(10): 868–873. DOI:10.1097/RLU.0000000000000555 |
| [6] | Holzer K, Sadikovic S, Esposito L, Bockelbrink A, Sander D, Hemmer B, Poppert H. Transcranial Doppler ultrasonography predicts cardiovascular events after TIA. BMC Med Imaging, 2009, 9: 13. DOI:10.1186/1471-2342-9-13 |
| [7] | Wang XY, Chen L, Ao Q, Sharma A, Shanker Sharma H. Progress in the research and development of nerve conduits. Transl Neurosci Clin, 2015, 1(2): 97–101. DOI:10.18679/CN11-6030/R.2015.012 |
| [8] | Guan JX, Zhou Q, Ouyang HQ, Zhang SF, Lu ZN. The diagnostic accuracy of TCD for intracranial arterial stenosis/occlusion in patients with acute ischemic stroke:The importance of time interval between detection of TCD and CTA. Neurol Res, 2013, 35(9): 930–936. DOI:10.1179/1743132813Y.0000000230 |
| [9] | Wong KS, Huang YN, Gao S, Lam WWM, Chan YL, Kay R. Intracranial stenosis in Chinese patients with acute stroke. Neurology, 1998, 50(3): 812–813. DOI:10.1212/WNL.50.3.812 |
| [10] | Teng MMH, Jen SL, Chiu FY, Kao YH, Lin CJ, Chang FC. Change in brain perfusion after extracranial-intracranial bypass surgery detected using the mean transit time of computed tomography perfusion. J Chin Med Assoc, 2012, 75(12): 649–653. DOI:10.1016/j.jcma.2012.08.008 |
| [11] | Suri MFK, Johnston SC. Epidemiology of intracranial stenosis. J Neuroimaging, 2009, 19(Suppl 1): 11S–16S. |
| [12] | Su LL, Huang HW, Tan SQ, Wu XH, Zhou GJ. Prevalence of asymptomatic intracranial artery stenosis in middle-aged and elderly population in the community of Foshan city, Guangdong province:A cross-sectional study. Chinese J Epidemiol, 2011, 32(5): 469–472. |
| [13] | Kang J, Kim N, Oh CW, Kwon OK, Jung CK, Kim WJ, Park JH, Ko Y, Noh WY, Jang MU, et al. Symptomatic steno-occlusion of cerebral arteries and subsequent ischemic events in patients with acute ischemic stroke. J Stroke Cerebrovasc Dis, 2014, 23(5): e347–e353. DOI:10.1016/j.jstrokecerebrovasdis.2013.12.028 |
| [14] | Kim BJ, Lee SH, Kang BS, Yoon BW, Roh JK. Diabetes increases large artery diseases, but not small artery diseases in the brain. J Neurol, 2008, 255(8): 1176–1181. DOI:10.1007/s00415-008-0864-0 |
| [15] | Lin T, Lin YH, Kao LL, Kao YH, Yang YH, Chou PS, Wu MN. An association between the location of white matter changes and the behavioral and psychological symptoms of dementia in Alzheimer's disease patients. Transl Neurosci Clin, 2016, 2(1): 8–16. DOI:10.18679/CN11-6030/R.2016.010 |
| [16] | Mendes I, Baptista P, Soares F, Oliveira V, Ferro JM. Diabetes mellitus and intracranial stenosis. Rev Neurol, 1999, 28(11): 1030–1033. |
| [17] | Miyazawa N, Akiyama I, Yamagata Z. Analysis of incidence and risk factors for progression in patients with intracranial steno-occlusive lesions by serial magnetic resonance angiography. Clin Neurol Neurosurg, 2007, 109(8): 680–685. DOI:10.1016/j.clineuro.2007.05.021 |
| [18] | Irace C, Pujia A, Motti C, Massimo F, Gnasso A. Carotid atherosclerosis in subjects with different hyperlipidaemia phenotypes. Int Angiol, 1998, 17(1): 15–21. |
| [19] | Shen Y, Wang J, Wu JW, Qu WK, Wang CX, Gao X, Zhou Y, Wang AX, Wu SL, Zhao XQ. Elevated plasma total cholesterol level is associated with the risk of asymptomatic intracranial arterial stenosis. PLoS One, 2014, 9(7): e101232. DOI:10.1371/journal.pone.0101232 |
| [20] | Xue MZ, Li YJ, Gao XG, Zhang CF. Atherosclerotic stenosis of intracranial and extracranial cerebral arteries in patients with cerebral infarction and the correlative factors. National Med J China, 2011, 91(11): 762–765. |
| [21] | Mackay MT, Prabhu SP, Coleman L. Childhood posterior circulation arterial ischemic stroke. Stroke, 2010, 41(10): 2201–2209. DOI:10.1161/STROKEAHA.110.583831 |
| [22] | Wu JW, Zhang Q, Yang HJ, Gao X, Zhou Y, Wang AX, Wang CX, Zhang SF, Wu SL, Zhao XQ. Association between non-high-density-lipoprotein-cholesterol levels and the prevalence of asymptomatic intracranial arterial stenosis. PLoS One, 2013, 8(5): e65229. DOI:10.1371/journal.pone.0065229 |
| [23] | Carvalho M, Oliveira A, Azevedo E, Bastos-Leite AJ. Intracranial arterial stenosis. J Stroke Cerebrovasc Dis, 2014, 23(4): 599–609. DOI:10.1016/j.jstrokecerebrovasdis.2013.06.006 |
| [24] | Tan TY, Kuo YL, Lin WC, Chen TY. Effect of lipidlowering therapy on the progression of intracranial arterial stenosis. J Neurol, 2009, 256(2): 187–193. DOI:10.1007/s00415-009-0960-9 |
| [25] | Amarenco P, Goldstein LB, Szarek M, Sillesen H, Rudolph AE, Callahan A, 3rd, Hennerici M, Simunovic L, Zivin JA, Welch KMA, et al. Effects of intense low-density lipoprotein cholesterol reduction in patients with stroke or transient ischemic attack:The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial. Stroke, 2007, 38(12): 3198–3204. DOI:10.1161/STROKEAHA.107.493106 |



