2. Department of Nephrology, Beijing Luhe Hospital, Capital Medical University, Beijing 101149, China;
3. Department of Psychiatry, Zaozhuang Mental Health Center, Zaozhuang 277103, China;
4. Department of Neurosurgery, The Fifth People's Hospital of Datong, Regional Medical Center of Shanxi Province, Datong 037006, China
Since September 2015, he has been working at the Department of Neurosurgery at the Beijing Tsinghua Changgung Hospital, School of Clinical Medicine at Tsinghua University.
He specializes in spine and spinal cord diseases, including Chiari malformation, syringomyelia, spinal cord tumors, lumbar spondylosis, and cervical spondylosis. At present, he has published 5 papers that are available in the Science Citation Index, 15 papers in core Chinese journals, and 3 bibliographic books. He has managed 2 National Natural Science Fund Projects and 1 Project from the Ministry of Health, Health Science, and Technology Development.
Dan Yuan graduated in July 2011 with the master degree from Beijing Youyi Hospital of Capital Medical University.
Since September 2011, she has been working at the Department of Nephrology at Beijing Luhe Hospital of Capital Medical University, Beijing, China. Currently, she has published 3 core Chinese journal articles and 2 Science Citation Index articles.
At present, her main research area is the clinical diagnosis and treatment of nephrological diseases.
Yaxing Sun received the bachelor degree from Taishan Medical University on July 2011. Since September 2011, he has been working at the Department of Psychiatry of Zaozhuang Mental Health Center in Shandong, China.
He specializes in mental diseases, including postoperative depression, vesania, and schizophrenia. At present, he has published 2 Science Citation Index articles and 5 core Chinese journal articles.
Zhanquan Zhang was born on May 22, 1971. In September 1991 to July 1996, he studied medicine at Lanzhou University. Currently, he works at the Department of Neurosurgery, Fifth People's Hospital in Datong City.
He serves as the deputy director of the Department and is an associate professor at Datong University. He studied micro-neurosurgery in April 2005 for one year at Tianjin Huanhu Hospital and neuro-endoscopic technology in 2011 at Qilu Hospital of Shandong University. In July 2015, he completed a spine course at Tsinghua Changgung Hospital.
James Wang is the deputy president and chief physician at the Department of Neurosurgery, Beijing Tsinghua Changgung Hospital, School of Clinical Medicine, Tsinghua University.
Dr. Wang acquired his USA Educational Commission for Foreign Medical Graduatescertified M.D. in 1993 and became an American Association of Neurological Surgeons-certified clinical neurosurgeon in 2003. Since 2010, Dr. Wang has been working as the chief physician at Seattle Providence Hospital and an associate professor at Washington University.
Dr. Wang has extensive experience in the surgical treatment of intracranial tumors, functional neurological diseases, and spine and spinal cord disorders. To date, Dr. Wang has published more than 30 academic articles in both domestic and foreign academic journals.
Guo Yi graduated in July 2012 from the Beijing Tiantan Hospital, Capital Medical University with the Ph.D. degree.
Since July 2015, he has been working at the Neurosurgery Department of the Beijing Tsinghua Changgung Hospital as an attending physician. Currently, he has published 7 core Chinese journal articles and 15 Science Citation Index articles and has been the principal investigator on four scientific studies.
At present, his main research areas are the clinical diagnosis and treatment of traumatic brain injury and cerebral vascular and spinal cord diseases.
Guoqin Wang graduated from Hubei Chinese Medicine University School of Nursing in June 2009.
Since August 2014, she has been working as an electrophysiologist at the Beijing Tsinghua Changgung Hospital.
Dongkang Liu graduated from Beijing Tiantan Hospital of Capital Medical University in July 2015 with the master degree.
Since September 2015, he has been a Ph.D. candidate in the Neurosurgery Department at the Beijing Tsinghua Changgung Hospital. To date, he has published 3 core Chinese journal articles and 1 Science Citation Index article.
His current main research area is the clinical diagnosis and treatment of spinal cord diseases.
Peng Cheng graduated from Beijing Union Medical College in February 2014 with the master degree.
Since March 2014, he has been working as an epidemiologist at Beijing Tsinghua Changgung Hospital.
Linkai Jing graduated in July 2016 from the Beijing Tiantan Hospital of Capital Medical University with the master degree.
Since September 2016, he has been a Ph.D. candidate in the Neurosurgery Department of the Beijing Tsinghua Changgung Hospital. His publications include 14 core Chinese journal articles and 5 Science Citation Index articles.
His current main area of research is the clinical diagnosis and treatment of spinal cord diseases.
Feng Yang graduated from Peking University in June 2014 and since has been working at the Beijing Tsinghua Changgung Hospital.
In addition, he has participated in a meta-analysis research project that was sponsored by the General Administration of Sports and in National Science Foundation-supported basic research on laser light-excitation at different wavelengths to evaluate different spectral features.
Peihai Zhang graduated from the seven-year clinical medicine program at China Medical University in July 2014 with the master degree.
Since August 2014, he has been a resident at the Neurosurgery Department of the Beijing Tsinghua Changgung Hospital. As a 4th-year resident, he has published 5 core Chinese journal articles and participated in 5 research projects, some of which were supported by the Beijing Natural Science Foundation. In addition, he participates in the Youth Fund of Tsinghua University.
His current main research areas include neuro-navigation technology, multimodal image fusion, and the clinical diagnosis and treatment of spinal cord diseases.
Huifang Zhang graduated from Jinlin Medical college in June 2009. From July 2009 to March 2015, she worked at Beijing Tiantan Hospital of Capital Medical University. Since then, she has been working at Beijing Tsinghua Changgung Hospital as a nurse practitioner and has recently completed the supervisor nurse examination.
Youtu Wu, M.D., is a resident physician at the Department of Neurosurgery, Beijing Tsinghua Changgung Hospital, School of Clinical Medicine, Tsinghua University, Beijing, China.
Dr. Wu graduated from Capital Medical University and began his residency in 2016 at Beijing Tsinghua Changgung Hospital. To date, Dr. Wu has first authored 3 academic articles in foreign journals and 2 academic articles in domestic journals.
Wei Shi graduated from the Medical School of the Chinese PLA in July 2015 with the Ph.D. degree.
In August 2015, he began a residency at the Neurosurgery Department of the Beijing Tsinghua Changgung Hospital. As a 3rd-year resident, he has published 1 core Chinese journal article and 2 Science Citation Index articles and participated in 4 research projects, some of which have been supported by the Beijing Natural Science Foundation.
At present, his main research areas include the clinical diagnosis and treatment of traumatic brain injury and cerebral vascular and spinal cord diseases.
Guaihuai Wang is the director and chief physician at the Department of Neurosurgery at the Beijing Tsinghua Changgung Hospital, School of Clinical Medicine at Tsinghua University. Dr. Wang has previously mentored by renown neurosurgeon and academician Zhongcheng Wang and has more than 25 years of experience in the surgical treatment of brain stem and spinal cord tumors, spinal congenital dysplasia, and spinal degeneration diseases.
To date, Dr. Wang has performed approximately 10, 000 operations for spinal cord diseases, including a successful resection of the intramedullary tumor from the medulla oblongata to level T6, which is one of the longest reported tumors in medical history.
Thus far, he has managed 10 funded research projects and published more than 80 articles in both domestic and foreign academic journals.
Spinal ependymomas (SEs) are the most common type of adult intramedullary tumor. SEs arise from ependymal epithelial cells at the center of the spinal cord, and often develop in the cervical cord[1-5]. SEs present with slow, longitudinal growth along the spinal cord and show expandability. Most SEs have clear boundaries, and surgical removal of the whole tumor is the primary treatment option[6-10]. However, absolute tumor boundaries can be difficult to determine, particularly in patients with tumor-associated strokes. Further, the spinal cord has numerous septa that tightly adhere to the cord, making tumor boundary determination and tumor residual detection difficult. Thus, assessing tumor boundaries and detecting tumor residuals during surgery is challenging. In the present study, we performed a retrospective study on the outcomes of fluorescein sodium (FS) use during surgical removal of SEs.
2 Materials and methods 2.1 Clinical documentationSpinal tumor patients (n = 193) admitted to Beijing Tsinghua Changgung Hospital and followed-up between December 2014 and December 2016 were included. Of these, 112 cases of SE were pathologically confirmed (78 men, 34 women; age range, 28-67 years; mean age, 41.3 ± 5.6 years). Prior to surgery, 106 patients exhibited sensory disorders in one (unilaterally) or two (bilaterally) limbs; 102 patients had movement disorders in all four extremities (including 92 with decreased muscle strength), five patients had lower limb paralysis (including two patients with sudden paralysis due to strokes, confirmed using magnetic resonance imaging (MRI)), four patients had respiratory dysfunction, and 24 patients had voiding dysfunction/ obstructed defecation. Preoperative McCormick spinal cord evaluations showed that 14 patients were at grade Ⅰ, 35 at grade Ⅱ, 54 at grade Ⅲ, and nine at grade Ⅳ.
2.2 Imaging findingsAll patients underwent 3.0-T MRI examinations; lesions were observed in the cervical cord (86 patients), thoracic spine (18), conus medullaris (5), and filum terminale (3). The lesions involved 1-2 segments (12 patients), 3-4 segments (58), 5-6 segments (26), or ≥7 segments (16) (Figures 1-3). There were 90 patients with combined syringomyelia, 94 with increased tumor solidification, 14 with intramedullary tumor-associated stroke and heterogeneous tumor solidification, and four with unchanged tumor solidification.
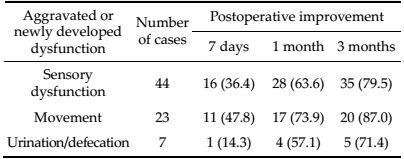
|
| Figure 1 Pre-and postoperative magnetic resonance imaging of intramedullary ependymomas at C1-T1, and intraoperative observations. (a) Preoperative sagittal T1 image reveals spinal cord thickening. (b) T2 image shows high intramedullary signal intensity at C1-T1, but no signal at either end. (c) Enhanced scanning reveals distinctive tumor enhancement. (d) Intact tumor with clear boundaries, as viewed under a microscope. (e) Tumor images, viewed via fluorescence microscopy, showing a distinctive basal structure. Sagittal T1 (f) and T2 (g) images, 3 months after surgery, do not show tumor residuals or relapses. |
Patients received FS (3-4 mg/kg intravenous; Lishede, Alcon, Fort Worth, TX, USA), 30 min before receiving general anesthesia. A Meditec Pentero 900 microscope (Carl Zeiss, Jena, Germany) equipped with a 560-nm, yellow fluorescence source was used for tumor imaging. Body positioning was determined by the tumor position. During each surgery, the somatosensory evoked potentials and surface electromyograms of the four extremities were monitored. Based on the tumor location and number of involved segments, a medial route via the posterior neck was adopted to expose the laminas along the upper and lower tumor boundaries. The corresponding laminas were then opened and removed with an ultrasound bone scalpel to further expose the dura mater. The dura mater was cut and lifted, followed by cutting of the arachnoid mater.
Each tumor's appearance and boundaries were microscopically observed under white light. During surgery, yellow fluorescence (560 nm) was used to image the tumor. A short posteromedial incision was made on the spinal cord to avoid causing tumor bleeding that would blur the tumor boundaries. The tumor boundaries were completely exposed. The majority of the tumors had visible boundaries and could be removed. However, tumor removal was relatively difficult in patients who had experienced tumor-associated strokes, and required careful surgery to avoid excessive traction and basal spinal cord damage. Tumor removal was performed with proper management of hemostasis. During surgery, tumors were exposed, imaged, and removed under alternating white and fluorescent illumination.
After completing tumor removal, fluorescence was also used to look for tumor residuals, particularly at the tops and bottoms of the cysts, and along the cyst walls. After the tumors were removed, the pia and dura maters were sutured closed. Suturing of the arachnoid was dependent on the existing conditions. The laminas that had been removed were replaced and stabilized using two-hole connectors from the craniomaxillofacial device. Layered closure was then performed, and postoperative respiration and movement were monitored.
2.4 Outcome evaluationClinical function changes, McCormick grading[11], tumor status, and complications (based on MRI examinations) were monitored at postoperative day 7 and months 1 and 3.
2.5 StatisticsAll data analyses were performed using statistical software (SAS v9.3; IBM, Armonk, NY, USA). Quantitative data are expressed as means ± standard deviations; count data are expressed as percentages or constituent ratios. The chi-square test was used to determine differences in postoperative functional improvements, and the rank sum test was used to assess differences in the McCormick spinal function; P < 0.05 was considered statistically significant.
3 Results 3.1 Intraoperative tumor statusIn the absence of FS, 80 patients demonstrated clear tumor boundaries and 32 had blurry tumor boundaries resulting from tumor adhesion to the spinal cord, including 14 with severe adhesions and intramedullary tumor-associated strokes (Figures 1-3). Under fluorescence, tumors were visible in 94 patients and invisible or indistinct in 18, including 14 with intramedullary tumor-associated strokes and indistinct MRI enhancement and four without MRI enhancement. Under fluorescence, complete tumor removal was accomplished in 92% (103/112) of the patients. Of the nine patients with tumors that could not be completely removed, five had myxopapillary ependymomas at the conus medullaris and four also had strokes and severe adhesions.
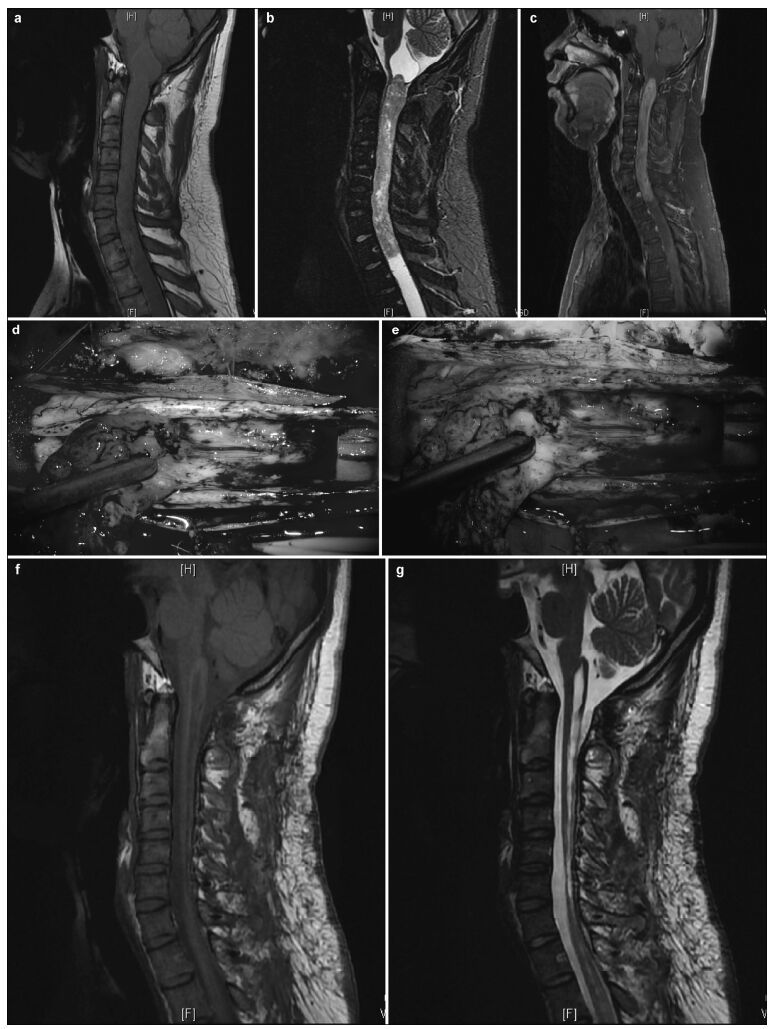
|
| Figure 2 Pre-and postoperative magnetic resonance imaging of intramedullary ependymomas at C2-T2, and intraoperative observations. (a) Preoperative sagittal T1 image reveals spinal cord thickening. (b) T2 image shows high intramedullary signal intensity at C2-T2, but no signal at either end. (c) Enhanced scanning reveals distinctive enhancement of the tumor mass and cyst wall. Tumor ends, as micrographed using white light (d) and fluorescence (e). (f) Cysts at the tumor ends, microscopically probed with white light, do not show the tumor. (g) Distinct cyst walls are evident under fluorescent illumination, revealing a suspicious tumor that was removed. At 3 months, the sagittal T1 (h) and T2 (i) images and the enhanced scan (j) reveal no evidence of tumor residuals or relapses. |
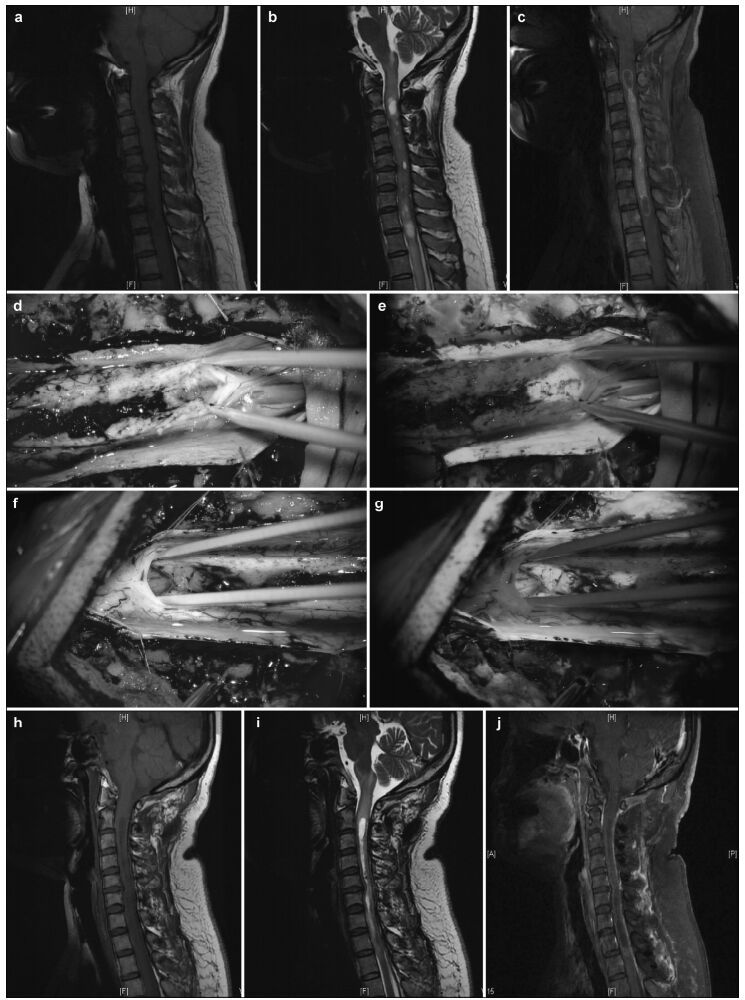
|
| Figure 3 Intramedullary ependymomas at C3-C7, and tumor-associated stroke. (a) Preoperative T1 image showing spinal cord thickening and cysts in the septum. (b) Preoperative T2 image of a mass at C3-C7 and cysts in the septum. Stroke was not detected. (c) Preoperative enhanced magnetic resonance image showing an intramedullary tumor as a distinctive mass at C3-C7. Stroke enhancement was undetected. (d) Indistinct tumor images, intraoperatively, under fluorescent illumination. (e) Distinct intraoperative image of a tumor under the microscope. (f) Intraoperative and 1-month postoperative T1 images show the absence of tumor residuals or relapses. (g) T2 images, 1 month after surgery, show the absence of tumor residuals and relapses. |
Three months after surgery, sensory function and movement were improved in most patients demonstrating preoperative dysfunction (Tables 1 and 2). Urination and defecation functions also improved in 66.7% (16/24) of the patients. At the 3-month time point, urination and defecation functions were unchanged, compared with observations on postoperative day 7 and 1 month (P > 0.05). Patients with preoperative respiratory dysfunction (n = 4) had recovered by postoperative day 7.
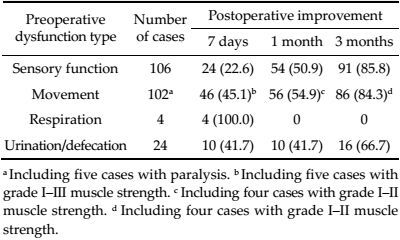 |
By 3 months after surgery, sensory function and movement were improved in most patients who had developed preoperative dysfunction or had demonstrated postoperative aggravation of their dysfunction. Of these patients, the rate of improved urination/ defecation function was 71.4% (10/14). Urination and defecation functions were unchanged at the 3-month follow-up, compared with functioning at postoperative day 7 and 1 month (P > 0.05). Surgery did not cause new cases of respiratory dysfunction.
3.3 Comparison of imaging and intraoperative fluorescence findingsOf the 112 cases, 94 (83.9%) had enhanced preoperative MRIs and distinct intraoperative fluorescent images, four had indistinct MRI enhancements and fluorescent images during surgery, and 14 had tumor-associated strokes associated with non-uniform MRI enhancement and indistinct fluorescent imaging, intraoperatively (Figure 3). The present study included 90 patients with combined syringomyelia, 88 of whom did not present with cyst wall enhancement during either their preoperative MRI examinations or with intraoperative fluorescent cyst walls, and one who presented with preoperative cyst wall MRI enhancement and intraoperative fluorescent cyst wall images.
3.4 Spinal cord functional classification improvementThe McCormick spinal cord functional classification, 3 months after surgery, was grade Ⅰ in 74.1% (83/112) of patients, grade Ⅱ in 16.9% (19/112), grade Ⅲ in 5.3% (6/112), and grade Ⅳ in 3.5% (4/112) of the patients; these proportions were significantly different from the preoperative classifications (P < 0.01). In the nine patients at grade Ⅳ, five had improved by 3 months, postoperatively. However, of the five patients with hemorrhagic paralysis, only one improved. At the 3-month follow-up, there was no evidence of tumor residuals or relapses, and no evidence of cerebrospinal fluid leakage, infection, respiratory dysfunction, or death during the follow-up period.
4 DiscussionFS is widely used for improving the diagnosis of malignant tumors and vascular lesions, although mainly in eye disease. The use of FS for the location and removal of an intracranial tumor was first reported in 1948[12-16]. However, the requirement for advanced imaging technology and the lack of specificity in the obtained images limited its applications. Under physiological conditions, circulating FS does not typically cross the blood-brain barrier to enter the brain. However, FS can accumulate within neurovascular tumors following endothelial lesions, and is visible under 560-nm yellow light[12]. Factors affecting the use of FS include malignant tumors, vascular permeability defects, blood-brain barrier defects, and angiogenesis[14, 15]. FS has a low molecular weight and reversibly binds to hemoglobin and red blood cells. Therefore, FS can permeate extravascular regions and stain the tissues surrounding a tumor. Recent studies have shown that fluorescent hemoglobin conjugates can also increase tumor contrast[16]. Thus, FS can increase tumor visibility and improve the range of tumor removal. Nevertheless, FS imaging reliability requires further improvement, particularly for determining tumor boundaries and for prognostic evaluations[16].Therefore, in the present study, we examined the use of FS in surgeries treating SEs.
In the present study, all 112 patients with SEs underwent FS-assisted tumor removal, and 94 (83.9%) had distinctive fluorescent imaging findings that aided complete tumor removals. There were 18 patients (16.1%) with indistinct fluorescent tumor images, and the tumors were removed under white light. Koc et al. reported a significantly higher rate of complete glioma removal, from the brain, when using FS (83%), compared with the rate obtained when FS was not used (55%); there were no differences in survival rates[11]. Shinoda et al. also reported an increase in the rates of complete removal of brain gliomas when using FS (84.4%) than when using conventional white light (30.1%)[13]. Theoretically, ependymomas and normal spinal tissues have clear boundaries, allowing for surgical removal of intact tumors. However, this procedure is difficult in patients with tumor-associated strokes, myxopapillary ependymomas at the conus medullaris, or anaplastic ependymomas. In the present study, the rate of complete ependymoma removal when using FS was 92%, higher than that previously reported for FS-assisted complete brain glioma removals.
The McCormick classification showed that spinal function was improved at postoperative day 1 and at the 1-and 3-month follow-ups. Although surgery was associated with temporary sensory (n = 88) and movement (n = 46) dysfunctions, these conditions were improved at the 3-month postoperative follow-ups (improvement rates of 79.5% (70/88) and 87% (40/46), respectively). Thus, both preoperative and surgeryassociated dysfunctions improved during follow-up. Preoperative respiratory dysfunction showed varying degrees of postoperative improvement, possibly because of pressure relief on the spinal cord following tumor removal, and improved blood supply. Patients with tumor-associated, preoperative strokes and paralysis (n = 5) demonstrated suboptimal postoperative improvements, indicating that the surgery timing requires optimization to prevent paralysis and other severe dysfunctions. These observations also indicated that, after paralysis, surgery is still beneficial, although the outcomes are suboptimal. During followup, there was no MRI evidence of tumor residuals or relapses. However, long-term follow-up studies are required to fully monitor tumor relapse.
For patients with tumor-associated stokes, white light is required intraoperatively to detect and remove the tumors. However, hemosiderin deposits can cause tumor cells to tightly adhere to normal spinal cords, increasing the difficulty of assessing tumor boundaries and residuals under both white light and fluorescent illumination. Hyperplasia of small blood vessels increases the blood supply to tumors, which also makes surgery more difficult. In the present study, one patient presented with cysts at both ends of the tumor (determined using MRI) and at the cyst wall (evidenced using intraoperative fluorescent imaging), indicating possible tumor cell infiltration and metastasis.
In summary, fluorescent imaging and preoperative MRI enhancement imaging are useful for detecting tumor infiltration and metastasis. In particular, we found that FS was useful for intraoperative SE removal. Further, a combination of white light and fluorescent microscopic imaging helps visualize tumors and tumor residuals, increasing the resection rate. However, tumors invisible under intraoperative fluorescence still require normal white light for detection. Further, there was no benefit of intraoperative fluorescent imaging for patients with tumor-associated stokes. Further studies in more patients are required to analyze the correlation of MRI imaging with intraoperative fluorescent imaging, and to assess the reliability of this technique, including the sensitivity and specificity of tumor imaging to decrease imaging noise resulting from swollen cells surrounding a tumor. Additionally, studies examining the effects of FS on tumor removal ranges and survival rates are required.
AcknowledgementsThis project was supported by the Beijing Tsinghua Changgung Hospital Fund (No. 12015C1045). The authors thank Peihai Zhang, Huifang Zhang, and Linkai Jing for collecting the archived data and Guihuai Wang and Jin Wang for technical help.
Conflict of interestsNone of the contributing authors have conflicts of interest.
| [1] | Keil VC, Schmitt AJ, Martin SC, Cadoux-Hudson TA, Pereira EA. Optimising treatment strategies in spinal ependymoma based on 20 years of experience at a single centre. J Clin Neurosci, 2016, 29: 52–58. DOI:10.1016/j.jocn.2016.01.003 |
| [2] | Ewelt C, Nemes A, Senner V, Wölfer J, Brokinkel B, Stummer W, Holling M. Fluorescence in neurosurgery:its diagnostic and therapeutic use. Review of the literature. J Photochem Photobiol B, 2015, 148: 302–309. DOI:10.1016/j.jphotobiol.2015.05.002 |
| [3] | Ehrhardt A, Stepp H, Irion KM, Stummer W, Zaak D, Baumgartner R, Hofstetter A. Fluorescence detection of human malignancies using incoherent light systems. Med Laser Appl, 2003, 18(1): 27–35. DOI:10.1078/1615-1615-00084 |
| [4] | Kukreja S, Ambekar S, Sharma M, Sin AM, Nanda A. Outcome predictors in the management of spinal myxopapillary ependymoma:an integrative survival analysis. World Neurosurg, 2015, 83(5): 852–859. DOI:10.1016/j.wneu.2014.08.006 |
| [5] | Xie TH, Qian Jun, Wu XJ, Lu YC, Hu GH, Luo C. Unilateral, multilevel, interlaminar fenestration in the removal of a multisegment cervical intramedullary ependymoma. Spine J, 2013, 13(7): 747–753. DOI:10.1016/j.spinee.2013.02.048 |
| [6] | Kim DH, Kim JH, Choi SH, Sohn Ch, Yun TJ, Kim CH, Chang KH. Differentiation between Intramedullary spinal ependymoma and astrocytoma:comparative MRI analysis. Clin Radiol, 2014, 69(1): 29–35. DOI:10.1016/j.crad.2013.07.017 |
| [7] | Butte PV, Mamelak AN, Nuno M, Bannykh SI, Black KL, Marcu L. Fluorescence lifetime spectroscopy for guided therapy of brain tumors. NeuroImage, 2011, 54(Suppl 1): S125–S135. |
| [8] | Wang GH, Yang J, Liu C, Han B, Li DZ, Chen SY, Yang B. Microneurosurgical management of intramedullary spinal cord ependymomas:report of 173 cases. Chin J Neuro-Oncol, 2007, 5(1): 9–12. |
| [9] | Chen B, Wang HF, Ge PF, Zhao JW, Li WC, Gu HZ, Wang GM, Luo YA, Chen DW. Gross total resection of glioma with the intraoperative fluorescence-guidance of fluorescein sodium. Int J Med Sci, 2012, 9(8): 708–714. DOI:10.7150/ijms.4843 |
| [10] | Chen Z, Jian FZ, Wang YL, Lin F. Microneurosurgery of multisegmental intramedullary spinal cord ependymomas. Chin J Neurosurg, 2006, 22(1): 14–17. |
| [11] | Koc K, Anik I, Cabuk B, Ceylan S. Fluorescein sodiumguided surgery in glioblastoma multiforme:a prospective evaluation. Br J Neurosurg, 2008, 22(1): 99–103. DOI:10.1080/02688690701765524 |
| [12] | Okuda T, Kataoka K, Yabuuchi T, Yugami H, Kato A. Fluorescence-guided surgery of metastatic brain tumors using fluorescein sodium. J Clin Neurosci, 2010, 17(1): 118–121. DOI:10.1016/j.jocn.2009.06.033 |
| [13] | Shinoda J, Yano H, Yoshimura SI, Okumura A, Kaku Y, Iwama T, Sakai N. Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium. J Neurosurg, 2003, 99(3): 597–603. DOI:10.3171/jns.2003.99.3.0597 |
| [14] | Schebesch KM, Proescholdt M, Höhne J, Hohenberger C, Hansen E, Riemenschneider M J, Ullrich W, Doenitz C, Schlaier J, Lange M, Brawanski A. Sodium fluorescein-guided resection under the YELLOW 560 nm surgical microscope filter in malignant brain tumor surgery-a feasibility study. Acta Neurochir, 2013, 155(4): 693–699. DOI:10.1007/s00701-013-1643-y |
| [15] | Kremer P, Fardanesh M, Ding R, Pritsch M, Zoubaa S, Frei E. Intraoperative fluorescence staining of malignant brain tumors using 5-aminofluorescein-labeled albumin. Neurosurgery, 2009, 64(3 Suppl): ons53–ons60. |
| [16] | Diaz RJ, Dios RR, Hattab EM, Burrell K, Rakopoulos P, Sabha N, Hawkins C, Zadeh G, Rutka JT, Cohen-Gadol AA. Study of the biodistribution of fluorescein in gliomainfiltrated mouse brain and histopathological correlation of intraoperative findings in high-grade gliomas resected under fluorescein fluorescence guidance. J Neurosurg, 2015, 122(6): 1360–1369. DOI:10.3171/2015.2.JNS132507 |