Due to the growing aging population and increasing awareness about health, leukoaraiosis (LA) has been attracting more attention. It is one of the most common causes of neurodegenerative disorders in the aging population with an incidence rate of 1.7%–8.6% in the healthy elderly, 78%–93% in the elderly patients with hypertension, and 30%–31% in the elderly patients with Alzheimer's disease[1, 2]. The Leukoaraiosis and Disability in the Elderly study (LADIS) found that the body's ability level was correlated with the extent of white matter damage; furthermore, the degree of white matter lesions was closely correlated to dementia disability assessment scores in the elderly without disability[3]. Currently, age, hypertension, diabetes, and other vascular risk factors are believed to contribute to the structural changes in the arteries of the brain, which eventually leads to LA and brain ischemic injury.
Therapeutic angiogenesis is a new concept introduced in the recent years, which aims to stimulate the growth of the small blood vessels and promote collateral circulation of the myocardium ischemic area, named "self-bypass"[4]. Currently, vascular endothelial growth factor (VEGF) is one of the most effective cytokines to stimulate the growth of blood vessels. The biological functions and characteristics of VEGF have been attracting increasing attention[5]. VEGF gene therapy is viewed as "molecular bypass surgery", which is expected to partially replace surgical bypass grafting and angioplasty. Various studies have demonstrated that VEGF and its receptors are upregulated after cerebral ischemia, and intracerebral injection of VEGF leads to the formation of new blood vessels in the brain by inducing the expression of VEGF receptors in astrocytes[6]. VEGF also plays a direct neuroprotective role independent of angiogenesis, which contributes to the damage of cortical neurons due to hypoxia and low sugar level, resulting in the stimulation of the axon growth.
Olfactory ensheathing cells (OECs) are the cells with dual characteristics of astrocytes and Schwann cells and still retain the characteristics of developmental stages and plasticity[7]. So far, the olfactory system is the only renewable hub in the brain, and its biological functions are extremely widespread, including facilitation of the growth, development, and survival of neurons and promotion of neurite outgrowth. Studies by Ramon-Cueto and others have shown that rodents OECs can accelerate axonal regeneration and remyelination of demyelinated nerve fibers in rats with spinal cord injury[8, 9]. Therefore, the identification of OECs has provided new hope for cell transplantation in the treatment of neurodegenerative disorders.
Based on the above studies, we devised the current study where OECs were isolated and cultured in vitro, and then VEGF was imported into OECs through lentivirus infection. The purpose of this study was to isolate, culture, and purify OECs from the olfactory bulb. The purity of OECs was confirmed using immunofluorescence staining of p74. Then, the lentivirus carrying human VEGF165 gene was constructed and transfected into OECs. The expression level of VEGF mRNA and protein in OECs was detected using Real Time-Polymerase Chain Reaction (RT-PCR) and Enzyme-linked immunosorbent assay (ELISA), respectively. In summary, our data showed that these engineered OECs-VEGF highly express functional VEGF and retain the characteristics of astrocytes and Schwann cells, providing an alternative cell source for cell therapy in LA.
2 Methods 2.1 Purification and identification of OECs from the rat olfactory bulbThe purification of OECs was carried out according to the modified protocol of Cao et al.[10]. Briefly, the olfactory bulbs were harvested from 2.5-month-old male Sprague Dawley rats. The OEC-rich olfactory nerve layer and granular layer were isolated, retained, and trypsinized into single-cell suspension in DF12 medium containing 10% fetal bovine serum (FBS). The OECs were purified using the differential adherence method. The cell suspension was added on uncoated dishes twice, each for 12 h culture at 37 ℃ in 5% CO2, and the floating cell suspension was collected. The purity of cultured OECs was determined by the number of immunoreactive cells for p75 compared with the number of Hoechst-labeled nuclei under a microscope.
2.2 Immunofluorescence stainingCells grown on 5 μg/mL fibronectin and 100 μg/mL poly-D-lysine (PDL) coated coverslips were fixed in 4% paraformaldehyde (PFA) at room temperature for 15-20 min, and then permeabilized with 0.2% Triton X-100 in phosphate buffered saline (PBS) at room temperature for 15 min. The primary antibody of p75 was diluted in 2% bovine serum albumin (BSA) in PBS with 0.1% Triton X-100. After incubation with the fluorophore-conjugated secondary antibodies (Molecular Probes) and Hoechst for nuclei labeling, the cells were mounted with SlowFade Antifade reagent (Molecular Probes) and analyzed using Zeiss Axiovert microscopy.
2.3 Real Time-Polymerase Chain Reaction (RT-PCR)Total mRNA was isolated from cells by TriZOL (Invitrogen) and was incubated with DNaseI for 15 min to remove any contaminating genomic DNA. The cDNA was generated through Oligo-dT priming method from 1 μg of total RNA with Transcriptor First Strand cDNA Synthesis Kit (Roche). Quantitative RT-PCR analysis was carried out using the ABI 7500 real time PCR system (Applied Biosystems). Amplification was performed in a 10-μL reaction system with 2X SYBR Green PCR Master Mix (Applied Biosystems) and specific primer sets for VEGF and GAPDH under the following conditions: 95 ℃ for 10 min, 40 cycles of 15 s at 95 ℃, and 1 min at 60 ℃. A melting curve was added at the end of the program.
2.4 Enzyme-linked immunosorbent assay (ELISA)The level of VEGF secreted by engineered OECs was detected using ELISA. According to the manufacturer's protocol, standards and controls were included. First, the log/log standard curves were obtained by averaging the optical density readings from the standard results. To determine the biomarker concentrations, each sample was assayed in duplicates, and the sample optical density (OD) readings were averaged. The protein concentrations were determined from the log/log standard curve.
3 Results 3.1 Sampling, separation, cultivation, purification, and identification of OECs from the olfactory bulbThe primary OECs, isolated using the differential adherent method, showed a typical spindle-like morphology with two or three processes (Figure 1a) and accounted approximately 98% of all cells[11]. There were other common forms, with multiple swellings and irregular shape. In addition, the cultivated OECs showed the capability of colony formation (Figure 1b). With full and complete culture nutrients, cells with poor activity died gradually and separated from the bottom of the culture plate. However, most of the cells transformed from multiple projections to spindleshaped form with strong proliferation ability and continued to proliferate.
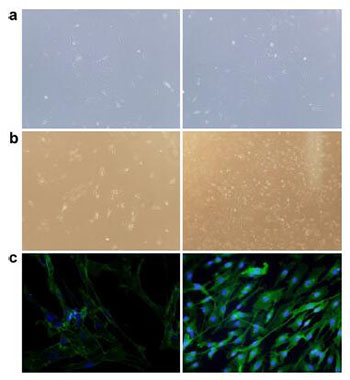
|
| Figure 1 Morphology and identification of OECs isolated from rat olfactory bulbs. a. The primary OECs showed a typical spindlelike morphology with two or three processes or appearance (left picture: 40× magnification and right picture: 100× magnification). b. OECs formed colonies in the DF12 culture medium supplemented with 15% FBS, 2 μM forskolin, and 10 ng/mL bFGF (left picture: 40× magnification and right picture: 100× magnification). c. OECs were subjected for the immunofluorescence staining of p75 (Green) with Hoechst for nuclei labeling (left picture with 40× magnification and right picture with 100× magnification). |
Immunofluorescence staining demonstrated that most of the cells were p75+ (Figure 1c). The purity of OECs became much lower after several passages, and the percentage of OECs declined to 78% and 58% in the second and third generation, respectively (data not shown). Moreover, cellular morphology dramatically changed from spindle-like shape to that with multiple swellings after passage with the disappearance of tight junctions. The proliferative capacity of primary OECs was strong. However, as the number of passages increased, the purity of OECs decreased. The cell shape diversified, and the proliferation rate slowed down. Thus, only the primary and second passages of OECs were used to avoid the variance of low purity after several passages.
3.2 Construction of the lentivirus for VEGF overexpression in OECsThe target gene fragment was amplified from the plasmid containing human VEGF gene and re-linked to the plasmid used as lentivirus packaging vectors. The accuracy of the target gene sequence was confirmed by sequencing the positive transformants and comparing with the NCBI on the target gene sequences. As the lentivirus vector contained EGFP gene, the transfection efficiency could be easily monitored using fluorescence microscopy. Twenty-four hours after transfection in 293T cells, EGFP+ cells were observed on the plate, indicating that the expression of the target gene and EGFP was normal (Figure 2a). The predicted size of VEGF protein is 22 kDa. The protein at 22 kDa near the bands extracted from VEGF plasmid-transfected 293T cells was detected using western blot with specific antibody, which was consistent with the protein synthesized by the target gene (Figure 2b). Our results indicated that the construction of VEGF lentivirus was successful.
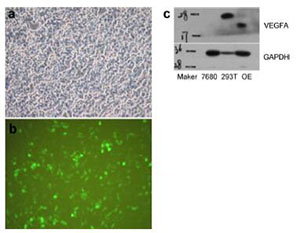
|
| Figure 2 Construction of VEGF lentivirus. a–b. Human VEGF gene was cloned into the lentivirus vector with the marker of EGFP. The representative picture of transfected 293T cells with the VEGF lentivirus vector. c. The total protein of transfected 293T cells was subjected to western blotting for the VEGF specific antibody detection. |
To find the optional infection condition of VEGF lentivirus, OECs were infected with different multiplicity of infection (MOI): 1, 10, 50, and 100 (Figure 3). At MOI = 10, the efficiency of infection was more than 80%, thus the MOI = 10 was used in our following experiments (Figure 4a).
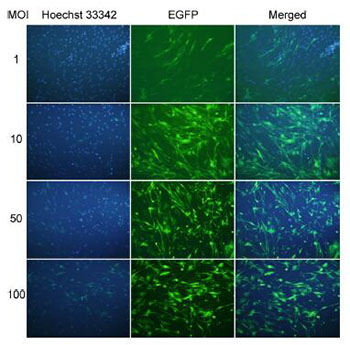
|
| Figure 3 The infection efficiency with serial MOIs of control lentivirus. The OECs were infected with serial MOIs of control lentivirus: 1, 10, 50, and 100. The infection efficiency was monitored through fluorescence microscope with Hoechst for nuclei labeling. |
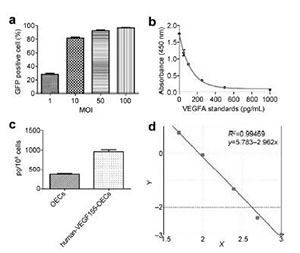
|
| Figure 4 Quantification of infection efficiency for control lentivirus and the detection of secreted VEGF by OECs-VEGF. a. The percentage of GFP+ cells with different MOIs of control lentivirus: 1, 10, 50, and 100. b. The VEGF standards and their absorbance at 450 nm were plotted. c. Quantification of secreted VEGF by control OECs and OECs-VEGF. D. The log/log curve for standards with R2 = 0.99. |
To verify the overexpression of VEGF in OECs, the supernatant of infected OECs was collected and subjected to VEGF ELISA, performed according to the manufacturer's protocol. The absorbance at 450 nm gradually increased with the increasing VEGF standards (Figure 4b), and absorbance values were highly linear correlated with the standard protein concentrations (Figure 4d). Moreover, the concentration of VEGF in OECs was 1000 pg/108 cells, compared to the control group with 430 pg/108 cells (Figure 4c). In addition, EGFP+ cells were also observed in the OECs infected with VEGF lentivirus. The expression level of VEGF was confirmed by RT-with specific primers for GAPDH and VEGF, and the melting curved showed the specific polymerase product.
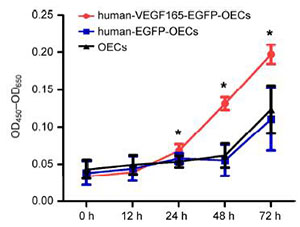
VEGF is believed to be a potent proangiogenic factor, which plays a critical role in both physiological and pathological angiogenesis; furthermore, it promotes the proliferation of human umbilical vein endothelial cells (HUVECs) by activating p38 MAPK kinase[12–14]. To test the functional activation of VEGF secreted by genetically engineered OECs-VEGF, HUVECs were treated with the supernatant of OECs-VEGF, and the proliferation rate was investigated. Our results showed that the OD450-OD460 of the HUVECs treated with the supernatant from OECs-VEGF was much higher than the group treated with the control supernatant (Figure 5). This indicates that the genetically engineered OECsVEGF produced VEGF with functional activity.
|
| Figure 5 The functional activity of secreted VEGF by OECs-VEGF. The supernatant of cultivated OECs-VEGF was collected and the HUVECs were treated with the supernatant for up to 72 h. The OD450-OD460 values of HUVECs were compared between OECs, EGFP-OECs, and VEGF-OECs groups (* demonstrates P < 0.05). |
In summary, our data demonstrated that the primary OECs were successfully isolated from rat olfactory bulbs, and the purity of these cells were extremely high at the first and second passages. Furthermore, VEGF was introduced into OECs through lentivirus, and the genetically engineered OECs-VEGF was successfully generated. The overexpression of VEGF in OECs was confirmed using RT-PCR and ELISA at mRNA and protein levels, respectively. Finally, the secreted VEGF in the supernatant significantly promoted the proliferation of HUVECs, indicating the biological activity of genetically engineered OECs-VEGF.
As LA is a chronic ischemic disease, two major pathophysiological processes (neurogenesis and angiogenesis) are triggered after cerebral ischemia, which have an important role in the recovery of damaged nerve function[15]. To improve the local blood supply is a fundamental solution to LA treatment, which prevents the progression of the disease to the more serious dementia and cerebral infarction[16]. Thus, OECs possess high potential in the cell therapy of patients with LA, due to their dual characteristics of astrocytes and Schwann cells. In addition, VEGF is a notable chemokine, which functions both in neurogenesis and in angiogenesis, thus having high potential as a therapeutic agent[17]. Until now, there are rare reports about VEGF changes in the lesions and treatment of LA. The success of VEGF therapy in the heart-cerebrovascular experiments paved a way for the study of LA[18]. Further explorations are required for how to introduce exogenous VEGF protein into lesions and apply VEGF gene therapy in cerebrovascular disease. Ideally, the exogenous VEGF should improve the blood supply to the damaged brain tissue and reduce the extent of their ischemia.
Moreover, the main LA pathological changes are ischemic demyelination and decline in myelin content[19]. Thus, VEGF was introduced into OECs in this study to improve remyelination and slow down the progress of LA. Based on the biological role of OECs, many studies have focused on the application of olfactory ensheathing cell transplantation to treat the demyelination of nerve tissue[20]. Based on the study on the relationship between OECs and apoptosis, it is possible to enhance the understanding of the therapeutic effect and mechanism of OECs and expand its range of treatment and improve their treatment[21]. With further understanding of the mechanism at the molecular level, gene therapy has become the new entry point for cerebrovascular disease prevention and treatment.
The development of gene transfection technology enables the application of genetically engineered cells in the treatment of neurodegenerative disorders[22]. They not only can exert a synergic effect but also resolve the low expression and poor continuity of VEGF, which is the bottleneck to provide effective solution for cell therapy. The idea of cell transplantation and gene therapy in LA represents a new kind of practical breakthrough. Engineered OECs-VEGF are passaged and amplified in large quantities in the LA model, and eventually differentiated into cells of the nervous system, which may have an important role in the promotion of axonal regeneration and repair of myelin. This study is ongoing in our lab. In conclusion, the genetically engineered OECs-VEGF have strong biological activity and continually secret functional VEGF to promote vascular endothelial regeneration, thereby improving LA nerve tissue microcirculation, promoting angiogenesis, and reducing ischemic injury of the nerve tissue.
Conflict of interests
All contributing authors have no conflict of interests.
| [1] | Lin TC, Lin YH, Kao LL, Kao YH, Yang YH, Chou PS, Wu MN. An association between the location of white matter changes and the behavioral and psychological symptoms of dementia in Alzheimer's disease patients. Transl Neurosci Clin, 2016, 2(1): 8–16. DOI:10.18679/CN11-6030/R.2016.010 |
| [2] | Sarabia-Cobo CM, Pérez V, Hermosilla C, Nuñez MJ, de Lorena P. Apathy and leukoaraiosis in mild cognitive impairment and Alzheimer's disease:Multicenter diagnostic criteria according to the latest studies. Dement Geriatr Cogn Dis Extra, 2014, 4(2): 228–235. DOI:10.1159/000363227 |
| [3] | Schmidt R, Ropele S, Ferro J, Madureira S, Verdelho A, Petrovic K, Gouw A, van der Flier WM, Enzinger C, Pantoni L, et al. Diffusion-weighted imaging and cognition in the leukoariosis and disability in the elderly study. Stroke, 2010, 41(5): e402–e408. DOI:10.1161/STROKEAHA.109.576629 |
| [4] | Brown WR, Thore CR. Review:Cerebral microvascular pathology in ageing and neurodegeneration. Neuropathol Appl Neurobiol, 2011, 37(1): 56–74. DOI:10.1111/nan.2011.37.issue-1 |
| [5] | Dehghanian F, Hojati Z, Kay M. New insights into VEGF-A alternative splicing:Key regulatory switching in the pathological process. Avicenna J Med Biotechnol, 2014, 6(4): 192–199. |
| [6] | Liu F, Ni JJ, Huang JJ, Kou ZW, Sun FY. VEGF overexpression enhances the accumulation of phospho-S292 MeCP2 in reactive astrocytes in the adult rat striatum following cerebral ischemia. Brain Res, 2015, 1599: 32–43. DOI:10.1016/j.brainres.2014.12.014 |
| [7] | Vincent AJ, Taylor JM, Choi-Lundberg DL, West AK, Chuah MI. Genetic expression profile of olfactory ensheathing cells is distinct from that of Schwann cells and astrocytes. Glia, 2005, 51(2): 132–147. DOI:10.1002/(ISSN)1098-1136 |
| [8] | Ramón-Cueto A. Olfactory ensheathing glia for nervous system repair. Exp Neurol, 2011, 229(1): 1. DOI:10.1016/j.expneurol.2011.03.011 |
| [9] | Wang DJ. Why does a little mean a lot when you have nothing? A brief review of cell therapy strategies for spinal cord injury. Transl Neurosci Clin, 2015, 1(2): 102–109. DOI:10.18679/CN11-6030/R.2015.013 |
| [10] | Cao L, Liu L, Chen ZY, Wang LM, Ye JL, Qiu HY, Lu CL, He C. Olfactory ensheathing cells genetically modified to secrete GDNF to promote spinal cord repair. Brain, 2004, 127(3): 535–549. |
| [11] | Wewetzer K, Verdú E, Angelov DN, Navarro X. Olfactory ensheathing glia and Schwann cells:Two of a kind?. Cell Tissue Res, 2002, 309(3): 337–345. DOI:10.1007/s00441-002-0607-y |
| [12] | Ali N, Yoshizumi M, Fujita Y, Izawa Y, Kanematsu Y, Ishizawa K, Tsuchiya K, Yano S, Sone S, Tamaki T. A novel Src kinase inhibitor, M475271, inhibits VEGF-induced human umbilical vein endothelial cell proliferation and migration. J Pharmacol Sci, 2005, 98(2): 130–141. DOI:10.1254/jphs.FP0040850 |
| [13] | Rousseau S, Houle F, Landry J, Huot J. p38 MAP kinase activation by vascular endothelial growth factor mediates actin reorganization and cell migration in human endothelial cells. Oncogene, 1997, 15(18): 2169–2177. DOI:10.1038/sj.onc.1201380 |
| [14] | McMullen M, Keller R, Sussman M, Pumiglia K. Vascular endothelial growth factor-mediated activation of p38 is dependent upon Src and RAFTK/Pyk2. Oncogene, 2004, 23(6): 1275–1282. DOI:10.1038/sj.onc.1207243 |
| [15] | Morimoto T, Yasuhara T, Kameda M, Baba T, Kuramoto S, Kondo A, Takahashi K, Tajiri N, Wang FF, Meng J, et al. Striatal stimulation nurtures endogenous neurogenesis and angiogenesis in chronic-phase ischemic stroke rats. Cell Transplant, 2011, 20(7): 1049–1064. DOI:10.3727/096368910X544915 |
| [16] | Adib-Samii P, Devan W, Traylor M, Lanfranconi S, Zhang CR, Cloonan L, Falcone GJ, Radmanesh F, Fitzpatrick K, Kanakis A, et al. Genetic architecture of white matter hyperintensities differs in hypertensive and nonhypertensive ischemic stroke. Stroke, 2015, 46(2): 348–353. DOI:10.1161/STROKEAHA.114.006849 |
| [17] | Crafts TD, Jensen AR, Blocher-Smith EC, Markel TA. Vascular endothelial growth factor:Therapeutic possibilities and challenges for the treatment of ischemia. Cytokine, 2015, 71(2): 385–393. DOI:10.1016/j.cyto.2014.08.005 |
| [18] | Bouleti C, Mathivet T, Coqueran B, Serfaty JM, Lesage M, Berland E, Ardidie-Robouant C, Kauffenstein G, Henrion D, Lapergue B, et al. Protective effects of angiopoietin-like 4 on cerebrovascular and functional damages in ischaemic stroke. Eur Heart J, 2013, 34(47): 3657–3668. DOI:10.1093/eurheartj/eht153 |
| [19] | Grueter BE, Schulz UG. Age-related cerebral white matter disease (leukoaraiosis):A review. Postgrad Med J, 2012, 88(1036): 79–87. DOI:10.1136/postgradmedj-2011-130307 |
| [20] | Ekberg JAK, St John JA. Crucial roles for olfactory ensheathing cells and olfactory mucosal cells in the repair of damaged neural tracts. Anat Rec, 2014, 297(1): 121–128. DOI:10.1002/ar.v297.1 |
| [21] | Bartolomei JC, Greer CA. Olfactory ensheathing cells:Bridging the gap in spinal cord injury. Neurosurgery, 2000, 47(5): 1057–1069. DOI:10.1097/00006123-200011000-00006 |
| [22] | Mouhieddine TH, Kobeissy FH, Itani M, Nokkari A, Wang KKW. Stem cells in neuroinjury and neurodegenerative disorders:Challenges and future neurotherapeutic prospects. Neural Regen Res, 2014, 9(9): 901–906. DOI:10.4103/1673-5374.133129 |



