2. Department of Pharmacology and Toxicology, Wright State University School of Medicine, Dayton, Ohio 45345, USA
Yang Hu got her medical degree from Huazhong University of Science and Technology. She is now a clinician at Neurology Department, Tongji Hospital. Her research interests are focused on the immunoregulatory capacity of mesenchymal stem cells. She has published four papers as the first author and corresponding author.
Zhouping Tang, M.D., Ph.D., has served as chief physician, professor, doctoral supervisor, vice director of the Neurology Department, and assistant dean at Tongji Hospital (Guanggu Branch), Tongji Medical College, and Huazhong University of Science and Technology. He is a member of the Neurorestoratology Association of Chinese Medical Doctor Association (CMDA) and the Pain and Sensory Dysfunction Group of CMDA. He is in charge of 10 provincial funds, four of which are managed by the National Natural Science Foundation of China (NSFC). In the past five years, he has published 26 SCI papers under the headings of Endocrinology, CNS NEUROSCI THER, and MOL NEUROBIOL, among others. His research studies focus on treatments of intracerebral hemorrhage using transplantation of adipose stem cell-derived neural stem cells and minimally invasive stereotactic puncture.
Zhouping Tang, E-mail:ddjtzp@163.com.
Stem cells are defined as the cells capable of self-renewal and differentiation into one or more specialized cell types[1]. Generally, two types of stem cells have been described: embryonic stem cells (ESCs) and adult stem cells. Although ESCs are pluripotent and can be used to replace any tissue, they may have teratogenic effects, and the procedure to isolate them is ethically controversial. Adult stem cells that have the ability to differentiate into mesodermal lineage tissues such as bone, fat, and cartilage are called mesenchymal stem/stromal cells (MSCs)[2]. It has been traditionally thought that MSCs reside mainly in bone marrow. However, MSCs have been recently obtained from various tissues including adipose tissue[3], dental pulp[4], synovium[5], Wharton's jelly[6], hair follicles[7], and olfactory bulbs[8].
Systemically administered MSCs are a promising tool in the development of regenerative medicine. These cells have the ability to migrate and accumulate at sites of injury and inflammation and promote functional recovery by replacing damaged tissue through paracrine effects, activating endogenous progenitor cells, and/or modulating immune responses[9, 10]. The application of MSCs in cell therapies has increased in recent years because of extensive availability of these cells, convenient isolation methods, less ethical concerns, and relatively low immunogenicity[11]. Numerous preclinical and clinical trials targeting various diseases, such as specific congenital defects, traumatic or ischemic injuries (e.g., tibial fracture, brain and spinal cord injury, liver injury, stroke, and myocardial infarction), immune disorders (arthritis, multiple sclerosis, diabetes mellitus, and Crohn's disease), degenerative diseases, and graft-versus-host disease (GVHD)[10-14] have been performed.
It is generally thought that the efficiency of MSC therapy in attenuating damage after ischemia, inflammation, or injuries mainly depends on the quantity of transplanted cells recruited to the target tissue[15]. Substantial evidence has shown that infused MSCs tend to migrate to the site of lesion. However, studies investigating the homing and the long-term engraftment of these cells finally showed that only a small percentage of cells were detectable in the lesions given that most cells became trapped in the liver, spleen, and lungs in animal models[16-18]. Regarding the central nervous system (CNS), even fewer systematically delivered cells home to the sites of ischemia or traumatic injury because of the blood brain barrier (BBB). The low rate of cell homing, retention, and survival is one of the major limitations in current experimental and clinical studies investigating the therapeutic potential of different types of MSCs[19]. Therefore, understanding the mechanisms of homing may be crucial for enhancing the engraftment efficiency, particularly in cases of intravenous or arterial cell administration. Karp and Leng Teo proposed the definition of MSC homing in 2009[11] by referring to the active or positive arrest of MSCs within the vasculature of a tissue followed by transmigration across the endothelium. A series of complex steps are involved in the homing of MSCs. However, the mechanisms underlying the homing of exogenous and endogenous MSCs have not been clearly elucidated. This review will focus on factors determining the migration of MSCs and propose strategies to improve homing to the target tissue, with a particular focus on cell therapy for CNS disorders.
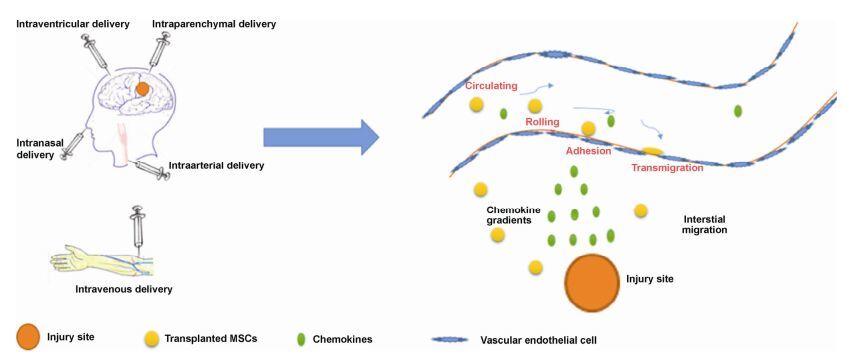
2 In vivo determinants of MSC migrationWhile the mechanisms underlying the homing of leukocytes to the sites of inflammation have been extensively described[20-22], those underlying MSC homing to the sites of interest remain unknown. As in the case of leukocyte homing, recruitment and incorporation of MSCs into ischemic or injured tissue may also require a coordinated multistep process including adhesion to the endothelium, transendothelial migration, chemotaxis, matrix degradation and invasion, and in situ differentiation (see Figure 1).Integrin blocking and knockout studies provided evidence for dependence of MSC migration on selectin and integrin interactions[15, 23]. These results suggest that the engraftment of MSCs within target tissues depends on specific molecular interactions prior to the transmigration step[11]. Besides adhesive interactions that mediate MSC homing to specific sites, chemokines released from the tissue or endothelial cells may promote activation of adhesion ligands, transendothelial migration, chemotaxis, and/or subsequent retention in the surrounding tissue[24-26]. Specific chemokines and their receptors are essential for the migration of MSCs to the sites of inflammation and injury. In addition, MSCs secrete proteases (i.e., matrix metalloproteinases, MMPs) that regulate transmigration and invasion of the endothelial basement membrane and degrade extracellular matrix (ECM) during chemotaxis[11, 27, 28].
|
| Figure 1 The migration of transplanted MSCs through different delivery routes. |
Chemokines are a family of small cytokines with a molecular weight of approximately 8-10 kD that act as chemoattractants to guide cell migration. Functional chemokine receptors CXCR1-7, CCR1-10, CX3CR1, and XCR1 are expressed by MSCs in vitro[29]. The migration of adipose-derived MSCs (AdMSCs) and bone marrowderived MSCs (BMSCs) is believed to occur in response to stimulation with various chemokines and cytokines. For example, the stromal-derived factor-1 (SDF-1) a/ CXCR4 interaction has been proven to affect the migration of MSCs in models of traumatic brain injury[30], spinal cord injury[31], myocardial infarction[32, 33], acute lung injury[34], and liver injury[14]. Akt, extracellular signal-regulated kinase (ERK), and p38 mitogen activated protein kinase (MAPK) signal transduction pathways may be involved in SDF-1-mediated migration of MSCs[33, 35]. In the rat model of bladder dysfunction, CXCL5/CXCR2 axis mediated chemotaxis of AdMSCs[36]. Monocyte chemoattractant protein-1 (MCP-1) /CCR2 signaling might promote the homing of MSCs to myocardium in dilated cardiomyopathy and a model of CNS inflammation[37, 38]. In addition, binding of fractalkine (CX3CL1) to CX3CR1 promotes chemotaxis of BMSCs towards ischemic brain lesions through Janus-Activated Kinase 2 (Jak2) signaling and cytoskeletal reorganization[39, 40].
However, certain studies have found that growth factors are more potent chemotactic agents than chemokines for human BMSCs and AdMSCs[26, 41]. Human adipose-derived MSCs (hAdMSCs) express transforming growth factor (TGF)-β receptor 2, tumor necrosis factor (TNF) receptor superfamily member 1A, platelet-derived growth factor (PDGF) receptor A, and PDGF receptor B at both protein and mRNA levels[26]. Moreover, PDGF-AB, TGF-β1, and TNF-α showed extraordinary chemoattractant activity in a transwell migration assay in vitro. A similar result was obtained for human bone marrow-derived MSCs (hBMSCs), as the growth factors PDGF-AB and insulin-like growth factor 1 (IGF-1) were demonstrated as the most potent chemoattractant, whereas the chemokines regulated on activation normal T cell expressed and secreted (RANTES) factor, macrophagederived chemokine (MDC), and SDF-1 had a limited effect on migration[25]. Besides, hBMSCs and umbilical cord blood derived MSCs (hUBMSCs) were strongly chemoattracted by hepatocyte growth factor (HGF) gradients in vitro, suggesting that HGF/Met might also play a role in the migration of MSCs[42].
2.2 Effects of adhesion molecules (integrins and selectins)Integrins are transmembrane receptors that function as the bridges for cell-cell and cell-extracellular matrix (ECM) interactions. The hMSCs were shown to express integrins alpha1, alpha2, alpha3, alpha5, alpha6, alphav, beta1, beta3, and beta4[43]. The integrin ligand vascular cell adhesion molecule (VCAM)-1 is involved in the adhesion of MSCs to endothelial cells. Fischer et al. found that most intravenously-infused BMSCs became trapped inside the lungs, which was prevented by a pretreatment with anti-CD49d (also called very late antigen-4, VLA4, or α4β1) antibody[44]. The adhesion molecules VLA-4/VCAM-1 expressed by MSCs/ endothelial cells were shown to play a role in the interactions between MSCs and lung tissue[45]. In addition, the VLA-4/VCAM-1 receptor axis has also been shown to mediate MSC migration in myocardial infarction[46]. Intravital microscopy performed on the mice with chronic experimental autoimmune encephalomyelitis and injected with AdMSCs revealed that a relevant subset of AdMSCs expresses activated alpha 4 integrins and adheres to inflamed brain venules, while bioluminescence imaging showed that alpha 4 integrins control AdMSCs accumulation in inflamed CNS[47].
Selectins are a large family of cell adhesion molecules (or CAMs). They can bind to sugar moieties and are therefore classified as a type of lectin. Blocking P-or E-selectins and CD44 knockout tests showed that both P-and E-selectins are involved in the homing of BMSCs to the infarct site after ischemic stroke, with CD44 acting as the critical ligand for selectin-mediated BMSC recruitment[48, 49]. However, it has also been shown that human MSCs express a CD44 glycoform bearing alpha-2, 3-sialyl modifications[50], but not E-selectin ligands, which may provide new insights for glycoengineering strategies as means to improve the migration of the MSCs.
2.3 Effects of enzymesBesides chemokines and adhesion molecules, enzymes secreted by certain invasive cells are essential for their migratory activity. During chemotaxis, MSCs secrete proteases that regulate transmigration and invasion of the endothelial basement membrane and degrade the extracellular matrix (ECM)[11]. MMPs are a family of zinc-dependent proteolytic enzymes that play an essential role in regulating the migratory activity of transplanted cells. The degradation products of ECM proteolysis by MMPs also mediate cell migration[51]. Several studies have shown that BMSCs highly express MMP-2, MT1-MMP (membrane-type MMP), tissuespecific inhibitor of metalloproteinase (TIMP)-1, and TIMP-2[52]. It was shown that MMP-2, MMP-9, MT1-MMP, and TIMP-2 could all promote the migration of MSCs, while TIMP-1 and TIMP-3 may have negative effects on cell motility[27]. In recent years, a new concept called biobridge has been proposed to elucidate the mechanism underlying the migration of endogenous progenitor cells to the lesion sites following MSCs therapy. Tajiri et al. used a model of traumatic brain injury and demonstrated that transplanted MSCs can form a biobridge connecting the area of injury to the neurogenic niche within the brain facilitating the repair by endogenous cells[53, 54]. Other studies hypothesized that, in a model of stroke[55, 56], a similar biobridge by injected MSCs is formed between the neurogenic niche and the stroke core and peri-infarct area. It has been found that the biobridge expresses high levels of extracellular MMPs, and that these may play an important role in the migration of endogenous neural stem cells[53].
3 In vitro determinants of MSC migrationThe migration of MSCs to the target tissue can be influenced by a number of factors related to the preparation of cells for transplantation, such as cell culture conditions, the origin of the MSCs, the conditioning medium, and the route of administration. For example, increased culture confluence has been shown to inhibit transendothelial MSC migration by increasing the production of a natural MMP inhibitor, TIMP-3[28]. Besides, increased number of cell passages can also affect the migration potential, as passaging the cells can modify the expression of receptors involved in the homing process[57]. MSCs of different origins may show differential motility due to differences in the expression profile of receptors and cell size[11]. Cheng et al. found that ASCs treated with high concentrations of glucose exhibited decreased cell migration in vitro[58]. Different administration routes may affect the efficacy of engraftment. Intravenous (Ⅳ) injection, intraperitoneal (IP) injection, intraarterial (IA) injection, and intracardiac (IC) injection are common methods of systematic administration. Although IV delivery is the least invasive one, most of the infused cells can become trapped in the liver, spleen, and lungs. Higher engraftment rates can be achieved by IC and IA delivery in certain models of myocardial infarction[59, 60] due to bypassing of filtration organs. In a model of acute stroke, IA stromal cell injection resulted in a highly variable efficacy of cerebral engraftment versus distant IV injection[61]. Although local infusion can be used to directly inject MSCs into the tissue of interest, this strategy is not clinically feasible because of its invasiveness (e.g., local injection into the heart or brain). Besides, locally administered cells often die before relieving the inflammatory response due to limitations in the diffusion of nutrients and oxygen[11].
4 Strategies to improve MSC migrationMany trials have been conducted in recent years to investigate potential strategies aiming to improve the migration and homing of MSCs to the sites of injury and inflammation. These existing strategies can be summarized as follows:
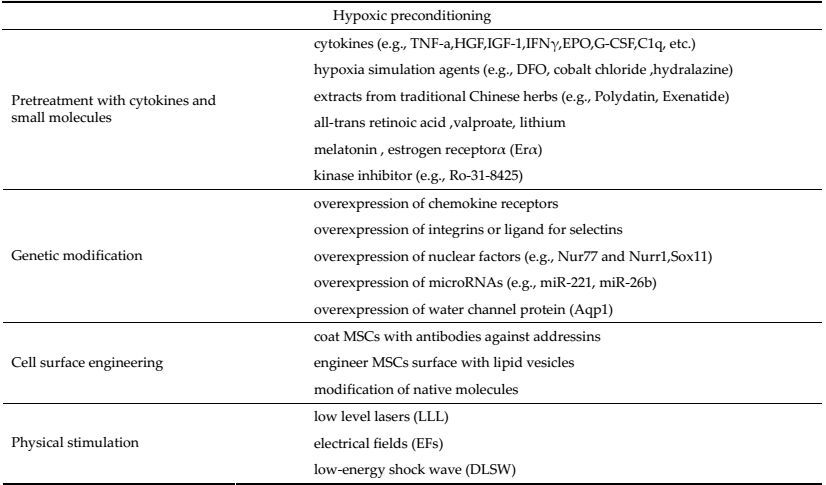
4.1 Culturing MSCs under hypoxic conditionsHypoxic preconditioning of MSCs has been proposed as a therapy for ischemic diseases[62]. Human MSCs exposed to hypoxic preconditioning have shown increased mobility and therapeutic potential compared to unconditioned control cells[63, 64]. The expression levels of CXCR4, CXCR7, CX3CR1, SDF-1a, and integrin αM[64-67] increased when MSCs were exposed to hypoxic conditions. Although the underlying mechanism is not completely clear, hypoxia-inducible factor-1 alpha (HIF-1α) is considered the master regulator of the adaptive response to hypoxia[68]. It has been suggested that enhanced activation of Ras homolog gene family, member A (RhoA), focal adhesion kinase (FAK), and Phosphatidylinositol-3-Kinase (PI3K)/Akt-HIF-1α-CXCR4/CXCR7 pathway mediates the increase in cell migration induced by hypoxic pretreatment[66-69]. These results suggest that short-term culture of MSCs under hypoxic conditions may provide novel means of enhancing their engraftment into a variety of tissues[70]. While hypoxic pretreatment is inexpensive and simple, the concentration of oxygen and the duration of hypoxia need to be further optimized to improve the therapeutic effect.
4.2 Pretreatment of MSCs with cytokines and small moleculesPrestimulating stromal cells with certain cytokines and small molecules in culture may facilitate their migration. The studies using an in vitro transwell assay showed that hAd-MSCs or hBMSCs prestimulated with TNF-a show increased migration compared with untreated cells[25, 26]. Prestimulation with a cocktail of five cytokines containing fms-like tyrosine kinase 3 (Flt-3) ligand, stem cell factor (SCF), interleukin (IL)-6, HGF, and IL-3 promotes the migration of FLK (+) MSCs to the bone marrow through up-regulation of both cell surface and intracellular CXCR4[71]. Other factors, such as IGF-1[72, 73], IL 1β[74], interferon γ (IFNγ)[4, 75], basic fibroblast growth factor (bFGF), PDGF[41], nitric oxide donor[76], or complement 1q (C1q)[77] have also been shown to improve MSC homing in different disease models possibly as a result of increased CXCR4 or MMP expression. Pretreating MSCs with a combination of hematopoietic growth factors erythropoietin (EPO) and granulocyte colony stimulating factor (G-CSF) can enhance their motility by increasing the MMP expression[78]. Tsai et al. showed that priming with valproate and/or lithium resulted into robust migration and homing of the MSCs towards the sites of ischemia in models of stroke and Huntington's disease[79, 80]. The underlying mechanism may involve an increase in CXCR4 expression by histone deacetylase (HDAC) inhibition, or elevation in MMP-9 levels through glycogen synthase kinase-3β inhibition. The treatment of MSCs with deferoxamine (DFO), cobalt chloride, or hydralazine can lead to increased surface expression of HIF-1a proteins, resulting in transcription of genes involved in cell migration[81-84]. In addition, pretreatment with all-trans retinoic acid[85], melatonin[86], estrogen receptorα (ERα)[87], and certain extracts from traditional Chinese herbs (i.e. Polydatin[88] and Exenatide[89]) can enhance the migration of MSCs as well[90]. Ro-31-8425, a kinase inhibitor identified as the most potent inducer of the homing integrin CD11a (integrin αL) surface expression, can significantly increase firm adhesion of MSCs to the endothelial ligand of CD11a, ICAM-1. In addition, it has been found that systemically infused MSCs pretreated with Ro-31-8425 display improved homing to the sites of inflammation and a stronger anti-inflammatory effect in a murine model of ear inflammation[90, 91]. Pretreating MSCs with cytokines and small molecules to control cell homing possesses multiple advantages over other bioengineering approaches. Cytokine-and small molecule-induced transient modifications of signaling pathways offer a potentially safe, simple, cost-effective, and highly scalable approach. Certain molecules, such as bFGF[92] and retinoic acid[93-95], can also promote differentiation of MSCs into neurons. While a few other molecules may change the differentiation and proliferation properties of the cell, such effects of most methods are thought to be negligible.
4.3 Genetic modification of MSCsSDF-1α/CXCR4 axis is one of the most important chemokine/receptor complexes regulating migration. Numerous experiments in which MSCs were modified by increasing the expression of the CXCR4 gene resulted in higher density of CXCR4 receptor, increased migration of MSCs toward SDF-1, and higher affinity to migrate to the sites of lesion[96]. In a rat model of cerebral ischemia, CXCR4 overexpression in the rMSCs promoted their mobilization and enhanced neuroprotective effects[97]. Apart from CXCR4, SDF-1 can also bind to CXCR7. Therefore, overexpressing CXCR7 receptor enhances migration as described above in cases of CXCR4 overexpression[98, 99]. In addition, overexpressing CXCR1, the receptor for IL-8, also improves migration[100]. Aquaporin-1 (Aqp1), the channel protein that transports water across the cell membrane, has been shown to interact with β-catenin to regulate cell migration. Therefore, MSCs overexpressing Aqp1 showed increased migration toward the sites of injury[101]. In addition, certain studies have shown that increased expression of nuclear factors, Nur77 and Nurr1[102, 103], and Sox11[104] or the overexpression of a4 subunit of the VLA-4 integrin[105] lead to enhanced cellular migration. In order to enhance the migration through vessel walls, MSCs were simultaneously transfected with mRNAs for P-selectin glycoprotein ligand-1 (PSGL-1) and Sialyl-Lewis (x) (SLeX). These cells produced functional ligands for P-selectins and E-selectins, resulting in significantly enhanced MSC homing to the inflamed spinal cord during experimental autoimmune encephalomyelitis (EAE) in mice[106]. Recently, microRNAs (miRNAs) have been described as regulators of MSC migration. The overexpression of miR-221[107, 108] or miR-26b[108] enhanced MSC migration through the activation of PI3K/Akt signaling. Genetic engineering, therefore, is a more directed method to promote migration, but this technique is difficult and expensive. In addition, the risk of tumorigenicity cannot be excluded.
4.4 Cell surface engineering of MSCsA new method that consists in coating MSCs with antibodies against addressins has been used to enhance the homing of these cells to the colon and increase the therapeutic efficacy in inflammatory bowel disease (IBD). It has been shown that vascular cell adhesion molecule antibody (Ab)-coated MSCs (Ab (VCAM-1) -MSCs) have the highest homing efficiency compared to untreated MSCs, Ab (isotype)-MSCs, and Ab (MAdCAM)-MSCs[109].
Since rolling is one of the first steps in the homing phase, an easy and generic approach to promote this process is to transiently engineer the cell surface using lipid vesicles. It was demonstrated that lipid vesicles could rapidly fuse with the cell membrane to introduce biotin moieties on the cell surface. Biotin can subsequently bind streptavidin and potentially any biotinylated homing ligand. The immobilization of sialyl Lewis X (SLeX) on MSCs to induce cell rolling on a P-selectin surface was significantly improved through a biotin-streptavidin bridge[110, 111]. While MSCs do not express E-selectin ligands, they highly express a CD44 glycoform bearing a-2, 3-sialyl modification. The native CD44 glycoform on MSCs can be converted to a ligand for E-selectin by treatment with a-1, 3-fucosyltransferase under specific enzymatic conditions without affecting cell viability or multipotency. Such modification of CD44 with an E-selectin binding motif increased specific homing of MSC to the BM[50, 65]. While cell surface engineering is a novel method to enhance migration in a controlled manner, the cost of this technique is elevated.
4.5 Physical stimulation of MSCs (light, electricity, and sound)Several studies have evaluated the effects of photobiomodulation (PBM) on the migration of MSCs. After irradiation of hMSCs in vitro by low level lasers (LLL), cell migration was accelerated via ERK1/2 and FAK pathway and upregulation of the expression of growth factors such as HGF and PDGF[112]. Electrical fields (EFs) were shown to induce rapid and directed migration of neural precursor cells[113, 114]. Griffin et al. found that BMSCs treated by EFs showed significantly increased expression of several migration-related molecules, including SDF-1/CXCR4, PDGF-BB-R, TGFbR1, IGF-1/IGF-1R, MMP-2, and MT1-MMP[115]. Besides, it has been found that defocused low-energy shock wave (DLSW) can activate MSCs. While treating rat adipose tissue-derived stromal cells (ADSCs) with DLSW did not alter the morphology and phenotype of ADSCs, the secretion, proliferation, and migration of ADSCs were promoted[36]. Thus, physical stimulation provides new means of improving the efficiency of cell therapy in the future.
5 Methods to promote the homing of MSCs in the treatment of CNS disordersMSCs have been used to treat various CNS disorders in preclinical and clinical studies, such as spinal cord and traumatic brain injury[116-118], ischemic stroke[31, 40, 56, 62, 69, 97, 119-123], intracranial hemorrhage (ICH)[124], Parkinson's disease[125, 126], multiple sclerosis[127], experimental autoimmune encephalomyelitis (EAE)[47, 128, 129], amyotrophic lateral sclerosis[130, 131], Alzheimer's disease[132], Huntington's disease[133-135], and so on. Potential repair mechanisms involve transdifferentiation to replace damaged neural cells and production of growth factors by MSCs. In addition, infused MSCs could modulate inflammation and stimulate endogenous neural stem cells to migrate to the target region[10, 106, 116, 136, 137]. The major obstacles for cell therapies are limited cell availability and engraftment, as well as reduced integration of grafted cells into the host tissue[138]. Apart from the abovementioned strategies to improve the migration of MSCs following cell therapy, several specific methods to enhance engraftment in the CNS have been tested.
The efficiency of cell-based therapies for neurodegenerative diseases largely depends on the mode of cell administration[139]. The IV (intravenous) route of administration has advantages of being more convenient, less invasive, and safer than intrathecal and intracerebral routes of administration[140], but the homing of the cells is limited. Administration of cells through intra-arterial injection during acute stroke resulted into a higher efficiency of cerebral engraftment[61]. In addition, it has been hypothesized that intranasally administered cells can bypass the bloodbrain barrier by migrating from the nasal mucosa through the cribriform plate along the olfactory neural pathway into the brain and cerebrospinal fluid (CSF). This method would minimize or eliminate the distribution of cellular grafts to peripheral organs and facilitate neurosurgical cell implantation[139]. Several studies have proven that intranasal transplantation promotes migration of infused MSCs to the sites of lesion[141, 142].
Blood brain barrier (BBB), consisting of cellular interactions between brain microvascular endothelial cells (BMECs), astrocytes, pericytes, and neurons[143], may restrict the number of stromal cells reaching injury sites. Whether systemically infused MSCs can actively migrate across BBB is still unclear. Rat MSCs were found to transmigrate across the BMEC monolayers in vitro through transient intercellular gaps between the BMECs[144]. However, the molecular mechanism underlying MSCs-BBB interaction is not known. It seems that MSCs undergo a leukocyte-like multistep homing cascade (i.e., rolling, adhesion, and transmigration) to engage with endothelial cells (ECs)[145]. Some studies indicate that MSCs may induce the disassembly of tight junctions in HBMEC through ERK1/2, PI3K, and ROCK signaling pathways[146, 147]. Since in vitro studies are performed using EC monolayers that do not fully recapitulate the in vivo BBB properties, further in vivo research is needed.
Crossing the BBB may be a prerequisite for MSCs to exert their therapeutic effects in treating neurological diseases[11, 145]. Thus, increasing the permeability of BBB appears as a feasible option to improve MSCs homing. Currently existing strategies for BBB opening are invasive, unspecific, and lack precise control over the site and timing of BBB opening. These disadvantages may limit their clinical translation. A novel approach has been described based on a combination of stromal cell delivery, heat-inducible gene expression, and mild heating with high-intensity focused ultrasound (HIFU) under MRI guidance to remotely permeabilize the BBB[148]. The principle of this method is to administrate stromal cells engineered to express pro-inflammatory factors that will permeabilize the BBB but only trigger their expression after heating with non-invasive imageguided HIFU. The aim of using this approach is to achieve a non-invasive way for BBB opening to restrict to the site where selected pro-inflammatory factors are secreted secondary to HIFU activation. This novel manner of BBB opening with unprecedented spatiotemporal precision may become an excellent method of improving the engraftment of stromal cells in the future.
6 Tracking migration and distribution of implanted MSCsMigration and precise location of infused MSCs can be tracked by several in vivo imaging methods including MRI, SPET/CT, and PET[11, 149-151]. In animal models, we can also track the cells by using techniques of fluorescent labeling[152-155], transduction of MSCs with reporter genes[59], taking advantage of species or sex mismatch by using specific genes as markers by fluorescent in situ hybridization or real-time PCR[11, 24]. In addition, nanoparticles have been used as a noninvasive technique of in vivo cellular tracking in cell therapy. Biocompatible superparamagnetic iron oxide nanoparticles (SPIONs) appear promising in investigation of homing, biodistribution, and fate of transplanted cells in preclinical and clinical studies because of their slower renal clearance, higher relaxation values, and low toxicity[156, 157]. Recently, a simple and well controlled stem cell imaging method was developed using imageable nanoparticles that bind bioorthogonal chemical receptors on the stromal cell surface[158]. These labeled stromal cells can be continuously tracked by noninvasive optical imaging over 15 days, and higher endocytic capacity can be achieved as compared with non-specific uptake of nanoparticles. Besides nanoparticles, a cellular barcoding tool was recently reported to enable clonal tracking in the hematopoietic system[159]. A small unique sequence or barcode was inserted into the genome of each individually hit cell through retroviral system. The progeny of all labeled cells can be traced as daughter cells carry the same barcode as mother cells[160]. This method might potentially provide a better clinical tool for studying the fate of transplanted MSCs. In addition, micro-optical sectioning tomography (MOST), a new imaging technique, is an attractive option for future studies of stromal cell migration. The MOST system consists of a microtome, light microscope, and image recorder, and it can perform imaging and sectioning simultaneously[161]. Combined with fluorescence imaging, the fluorescence micro-optical sectioning tomography (fMOST) has been used for whole mousebrain imaging to acquire sets of data relative to multiple types of neurons and projections. This technique has a single-neuron resolution and allows for precise tracing of long-range projections and accurately locating the nuclei[162-164]. We have used fMOST to image the neurons and microvessel density in a model of ICH[165]. Use of fMOST may provide novel means for exploring the migration and distribution of transplanted stromal cells in addition to study the formation of new neural circuits during brain inflammation and injury repair.
7 Summary and ProspectiveMSCs have been widely tested as a potential treatment for various diseases because of convenient isolation, less ethical issues, and low/negligible immunogenicity[11]. Low rates of cell homing, retention, and survival are the major limitations in current experimental and clinical studies for all different types of MSCs available[11, 15, 24, 138]. Many factors can influence the homing of MSCs (see Table 1). In this review, we have mainly discussed the potential roles of chemokines, growth factors, adhesion molecules, and MMPs in the process of migration. We then summarized the existing strategies to promote stromal cell migration (see Table 2). Finally, we discussed the migration of MSCs in the treatment of CNS diseases. Pretreatment with various small molecules and culturing cells under hypoxic environment has been shown to effectively improve MSC migration, and these methods are simple and cheap. However, these pretreatments may change certain properties of the original cells, and some of them show limited effects. Genetic modifications and cell surface engineering appear more efficient in improving cell homing. However, the difficulty and the cost of these techniques are elevated. In addition, unwanted genes may be introduced causing a series of safety problems. As for the physical stimulation of MSCs by light, sound, and electrical fields, this strategy may represent a new promising method to improve the efficiency of cell therapy in the future. When MSCs are used to treat CNS diseases, lower number of infused cells reaches the sites of lesion due to the existence of BBB. Adequate administration routes, such as intra-arterial and intranasal delivery of stromal cells can lead to improved cerebral engraftment via reducing cell entrapment by the liver, spleen, and lungs or by bypassing the blood-brain barrier. Recently, a new method based on a combination of stromal cell delivery, heat-inducible gene expression, and mild heating with high-intensity focused ultrasound (HIFU) was found to precisely and non-invasively permeabilize the BBB.
Our aim is to achieve increased homing and engraftment of MSC while using lower doses of transplanted cells, thereby decreasing the duration of cell culture, costs, availability, and the risk of transformations during culture expansion[160]. However, these trials should not compromise the viability, proliferation, and differentiation ability of the cells. A meta-analysis of preclinical results examining the intravenous route of cell delivery for the treatment of neurological disorders showed a good correlation between the dose of infused cells and the therapeutic effect. As such, correlation has not been shown between the therapeutic outcome and the number of cells engrafted within the lesioned brain area[166], different therapeutic mechanisms unrelated to the presence of infused cells within the injury site probably underlie the therapeutic effect. Recently, extracellular vesicles (EVs), membrane-surrounded structures released by most cell types, have been recognized as potent vehicles of intercellular communication that transmit biological signals between the cells. Paracrine effects of MSCs may be mediated by EVs[136]. The stromal cell derivedEVs do not replicate like the stromal cells, and they are less likely to become trapped inside the lungs after intravenous administration. In addition, given that exosomes, a subtype of secreted vesicles, are lipidbound nano-vesicles, they act as liposomes and can cross the blood-brain barrier[167]. Therefore, exosomes may represent an attractive therapeutic option for CNS disorders in the future.
Conflict of interestsAll contributing authors have no conflict of interests.
| [1] | Watt FM, Hogan BLM. Out of Eden:Stem cells and their niches. Science, 2000, 287(5457): 1427–1430. DOI:10.1126/science.287.5457.1427 |
| [2] | Neirinckx V, Coste C, Rogister B, Wislet-Gendebien S. Concise review:Adult mesenchymal stem cells, adult neural crest stem cells, and therapy of neurological pathologies:A state of play. Stem Cells Transl Med, 2013, 2(4): 284–296. DOI:10.5966/sctm.2012-0147 |
| [3] | Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue:Implications for cell-based therapies. Tissue Eng, 2001, 7(2): 211–228. DOI:10.1089/107632701300062859 |
| [4] | He XY, Jiang WK, Luo ZR, Qu TJ, Wang ZH, Liu NN, Zhang YQ, Cooper PR, He WX. IFN-γ regulates human dental pulp stem cells behavior via NF-κB and MAPK signaling. Sci Rep, 2017, 7: 40681. DOI:10.1038/srep40681 |
| [5] | Futami I, Ishijima M, Kaneko H, Tsuji K, IchikawaTomikawa N, Sadatsuki R, Muneta T, Arikawa-Hirasawa E, Sekiya I, Kaneko K. Isolation and characterization of multipotential mesenchymal cells from the mouse synovium. PLoS One, 2012, 7(9): e45517. DOI:10.1371/journal.pone.0045517 |
| [6] | Troyer DL, Weiss ML. Concise review:Wharton's JellyDerived cells are a primitive stromal cell population. Stem Cells, 2008, 26(3): 591–599. DOI:10.1634/stemcells.2007-0439 |
| [7] | Zhang XY, Wang YM, Gao YH, Liu XJ, Bai TT, Li MY, Li LS, Chi GF, Xu H, Liu FL, Liu JY, Li YL. Maintenance of high proliferation and multipotent potential of human hair follicle-derived mesenchymal stem cells by growth factors. Int J Mol Med, 2013, 31(4): 913–921. DOI:10.3892/ijmm.2013.1272 |
| [8] | Huang YS, Li IH, Chueh SH, Hueng DY, Tai MC, Liang CM, Lien SB, Sytwu HK, Ma KH. Mesenchymal stem cells from rat olfactory bulbs can differentiate into cells with cardiomyocyte characteristics. J Tissue Eng Regen Med, 2015, 9(12): E191–E201. DOI:10.1002/term.1684 |
| [9] | Ankrum J, Karp JM. Mesenchymal stem cell therapy:Two steps forward, one step back. Trends Mol Med, 2010, 16(5): 203–209. DOI:10.1016/j.molmed.2010.02.005 |
| [10] | Salem HK, Thiemermann C. Mesenchymal stromal cells:Current understanding and clinical status. Stem Cells, 2010, 28(3): 585–596. |
| [11] | Karp JM, Leng Teo GS. Mesenchymal stem cell homing:The devil is in the details. Cell Stem Cell, 2009, 4(3): 206–216. DOI:10.1016/j.stem.2009.02.001 |
| [12] | Liu S, Zhou JL, Zhang X, Liu Y, Chen J, Hu B, Song JL, Zhang YY. Strategies to optimize adult stem cell therapy for tissue regeneration. Int J Mol Sci, 2016, 17(6): 982. DOI:10.3390/ijms17060982 |
| [13] | Liu XF, Ye RD, Yan T, Yu SP, Wei L, Xu GL, Fan XY, Jiang YJ, Stetler RA, Liu G, Chen JL. Cell based therapies for ischemic stroke:From basic science to bedside. Prog Neurobiol, 2014, 115: 92–115. DOI:10.1016/j.pneurobio.2013.11.007 |
| [14] | Kuai XL, Li P, Zhang JFeng, Cao W, Yuan WY, Shao N, Guan CQ, Wang ZW. Stromal derived factor-1/CXCR4 axis involved in bone marrow mesenchymal stem cells recruitment to injured liver. Stem Cells Int, 2016, 2016: 8906945. |
| [15] | Chavakis E, Urbich C, Dimmeler S. Homing and engraftment of progenitor cells:A prerequisite for cell therapy. J Mol Cell Cardiol, 2008, 45(4): 514–522. DOI:10.1016/j.yjmcc.2008.01.004 |
| [16] | Tang YH, Zhang CF, Wang JX, Lin XJ, Zhang L, Yang Y, Wang YT, Zhang ZJ, Bulte JWM, Yang GY. MRI/SPECT/fluorescent tri-modal probe for evaluating the homing and therapeutic efficacy of transplanted mesenchymal stem cells in a rat ischemic stroke model. Adv Funct Mater, 2015, 25(7): 1024–1034. DOI:10.1002/adfm.v25.7 |
| [17] | Burst VR, Gillis M, Pütsch F, Herzog R, Fischer JH, Heid P, Müller-Ehmsen J, Schenk K, Fries JWU, Baldamus CA, Benzing T. Poor cell survival limits the beneficial impact of mesenchymal stem cell transplantation on acute kidney injury. Nephron Exp Nephrol, 2010, 114(3): e107–e116. DOI:10.1159/000262318 |
| [18] | Detante O, Moisan A, Dimastromatteo J, Richard MJ, Riou L, Grillon E, Barbier E, Desruet MD, De Fraipont F, Segebarth C, Jaillard A, Hommel M, Ghezzi C, Remy C. Intravenous administration of 99mTc-HMPAO-labeled human mesenchymal stem cells after stroke:In vivo imaging and biodistribution. Cell Transplant, 2009, 18(12): 1369–1379. DOI:10.3727/096368909X474230 |
| [19] | Chavakis E, Dimmeler S. Homing of progenitor cells to ischemic tissues. Antioxid Redox Signal, 2011, 15(4): 967–980. DOI:10.1089/ars.2010.3582 |
| [20] | Imhof BA, Aurrand-Lions M. Adhesion mechanisms regulating the migration of monocytes. Nat Rev Immunol, 2004, 4(6): 432–444. DOI:10.1038/nri1375 |
| [21] | Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation:The leukocyte adhesion cascade updated. Nat Rev Immunol, 2007, 7(9): 678–689. DOI:10.1038/nri2156 |
| [22] | Luster AD, Alon R, von Andrian UH. Immune cell migration in inflammation:Present and future therapeutic targets. Nat Immunol, 2005, 6(12): 1182–1190. DOI:10.1038/ni1275 |
| [23] | Rüster B, Göttig S, Ludwig RJ, Bistrian R, Müller S, Seifried E, Gille J, Henschler R. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood, 2006, 108(12): 3938–3944. DOI:10.1182/blood-2006-05-025098 |
| [24] | Belema-Bedada F, Uchida S, Martire A, Kostin S, Braun T. Efficient homing of multipotent adult mesenchymal stem cells depends on FROUNT-mediated clustering of CCR2. Cell Stem Cell, 2008, 2(6): 566–575. DOI:10.1016/j.stem.2008.03.003 |
| [25] | Ponte AL, Marais E, Gallay N, Langonné A, Delorme B, Hérault O, Charbord P, Domenech J. The in vitro migration capacity of human bone marrow mesenchymal stem cells:Comparison of chemokine and growth factor chemotactic activities. Stem Cells, 2007, 25(7): 1737–1745. DOI:10.1634/stemcells.2007-0054 |
| [26] | Baek SJ, Kang SK, Ra JC. In vitro migration capacity of human adipose tissue-derived mesenchymal stem cells reflects their expression of receptors for chemokines and growth factors. Exp Mol Med, 2011, 43(10): 596–603. DOI:10.3858/emm.2011.43.10.069 |
| [27] | Almalki SG, Agrawal DK. Effects of matrix metalloproteinases on the fate of mesenchymal stem cells. Stem Cell Res Ther, 2016, 7(1): 129. DOI:10.1186/s13287-016-0393-1 |
| [28] | De Becker A, Van Hummelen P, Bakkus M, Vande Broek I, De Wever J, De Waele M, Van Riet I. Migration of cultureexpanded human mesenchymal stem cells through bone marrow endothelium is regulated by matrix metalloproteinase-2 and tissue inhibitor of metalloproteinase-3. Haematologica, 2007, 92(4): 440–449. DOI:10.3324/haematol.10475 |
| [29] | De Becker A, Van Riet I. Homing and migration of mesenchymal stromal cells:How to improve the efficacy of cell therapy?. World J Stem Cells, 2016, 8(3): 73–87. DOI:10.4252/wjsc.v8.i3.73 |
| [30] | Ma JH, Liu N, Yi B, Zhang XC, Gao BB, Zhang YS, Xu RX, Li X, Dai YW. Transplanted hUCB-MSCs migrated to the damaged area by SDF-1/CXCR4 signaling to promote functional recovery after traumatic brain injury in rats. Neurol Res, 2015, 37(1): 50–56. DOI:10.1179/1743132814Y.0000000399 |
| [31] | Li J, Guo WC, Xiong M, Han H, Chen J, Mao D, Tang B, Yu HL, Zeng Y. Effect of SDF-1/CXCR4 axis on the migration of transplanted bone mesenchymal stem cells mobilized by erythropoietin toward lesion sites following spinal cord injury. Int J Mol Med, 2015, 36(5): 1205–1214. DOI:10.3892/ijmm.2015.2344 |
| [32] | Li L, Wu SZ, Liu Z, Zhuo ZX, Tan KB, Xia HM, Zhuo LS, Deng XJ, Gao YH, Xu YL. Ultrasound-targeted microbubble destruction improves the migration and homing of mesenchymal stem cells after myocardial infarction by upregulating SDF-1/CXCR4:A pilot study. Stem Cells Int, 2015, 2015: 691310. |
| [33] | Wiehe JM, Kaya Z, Homann JM, Wöhrle J, Vogt K, Nguyen T, Rottbauer W, Torzewski J, Fekete N, Rojewski M, Schrezenmeier H, Moepps B, Zimmermann O. GMP-adapted overexpression of CXCR4 in human mesenchymal stem cells for cardiac repair. Int J Cardiol, 2013, 167(5): 2073–2081. DOI:10.1016/j.ijcard.2012.05.065 |
| [34] | Yang JX, Zhang N, Wang HW, Gao P, Yang QP, Wen QP. CXCR4 receptor overexpression in mesenchymal stem cells facilitates treatment of acute lung injury in rats. J Biol Chem, 2015, 290(4): 1994–2006. DOI:10.1074/jbc.M114.605063 |
| [35] | Ryu CH, Park SA, Kim SM, Lim JY, Jeong CH, Jun JA, Oh JH, Park SH, Oh WI, Jeun SS. Migration of human umbilical cord blood mesenchymal stem cells mediated by stromal cell-derived factor-1/CXCR4 axis via Akt, ERK, and p38 signal transduction pathways. Biochem Biophys Res Commun, 2010, 398(1): 105–110. DOI:10.1016/j.bbrc.2010.06.043 |
| [36] | Xu LN, Zhao Y, Wang MW, Song W, Li B, Liu W, Jin XB, Zhang HY. Defocused low-energy shock wave activates adipose tissue-derived stem cells in vitro via multiple signaling pathways. Cytotherapy, 2016, 18(12): 1503–1514. DOI:10.1016/j.jcyt.2016.08.009 |
| [37] | Guo J, Zhang HF, Xiao JJ, Wu J, Ye Y, Li Z, Zou YZ, Li XL. Monocyte chemotactic protein-1 promotes the myocardial homing of mesenchymal stem cells in dilated cardiomyopathy. Int J Mol Sci, 2013, 14(4): 8164–8178. DOI:10.3390/ijms14048164 |
| [38] | Stamatovic SM, Keep RF, Kunkel SL, Andjelkovic AV. Potential role of MCP-1 in endothelial cell tight junction 'opening':Signaling via Rho and Rho kinase. J Cell Sci, 2003, 116(22): 4615–4628. DOI:10.1242/jcs.00755 |
| [39] | Zhang Y, Zheng J, Zhou ZJ, Zhou HD, Wang YJ, Gong ZL, Zhu J. Fractalkine promotes chemotaxis of bone marrowderived mesenchymal stem cells towards ischemic brain lesions through Jak2 signaling and cytoskeletal reorganization. FEBS J, 2015, 282(5): 891–903. DOI:10.1111/febs.13187 |
| [40] | Zhu J, Zhou ZJ, Liu Y, Zheng J. Fractalkine and CX3CR1 are involved in the migration of intravenously grafted human bone marrow stromal cells toward ischemic brain lesion in rats. Brain Res, 2009, 1287: 173–183. DOI:10.1016/j.brainres.2009.06.068 |
| [41] | Naaldijk Y, Johnson AA, Ishak S, Meisel HJR, Hohaus C, Stolzing A. Migrational changes of mesenchymal stem cells in response to cytokines, growth factors, hypoxia, and aging. Exp Cell Res, 2015, 338(1): 97–104. DOI:10.1016/j.yexcr.2015.08.019 |
| [42] | Son BR, Marquez-Curtis LA, Kucia M, Wysoczynski M, Turner AR, Ratajczak J, Ratajczak MZ, Janowska-Wieczorek A. Migration of bone marrow and cord blood mesenchymal stem cells in vitro is regulated by stromal-derived factor-1-CXCR4 and hepatocyte growth factor-c-met axes and involves matrix metalloproteinases. Stem Cells, 2006, 24(5): 1254–1264. DOI:10.1634/stemcells.2005-0271 |
| [43] | Majumdar MK, Keane-Moore M, Buyaner D, Hardy WB, Moorman MA, McIntosh KR, Mosca JD. Characterization and functionality of cell surface molecules on human mesenchymal stem cells. J Biomed Sci, 2003, 10(2): 228–241. DOI:10.1007/BF02256058 |
| [44] | Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue HS, Savitz SI, Laine GA, Cox Jr CS. Pulmonary passage is a major obstacle for intravenous stem cell delivery:The pulmonary first-pass effect. Stem Cells Dev, 2009, 18(5): 683–692. DOI:10.1089/scd.2008.0253 |
| [45] | Leibacher J, Henschler R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res Ther, 2016, 7(1): 7. DOI:10.1186/s13287-015-0271-2 |
| [46] | Segers VFM, Van Riet I, Andries LJ, Lemmens K, Demolder MJ, De Becker AJML, Kockx MM, De Keulenaer GW. Mesenchymal stem cell adhesion to cardiac microvascular endothelium:Activators and mechanisms. Am J Physiol Heart Circ Physiol, 2006, 290(4): H1370–H1377. |
| [47] | Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, Gini B, Bach SD, Martinello M, Bifari F, Galiè M, Turano E, Budui S, Sbarbati A, Krampera M, Bonetti B. Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells, 2009, 27(10): 2624–2635. DOI:10.1002/stem.v27:10 |
| [48] | Yilmaz G, Vital S, Yilmaz CE, Stokes KY, Alexander JS, Granger DN. Selectin-mediated recruitment of bone marrow stromal cells in the postischemic cerebral microvasculature. Stroke, 2011, 42(3): 806–811. DOI:10.1161/STROKEAHA.110.597088 |
| [49] | Lawton B, Boyette JR, Hu M, Lian TS. Selectin blockade decreases postischemic recruitment of bone marrow stromal cells. Laryngoscope, 2013, 123(12): 2993–2995. DOI:10.1002/lary.24217 |
| [50] | Sackstein R, Merzaban JS, Cain DW, Dagia NM, Spencer JA, Lin CP, Wohlgemuth R. Ex vivo glycan engineering of CD44 programs human multipotent mesenchymal stromal cell trafficking to bone. Nat Med, 2008, 14(2): 181–187. DOI:10.1038/nm1703 |
| [51] | Brauer PR. Mmps-role in cardiovascular development and disease. Front Biosci, 2006, 11(1): 447–478. DOI:10.2741/1810 |
| [52] | Ries C, Egea V, Karow M, Kolb H, Jochum M, Neth P. MMP-2, MT1-MMP, and TIMP-2 are essential for the invasive capacity of human mesenchymal stem cells:Differential regulation by inflammatory cytokines. Blood, 2007, 109(9): 4055–4063. DOI:10.1182/blood-2006-10-051060 |
| [53] | Tajiri N, Kaneko Y, Shinozuka K, Ishikawa H, Yankee E, McGrogan M, Case C, Borlongan CV. Stem cell recruitment of newly formed host cells via a successful seduction? Filling the gap between neurogenic niche and injured brain site. PLoS One, 2013, 8(9): e74857. DOI:10.1371/journal.pone.0074857 |
| [54] | Tajiri N, Duncan K, Antoine A, Pabon M, Acosta SA, de la Pena I, Hernadez-Ontiveros DG, Shinozuka K, Ishikawa H, Kaneko Y, Yankee E, McGrogan M, Case C, Borlongan CV. Stem cell-paved biobridge facilitates neural repair in traumatic brain injury. Front Syst Neurosci, 2014, 8: 116. |
| [55] | Liska MG, Crowley MG, Nguyen H, Borlongan CV. Biobridge concept in stem cell therapy for ischemic stroke. J Neurosurg Sci, 2017, 61(2): 173–179. |
| [56] | Duncan K, Gonzales-Portillo GS, Acosta SA, Kaneko Y, Borlongan CV, Tajiri N. Stem cell-paved biobridges facilitate stem transplant and host brain cell interactions for stroke therapy. Brain Res, 2015, 1623: 160–165. DOI:10.1016/j.brainres.2015.03.007 |
| [57] | Rombouts WJC, Ploemacher RE. Primary murine MSC show highly efficient homing to the bone marrow but lose homing ability following culture. Leukemia, 2003, 17(1): 160–170. DOI:10.1038/sj.leu.2402763 |
| [58] | Cheng NC, Hsieh TY, Lai HS, Young TH. High glucoseinduced reactive oxygen species generation promotes stemness in human adipose-derived stem cells. Cytotherapy, 2016, 18(3): 371–383. DOI:10.1016/j.jcyt.2015.11.012 |
| [59] | Barbash IM, Chouraqui P, Baron J, Feinberg MS, Etzion S, Tessone A, Miller L, Guetta E, Zipori D, Kedes LH, Kloner RA, Leor J. Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium:Feasibility, cell migration, and body distribution. Circulation, 2003, 108(7): 863–868. DOI:10.1161/01.CIR.0000084828.50310.6A |
| [60] | Beggs KJ, Lyubimov A, Borneman JN, Bartholomew A, Moseley A, Dodds R, Archambault MP, Smith AK, Mcintosh KR. Immunologic consequences of multiple, highdose administration of allogeneic mesenchymal stem cells to baboons. Cell Transplant, 2006, 15(8-9): 711–721. |
| [61] | Walczak P, Zhang J, Gilad AA, Kedziorek DA, RuizCabello J, Young RG, Pittenger MF, van Zijl PCM, Huang J, Bulte JWM. Dual-modality monitoring of targeted intraarterial delivery of mesenchymal stem cells after transient ischemia. Stroke, 2008, 39(5): 1569–1574. DOI:10.1161/STROKEAHA.107.502047 |
| [62] | Wei N, Yu SP, Gu XH, Taylor TM, Song D, Liu XF, Wei L. Delayed intranasal delivery of hypoxic-preconditioned bone marrow mesenchymal stem cells enhanced cell homing and therapeutic benefits after ischemic stroke in mice. Cell Transplant, 2013, 22(6): 977–991. DOI:10.3727/096368912X657251 |
| [63] | Rosová I, Dao M, Capoccia B, Link D, Nolta JA. Hypoxic preconditioning results in increased motility and improved therapeutic potential of human mesenchymal stem cells. Stem Cells, 2008, 26(8): 2173–2182. DOI:10.1634/stemcells.2007-1104 |
| [64] | Kubo M, Li TS, Kamota T, Ohshima M, Qin SL, Hamano K. Increased expression of CXCR4 and integrin αM in hypoxia-preconditioned cells contributes to improved cell retention and angiogenic potency. J Cell Physiol, 2009, 220(2): 508–514. DOI:10.1002/jcp.v220:2 |
| [65] | Naderi-Meshkin H, Bahrami AR, Bidkhori HR, Mirahmadi M, Ahmadiankia N. Strategies to improve homing of mesenchymal stem cells for greater efficacy in stem cell therapy. Cell Biol Int, 2015, 39(1): 23–34. DOI:10.1002/cbin.v39.1 |
| [66] | Liu HB, Xue WJ, Ge GQ, Luo XH, Li Y, Xiang HL, Ding XM, Tian PX, Tian XH. Hypoxic preconditioning advances CXCR4 and CXCR7 expression by activating HIF-1α in MSCs. Biochem Biophys Res Commun, 2010, 401(4): 509–515. DOI:10.1016/j.bbrc.2010.09.076 |
| [67] | Liu HB, Liu SB, Li Y, Wang XH, Xue WJ, Ge GQ, Luo XH. The role of SDF-1-CXCR4/CXCR7 axis in the therapeutic effects of hypoxia-preconditioned mesenchymal stem cells for renal ischemia/reperfusion injury. PLoS One, 2012, 7(4): e34608. DOI:10.1371/journal.pone.0034608 |
| [68] | Cerrada I, Ruiz-Saurí A, Carrero R, Trigueros C, Dorronsoro A, Sanchez-Puelles JM, Diez-Juan A, Montero JA, Sepúlveda P. Hypoxia-inducible factor 1 Alpha contributes to cardiac healing in mesenchymal stem cells-mediated cardiac repair. Stem Cells Dev, 2013, 22(3): 501–511. DOI:10.1089/scd.2012.0340 |
| [69] | Yu Q, Liu LZ, Lin J, Wang Y, Xuan XB, Guo Y, Hu SJ. SDF-1α/CXCR4 axis mediates the migration of mesenchymal stem cells to the hypoxic-ischemic brain lesion in a rat model. Cell J, 2015, 16(4): 440–447. |
| [70] | Choi JH, Lee YB, Jung J, Hwang SG, Oh IH, Kim GJ. Hypoxia inducible factor-1α regulates the migration of bone marrow mesenchymal stem cells via integrin α4. Stem Cells Int, 2016, 2016: 7932185. |
| [71] | Shi MX, Li J, Liao LM, Chen B, Li BZ, Chen L, Jia HR, Zhao RC. Regulation of CXCR4 expression in human mesenchymal stem cells by cytokine treatment:Role in homing efficiency in nod/scid mice. Haematologica, 2007, 92(7): 897–904. DOI:10.3324/haematol.10669 |
| [72] | Li YX, Yu XY, Lin SG, Li XH, Zhang SD, Song YH. Insulin-like growth factor 1 enhances the migratory capacity of mesenchymal stem cells. Biochem Biophys Res Commun, 2007, 356(3): 780–784. DOI:10.1016/j.bbrc.2007.03.049 |
| [73] | Xinaris C, Morigi M, Benedetti V, Imberti B, Fabricio AS, Squarcina E, Benigni A, Gagliardini E, Remuzzi G. A novel strategy to enhance mesenchymal stem cell migration capacity and promote tissue repair in an injury specific fashion. Cell Transplant, 2013, 22(3): 423–436. DOI:10.3727/096368912X653246 |
| [74] | Fan HY, Zhao GF, Liu L, Liu F, Gong W, Liu XQ, Yang L, Wang JJ, Hou YY. Pre-treatment with IL-1β enhances the efficacy of MSC transplantation in DSS-induced colitis. Cell Mol Immunol, 2012, 9(6): 473–481. DOI:10.1038/cmi.2012.40 |
| [75] | Duijvestein M, Wildenberg ME, Welling MM, Hennink S, Molendijk I, van Zuylen VL, Bosse T, Vos ACW, de Jonge-Muller ESM, Roelofs H, van der Weerd L, Verspaget HW, Fibbe WE, te Velde AA, van den Brink GR, Hommes DW. Pretreatment with interferon-γ enhances the therapeutic activity of mesenchymal stromal cells in animal models of colitis. Stem Cells, 2011, 29(10): 1549–1558. DOI:10.1002/stem.698 |
| [76] | Cui X, Chen JL, Zacharek A, Li Y, Roberts C, Kapke A, Savant-Bhonsale S, Chopp M. Nitric oxide donor upregulation of stromal cell-derived factor-1/chemokine (CXC motif) receptor 4 enhances bone marrow stromal cell migration into ischemic brain after stroke. Stem Cells, 2007, 25(11): 2777–2785. DOI:10.1634/stemcells.2007-0169 |
| [77] | Qiu YY, Marquez-Curtis LA, Janowska-Wieczorek A. Mesenchymal stromal cells derived from umbilical cord blood Migrate in response to complement C1q. Cytotherapy, 2012, 14(3): 285–295. DOI:10.3109/14653249.2011.651532 |
| [78] | Yu Q, Chen L, You Y, Zou C, Zhang YS, Liu QH, Cheng FJ. Erythropoietin combined with granulocyte colony-stimulating factor enhances MMP-2 expression in mesenchymal stem cells and promotes cell migration. Mol Med Rep, 2011, 4(1): 31–36. |
| [79] | Tsai LK, Wang ZF, Munasinghe J, Leng Y, Leeds P, Chuang DM. Mesenchymal stem cells primed with valproate and lithium robustly migrate to infarcted regions and facilitate recovery in a stroke model. Stroke, 2011, 42(10): 2932–2939. DOI:10.1161/STROKEAHA.110.612788 |
| [80] | Linares GR, Chiu CT, Scheuing L, Leng Y, Liao HM, Maric D, Chuang DM. Preconditioning mesenchymal stem cells with the mood stabilizers lithium and valproic acid enhances therapeutic efficacy in a mouse model of Huntington's disease. Exp Neurol, 2016, 281: 81–92. DOI:10.1016/j.expneurol.2016.04.003 |
| [81] | Dendorfer A, Heidbreder M, Hellwig-Bürgel T, Jöhren O, Qadri F, Dominiak P. Deferoxamine induces prolonged cardiac preconditioning via accumulation of oxygen radicals. Free Radic Biol Med, 2005, 38(1): 117–124. DOI:10.1016/j.freeradbiomed.2004.10.015 |
| [82] | Nouri F, Salehinejad P, Nematollahi-Mahani SN, Kamarul T, Zarrindast MR, Sharifi AM. Deferoxamine preconditioning of neural-like cells derived from human Wharton's Jelly mesenchymal stem cells as a strategy to promote their tolerance and therapeutic potential:An in vitro study. Cell Mol Neurobiol, 2016, 36(5): 689–700. DOI:10.1007/s10571-015-0249-8 |
| [83] | Knowles HJ, Tian YM, Mole DR, Harris AL. Novel mechanism of action for hydralazine:Induction of hypoxiainducible factor-1α, vascular endothelial growth factor, and angiogenesis by inhibition of prolyl hydroxylases. Circ Res, 2004, 95(2): 162–169. DOI:10.1161/01.RES.0000134924.89412.70 |
| [84] | Yu XF, Lu CL, Liu H, Rao SX, Cai JR, Liu SP, Kriegel AJ, Greene AS, Liang MY, Ding XQ. Hypoxic preconditioning with cobalt of bone marrow mesenchymal stem cells improves cell migration and enhances therapy for treatment of ischemic acute kidney injury. PLoS One, 2013, 8(5): e62703. DOI:10.1371/journal.pone.0062703 |
| [85] | Pourjafar M, Saidijam M, Mansouri K, Ghasemibasir H, Karimi Dermani F, Najafi R. All-trans retinoic acid preconditioning enhances proliferation, angiogenesis and migration of mesenchymal stem cell in vitro and enhances wound repair in vivo. Cell Prolif, 2017, 50(1): e12315. DOI:10.1111/cpr.2017.50.issue-1 |
| [86] | Mortezaee K, Pasbakhsh P, Ragerdi Kashani I, Sabbaghziarani F, Omidi A, Zendedel A, Ghasemi S, Dehpour AR. Melatonin pretreatment enhances the homing of bone marrowderived mesenchymal stem cells following transplantation in a rat model of liver fibrosis. Iran Biomed J, 2016, 20(4): 207–216. |
| [87] | Zhang W, Schmull S, Du M, Liu J, Lu Z, Zhu H, Xue S, Lian F. Estrogen receptor α and β in mouse:Adipose-derived stem cell proliferation, migration, and brown adipogenesis in vitro. Cell Physiol Biochem, 2016, 38(6): 2285–2299. DOI:10.1159/000445583 |
| [88] | Chen ZQ, Wei QS, Hong GJ, Chen D, Liang J, He W, Chen MH. Polydatin induces bone marrow stromal cells migration by activation of ERK1/2. Biomed Pharmacother, 2016, 82: 49–53. DOI:10.1016/j.biopha.2016.04.059 |
| [89] | Ma Q, Yang JJ, Zhou H, Zhang Y, Chen YD. Exenatide promotes chemotactic migration of adipose-derived stem cells through SDF-1/CXCR-4/Rho GTPase pathway. Journal of Southern Medical University, 2016, 36(8): 1034–1040. |
| [90] | Heinelt M, Karp JM, Levy O. Cell therapy-showing cells the way home. Oncotarget, 2015, 6(20): 17857–17858. DOI:10.18632/oncotarget.v6i20 |
| [91] | Levy O, Mortensen LJ, Boquet G, Tong ZX, Perrault C, Benhamou B, Zhang JD, Stratton T, Han E, Safaee H, Musabeyezu J, Yang ZJ, Multon MC, Rothblatt J, Deleuze JF, Lin CP, Karp JM. A small-molecule screen for enhanced homing of systemically infused cells. Cell Rep, 2015, 10(8): 1261–1268. DOI:10.1016/j.celrep.2015.01.057 |
| [92] | Jang S, Cho HH, Cho YB, Park JS, Jeong HS. Functional neural differentiation of human adipose tissue-derived stem cells using bFGF and forskolin. BMC Cell Biol, 2010, 11(1): 25. DOI:10.1186/1471-2121-11-25 |
| [93] | Abdullah RH, Yaseen NY, Salih SM, Al-Juboory AA, Hassan A, Al-Shammari AM. Induction of mice adult bone marrow mesenchymal stem cells into functional motor neuron-like cells. J Chem Neuroanat, 2016, 77: 129–142. DOI:10.1016/j.jchemneu.2016.07.003 |
| [94] | Marei HE, El-Gamal A, Althani A, Afifi N, Abd-Elmaksoud A, Farag A, Cenciarelli C, Caceci T, Hasan A. Cholinergic and dopaminergic neuronal differentiation of human adipose tissue derived mesenchymal stem cells. J Cell Physiol, in Press, DOI10. 1002/jcp. 25937. |
| [95] | Salehi H, Amirpour N, Niapour A, Razavi S. An overview of neural differentiation potential of human adipose derived stem cells. Stem Cell Rev Rep, 2016, 12(1): 26–41. DOI:10.1007/s12015-015-9631-7 |
| [96] | Nowakowski A, Walczak P, Lukomska B, Janowski M. Genetic engineering of mesenchymal stem cells to induce their migration and survival. Stem Cells Int, 2016, 2016: 4956063. |
| [97] | Yu XL, Chen DP, Zhang Y, Wu XL, Huang ZX, Zhou HT, Zhang YD, Zhang ZJ. Overexpression of CXCR4 in mesenchymal stem cells promotes migration, neuroprotection and angiogenesis in a rat model of stroke. J Neurol Sci, 2012, 316(1-2): 141–149. DOI:10.1016/j.jns.2012.01.001 |
| [98] | Wang YL, Fu W, Zhang SC, He XM, Liu ZA, Gao D, Xu TJ. CXCR-7 receptor promotes SDF-1α-induced migration of bone marrow mesenchymal stem cells in the transient cerebral ischemia/reperfusion rat hippocampus. Brain Res, 2014, 1575: 78–86. DOI:10.1016/j.brainres.2014.05.035 |
| [99] | Li H, Jiang YM, Jiang XX, Guo XM, Ning HM, Li YH, Liao L, Yao HY, Wang XY, Liu YL, Zhang Y, Chen H, Mao N. CCR7 guides migration of mesenchymal stem cell to secondary lymphoid organs:A novel approach to separate GvHD from GvL effect. Stem Cells, 2014, 32(7): 1890–1903. DOI:10.1002/stem.1656 |
| [100] | Kim SM, Kim DS, Jeong CH, Kim DH, Kim JH, Jeon HB, Kwon SJ, Jeun SS, Yang YS, Oh W, Chang JW. CXC chemokine receptor 1 enhances the ability of human umbilical cord blood-derived mesenchymal stem cells to migrate toward gliomas. Biochem Biophys Res Commun, 2011, 407(4): 741–746. DOI:10.1016/j.bbrc.2011.03.093 |
| [101] | Meng FB, Rui YF, Xu LL, Wan C, Jiang XH, Li G. Aqp1 enhances migration of bone marrow mesenchymal stem cells through regulation of FAK and β-catenin. Stem Cells Dev, 2014, 23(1): 66–75. DOI:10.1089/scd.2013.0185 |
| [102] | Maijenburg MW, Gilissen C, Melief SM, Kleijer M, Weijer K, ten Brinke A, Roelofs H, Van Tiel CM, Veltman JA, de Vries CJM, van der Schoot CE, Voermans C. Nuclear receptors Nur77 and Nurr1 modulate mesenchymal stromal cell migration. Stem Cells Dev, 2012, 21(2): 228–238. DOI:10.1089/scd.2011.0076 |
| [103] | Zhang X, Yan G, Diao Z, Sun H, Hu Y. NUR77 inhibits the expression of TIMP2 and increases the migration and invasion of HTR-8/SVneo cells induced by CYR61. Placenta, 2012, 33(7): 561–567. DOI:10.1016/j.placenta.2012.04.005 |
| [104] | Xu LL, Huang S, Hou YH, Liu Y, Ni M, Meng FB, Wang KX, Rui YF, Jiang XH, Li G. Sox11-modified mesenchymal stem cells (MSCs) accelerate bone fracture healing:Sox11 regulates differentiation and migration of MSCs. FASEB J, 2015, 29(4): 1143–1152. DOI:10.1096/fj.14-254169 |
| [105] | Kumar S, Ponnazhagan S. Bone homing of mesenchymal stem cells by ectopic α4 integrin expression. FASEB J, 2007, 21(14): 3917–3927. DOI:10.1096/fj.07-8275com |
| [106] | Liao WB, Pham V, Liu LN, Riazifar M, Pone EJ, Zhang SX, Ma FX, Lu MR, Walsh CM, Zhao WA. Mesenchymal stem cells engineered to express selectin ligands and IL-10 exert enhanced therapeutic efficacy in murine experimental autoimmune encephalomyelitis. Biomaterials, 2016, 77: 87–97. DOI:10.1016/j.biomaterials.2015.11.005 |
| [107] | Hu XY, Chen PP, Wu Y, Wang K, Xu YC, Chen H, Zhang L, Wu RR, Webster KA, Yu H, Zhu W, Wang JA. MiR-211/STAT5A signaling modulates migration of mesenchymal stem cells to improve its therapeutic efficacy. Stem Cells, 2016, 34(7): 1846–1858. DOI:10.1002/stem.2391 |
| [108] | Zhu AS, Kang NX, He LH, Li XY, Xu XJ, Zhang HX. MiR-221 and miR-26b regulate chemotactic migration of MSCs toward HGF through activation of Akt and FAK. J Cell Biochem, 2016, 117(6): 1370–1383. DOI:10.1002/jcb.v117.6 |
| [109] | Ko IK, Kim BG, Awadallah A, Mikulan J, Lin P, Letterio JJ, Dennis JE. Targeting improves MSC treatment of inflammatory bowel disease. Mol Ther, 2010, 18(7): 1365–1372. DOI:10.1038/mt.2010.54 |
| [110] | Sarkar D, Vemula PK, Zhao WA, Gupta A, Karnik R, Karp JM. Engineered mesenchymal stem cells with self-assembled vesicles for systemic cell targeting. Biomaterials, 2010, 31(19): 5266–5274. DOI:10.1016/j.biomaterials.2010.03.006 |
| [111] | Sarkar D, Vemula PK, Teo GSL, Spelke D, Karnik R, Wee LY, Karp JM. Chemical engineering of mesenchymal stem cells to induce a cell rolling response. Bioconjugate Chem, 2008, 19(11): 2105–2109. DOI:10.1021/bc800345q |
| [112] | Yin K, Zhu R, Wang S, Zhao RC. Low-level laser effect on proliferation, migration, and antiapoptosis of mesenchymal stem cells. Stem Cells Dev, 2017, 26(10): 762–775. DOI:10.1089/scd.2016.0332 |
| [113] | Babona-Pilipos R, Droujinine IA, Popovic MR, Morshead CM. Adult subependymal neural precursors, but not differentiated cells, undergo rapid cathodal migration in the presence of direct current electric fields. PLoS One, 2011, 6(8): e23808. DOI:10.1371/journal.pone.0023808 |
| [114] | Feng JF, Liu J, Zhang XZ, Zhang L, Jiang JY, Nolta J, Zhao M. Guided migration of neural stem cells derived from human embryonic stem cells by an electric field. Stem Cells, 2012, 30(2): 349–355. DOI:10.1002/stem.779 |
| [115] | Griffin M, Iqbal SA, Sebastian A, Colthurst J, Bayat A. Degenerate wave and capacitive coupling increase human MSC invasion and proliferation while reducing cytotoxicity in an in vitro wound healing model. PLoS One, 2011, 6(8): e23404. DOI:10.1371/journal.pone.0023404 |
| [116] | Zhang R, Liu Y, Yan K, Chen L, Chen XR, Li P, Chen FF, Jiang XD. Anti-inflammatory and immunomodulatory mechanisms of mesenchymal stem cell transplantation in experimental traumatic brain injury. J Neuroinflammation, 2013, 10(1): 871. DOI:10.1186/1742-2094-10-106 |
| [117] | Karamouzian S, Nematollahi-Mahani SN, Nakhaee N, Eskandary H. Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin Neurol Neurosurg, 2012, 114(7): 935–939. DOI:10.1016/j.clineuro.2012.02.003 |
| [118] | Alexanian AR, Fehlings MG, Zhang ZY, Maiman DJ. Transplanted neurally modified bone marrow-derived mesenchymal stem cells promote tissue protection and locomotor recovery in spinal cord injured rats. Neurorehabil Neural Repair, 2011, 25(9): 873–880. DOI:10.1177/1545968311416823 |
| [119] | Bang OY, Lee JS, Lee PH, Lee G. Autologous mesenchymal stem cell transplantation in stroke patients. Ann Neurol, 2005, 57(6): 874–882. DOI:10.1002/(ISSN)1531-8249 |
| [120] | Honmou O, Houkin K, Matsunaga T, Niitsu Y, Ishiai S, Onodera R, Waxman SG, Kocsis JD. Intravenous administration of auto serum-expanded autologous mesenchymal stem cells in stroke. Brain, 2011, 134(6): 1790–1807. DOI:10.1093/brain/awr063 |
| [121] | Sasaki Y, Honmou O. Bone marrow stem cell therapy for stroke. Nihon Rinsho, 2011, 69(12): 2203–2208. |
| [122] | Honmou O. Cell therapy for stroke. Rinsho Shink, 2013, 53(11): 1175–1176. DOI:10.5692/clinicalneurol.53.1175 |
| [123] | Zhang Y, Deng H, Pan C, Hu Y, Wu Q, Liu N, Tang ZP. Mesenchymal stromal cell therapy in ischemic stroke. J Neurorestoratol, 2016, 4: 79–83. DOI:10.2147/JN |
| [124] | Fatar M, Stroick M, Griebe M, Marwedel I, Kern S, Bieback K, Giesel FL, Zechmann C, Kreisel S, Vollmar F, Vollmar, Alonso A, Back W, Meairs S, Hennerici MG. Lipoaspirate-derived adult mesenchymal stem cells improve functional outcome during intracerebral hemorrhage by proliferation of endogenous progenitor cells:Stem cells in intracerebral hemorrhages. Neurosci Lett, 2008, 443(3): 174–178. DOI:10.1016/j.neulet.2008.07.077 |
| [125] | Camp DM, Loeffler DA, Farrah DM, Borneman JN, Lewitt PA. Cellular immune response to intrastriatally implanted allogeneic bone marrow stromal cells in a rat model of Parkinson's disease. J Neuroinflammation, 2009, 6(1): 17. DOI:10.1186/1742-2094-6-17 |
| [126] | Teixeira FG, Carvalho MM, Panchalingam KM, Rodrigues AJ, Mendes-Pinheiro B, Anjo S, Manadas B, Behie LA, Sousa N, Salgado AJ. Impact of the secretome of human mesenchymal stem cells on brain structure and animal behavior in a rat model of Parkinson's disease. Stem Cells Transl Med, 2017, 6(2): 634–646. DOI:10.5966/sctm.2016-0071 |
| [127] | Karussis D, Karageorgiou C, Vaknin-Dembinsky A, Gowda-Kurkalli B, Gomori JM, Kassis I, Bulte JW, Petrou P, Ben-Hur T, Abramsky O, Slavin S. Safety and immunological effects of mesenchymal stem cell transplantation in patients with multiple sclerosis and amyotrophic lateral sclerosis. Arch Neurol, 2010, 67(10): 1187–1194. |
| [128] | Agah EM, Parivar K, Joghataei MT. Therapeutic effect of transplanted human Wharton's Jelly stem cell-derived oligodendrocyte progenitor cells (hWJ-MSC-derived OPCs) in an animal model of multiple sclerosis. Mol Neurobiol, 2014, 49(2): 625–632. DOI:10.1007/s12035-013-8543-2 |
| [129] | Hou Y, Ryu CH, Park KY, Kim SM, Jeong CH, Jeun SS. Effective combination of human bone marrow mesenchymal stem cells and minocycline in experimental autoimmune encephalomyelitis mice. Stem Cell Res Ther, 2013, 4(4): 77. DOI:10.1186/scrt228 |
| [130] | Mazzini L, Ferrero I, Luparello V, Rustichelli D, Gunetti M, Mareschi K, Testa L, Stecco A, Tarletti R, Miglioretti M, Vercelli R, Oggioni GD, Carriero A, Cantello R, Monaco F, Fagioli F. Mesenchymal stem cell transplantation in amyotrophic lateral sclerosis:A phase I clinical trial. Exp Neurol, 2010, 223(1): 229–237. DOI:10.1016/j.expneurol.2009.08.007 |
| [131] | Vercelli A, Mereuta OM, Garbossa D, Muraca G, Mareschi K, Rustichelli D, Ferrero I, Mazzini L, Madon E, Fagioli F. Human mesenchymal stem cell transplantation extends survival, improves motor performance and decreases neuroinflammation in mouse model of amyotrophic lateral sclerosis. Neurobiol Dis, 2008, 31(3): 395–405. DOI:10.1016/j.nbd.2008.05.016 |
| [132] | Yun HM, Kim HS, Park KR, Shin JM, Kang AR, Lee KI, Song S, Kim YB, Han SB, Chung HM, Hong JT. Placentaderived mesenchymal stem cells improve memory dysfunction in an Aβ1-42-infused mouse model of Alzheimer's disease. Cell Death Dis, 2013, 4(12): e958. DOI:10.1038/cddis.2013.490 |
| [133] | Lin YT, Chern Y, Shen CKJ, Wen HL, Chang YC, Li H, Cheng TH, Hsieh-Li HM. Human mesenchymal stem cells prolong survival and ameliorate motor deficit through trophic support in Huntington's disease mouse models. PLoS One, 2011, 6(8): e22924. DOI:10.1371/journal.pone.0022924 |
| [134] | Snyder BR, Chiu AM, Prockop DJ, Chan AWS. Human multipotent stromal cells (MSCs) increase neurogenesis and decrease atrophy of the striatum in a transgenic mouse model for Huntington's disease. PLoS One, 2010, 5(2): e9347. DOI:10.1371/journal.pone.0009347 |
| [135] | Sadan O, Shemesh N, Barzilay R, Dadon-Nahum M, Blumenfeld-Katzir T, Assaf Y, Yeshurun M, Djaldetti R, Cohen Y, Melamed E, Offen D. Mesenchymal stem cells induced to secrete neurotrophic factors attenuate quinolinic acid toxicity:A potential therapy for Huntington's disease. Exp Neurol, 2012, 234(2): 417–427. DOI:10.1016/j.expneurol.2011.12.045 |
| [136] | Koniusz S, Andrzejewska A, Muraca M, Srivastava AK, Janowski M, Lukomska B. Extracellular vesicles in physiology, pathology, and therapy of the immune and central nervous system, with focus on extracellular vesicles derived from mesenchymal stem cells as therapeutic tools. Front Cell Neurosci, 2016, 10: 109. |
| [137] | Gervois P, Wolfs E, Ratajczak J, Dillen Y, Vangansewinkel T, Hilkens P, Bronckaers A, Lambrichts I, Struys T. Stem cell-based therapies for ischemic stroke:Preclinical results and the potential of imaging-assisted evaluation of donor cell fate and mechanisms of brain regeneration. Med Res Rev, 2016, 36(6): 1080–1126. DOI:10.1002/med.2016.36.issue-6 |
| [138] | Daviaud N, Garbayo E, Schiller PC, Perez-Pinzon M, Montero-Menei CN. Organotypic Cultures as tools for optimizing central nervous system cell therapies. Exp Neurol, 2013, 248: 429–440. DOI:10.1016/j.expneurol.2013.07.012 |
| [139] | Danielyan L, Schäfer R, von Ameln-Mayerhofer A, Buadze M, Geisler J, Klopfer T, Burkhardt U, Proksch B, Verleysdonk S, Ayturan M, Buniatian GH, Gleiter CH, Frey Ⅱ WH. Intranasal delivery of cells to the brain. Eur J Cell Biol, 2009, 88(6): 315–324. DOI:10.1016/j.ejcb.2009.02.001 |
| [140] | Lanza C, Morando S, Voci A, Canesi L, Principato MC, Serpero LD, Mancardi G, Uccelli A, Vergani L. Neuroprotective mesenchymal stem cells are endowed with a potent antioxidant effect in vivo. J Neurochem, 2009, 110(5): 1674–1684. DOI:10.1111/jnc.2009.110.issue-5 |
| [141] | Fransson M, Piras E, Wang H, Burman J, Duprez I, Harris RA, LeBlanc K, Magnusson PU, Brittebo E, Loskog ASI. Intranasal delivery of central nervous system-retargeted human mesenchymal stromal cells prolongs treatment efficacy of experimental autoimmune encephalomyelitis. Immunology, 2014, 142(3): 431–441. DOI:10.1111/imm.2014.142.issue-3 |
| [142] | Donega V, Nijboer CH, van Tilborg G, Dijkhuizen RM, Kavelaars A, Heijnen CJ. Intranasally administered mesenchymal stem cells promote a regenerative niche for repair of neonatal ischemic brain injury. Exp Neurol, 2014, 261: 53–64. DOI:10.1016/j.expneurol.2014.06.009 |
| [143] | Abbott NJ, Patabendige AAK, Dolman DEM, Yusof SR, Begley DJ. Structure and function of the blood-brain barrier. Neurobiol Dis, 2010, 37(1): 13–25. DOI:10.1016/j.nbd.2009.07.030 |
| [144] | Matsushita T, Kibayashi T, Katayama T, Yamashita Y, Suzuki S, Kawamata J, Honmou O, Minami M, Shimohama S. Mesenchymal stem cells transmigrate across brain microvascular endothelial cell monolayers through transiently formed inter-endothelial gaps. Neurosci Lett, 2011, 502(1): 41–45. DOI:10.1016/j.neulet.2011.07.021 |
| [145] | Liu LN, Eckert MA, Riazifar H, Kang DK, Agalliu D, Zhao WA. From blood to the brain:Can systemically transplanted mesenchymal stem cells cross the blood-brain barrier?. Stem Cells Int, 2013, 2013: 435093. |
| [146] | Lin MN, Shang DS, Sun W, Li B, Xu X, Fang WG, Zhao WD, Cao L, Chen YH. Involvement of PI3K and ROCK signaling pathways in migration of bone marrow-derived mesenchymal stem cells through human brain microvascular endothelial cell monolayers. Brain Res, 2013, 1513: 1–8. DOI:10.1016/j.brainres.2013.03.035 |
| [147] | Feng Y, Yu HM, Shang DS, Fang WG, He ZY, Chen YH. The involvement of CXCL11 in bone marrow-derived mesenchymal stem cell migration through human brain microvascular endothelial cells. Neurochem Res, 2014, 39(4): 700–706. DOI:10.1007/s11064-014-1257-7 |
| [148] | Xiong XB, Sun Y, Sattiraju A, Jung Y, Mintz A, Hayasaka S, Li KCP. Remote spatiotemporally controlled and biologically selective permeabilization of blood-brain barrier. J Control Release, 2015, 217: 113–120. DOI:10.1016/j.jconrel.2015.08.044 |
| [149] | Rahmi G, Pidial L, Silva AKA, Blondiaux E, Meresse B, Gazeau F, Autret G, Balvay D, Cuenod CA, Perretta S, Tavitian B, Wilhelm C, Cellier C, Clément O. Designing 3D mesenchymal stem cell sheets merging magnetic and fluorescent features:When cell sheet technology meets image-guided cell therapy. Theranostics, 2016, 6(5): 739–751. DOI:10.7150/thno.14064 |
| [150] | Yang BX, Brahmbhatt A, Nieves Torres E, Thielen B, McCall DL, Engel S, Bansal A, Pandey MK, Dietz AB, Leof EB, DeGrado TR, Mukhopadhyay D, Misra S. Tracking and therapeutic value of human adipose tissue-derived mesenchymal stem cell transplantation in reducing venous neointimal hyperplasia associated with arteriovenous fistula. Radiology, 2016, 279(2): 513–522. DOI:10.1148/radiol.2015150947 |
| [151] | Ribot EJ, Gaudet JM, Chen YH, Gilbert KM, Foster PJ. In vivo MR detection of fluorine-labeled human MSC using the bSSFP sequence. Int J Nanomedicine, 2014, 9: 1731–1739. |
| [152] | Dong SS, Liu N, Hu Y, Zhang P, Pan C, Zhang YP, Tang YX, Tang ZP. Transplantation of neural progenitor cells differentiated from adipose tissue-derived stem cells for treatment of sciatic nerve injury. Transl Neurosci Clin, 2016, 2(2): 108–119. DOI:10.18679/CN11-6030/R.2016.016 |
| [153] | Lee SH, Kim Y, Rhew D, Kim A, Jo KR, Yoon Y, Choi KU, Jung T, Kim WH, Kweon OK. Impact of local injection of brain-derived neurotrophic factor-expressing mesenchymal stromal cells (MSCs) combined with intravenous MSC delivery in a canine model of chronic spinal cord injury. Cytotherapy, 2017, 19(1): 75–87. DOI:10.1016/j.jcyt.2016.09.014 |
| [154] | Ezquer M, Ezquer F, Ricca M, Allers C, Conget P. Intravenous administration of multipotent stromal cells prevents the onset of non-alcoholic steatohepatitis in obese mice with metabolic syndrome. J Hepatol, 2011, 55(5): 1112–1120. DOI:10.1016/j.jhep.2011.02.020 |
| [155] | Li M, Zhang YX, Zhang Z, Zhou XY, Zuo XL, Cong YZ, Li YQ. Endomicroscopy will track injected mesenchymal stem cells in rat colitis models. Inflamm Bowel Dis, 2015, 21(9): 2068–2077. DOI:10.1097/MIB.0000000000000458 |
| [156] | Jasmin, de Souza GT, Andrade Louzada R, Rosado-deCastro PH, Mendez-Otero R, de Carvalho ACC. Tracking stem cells with superparamagnetic iron oxide nanoparticles:Perspectives and considerations. Int J Nanomedicine, 2017, 12: 779–793. DOI:10.2147/IJN |
| [157] | Mahmoudi M, Hosseinkhani H, Hosseinkhani M, Boutry S, Simchi A, Journeay WS, Subramani K, Laurent S. Magnetic resonance imaging tracking of stem cells in vivo using iron oxide nanoparticles as a tool for the advancement of clinical regenerative medicine. Chem Rev, 2011, 111(2): 253–280. DOI:10.1021/cr1001832 |
| [158] | Lee S, Yoon HI, Na JH, Jeon S, Lim S, Koo H, Han SS, Kang SW, Park SJ, Moon SH, Park JH, Cho YW, Kim BS, Kim SK, Lee T, Kim D, Lee S, Pomper MG, Kwon IC, Kim K. In vivo stem cell tracking with imageable nanoparticles that bind bioorthogonal chemical receptors on the stem cell surface. Biomaterials, 2017, 139: 12–29. DOI:10.1016/j.biomaterials.2017.05.050 |
| [159] | Gerrits A, Dykstra B, Kalmykowa OJ, Klauke K, Verovskaya E, Broekhuis MJC, de Haan G, Bystrykh LV. Cellular barcoding tool for clonal analysis in the hematopoietic system. Blood, 2010, 115(13): 2610–2618. DOI:10.1182/blood-2009-06-229757 |
| [160] | Maijenburg MW, van der Schoot CE, Voermans C. Mesenchymal stromal cell migration:Possibilities to improve cellular therapy. Stem Cells Dev, 2012, 21(1): 19–29. DOI:10.1089/scd.2011.0270 |
| [161] | Li AA, Gong H, Zhang B, Wang QD, Yan C, Wu JP, Liu Q, Zeng SQ, Luo QM. Micro-optical sectioning tomography to obtain a high-resolution atlas of the mouse brain. Science, 2010, 330(6009): 1404–1408. DOI:10.1126/science.1191776 |
| [162] | Gong H, Xu DL, Yuan J, Li XN, Guo CD, Peng J, Li YX, Schwarz LA, Li AA, Hu BH, Xiong BY, Sun QT, Zhang YL, Liu JP, Zhong QY, Xu TH, Zeng SQ, Luo QM. High-throughput dual-colour precision imaging for brain-wide connectome with cytoarchitectonic landmarks at the cellular level. Nat Commun, 2016, 7: 12142. DOI:10.1038/ncomms12142 |
| [163] | Gong H, Zeng SQ, Yan C, Lv XH, Yang ZQ, Xu TH, Feng Z, Ding WX, Qi XL, Li AA, Wu JP, Luo QM. Continuously tracing brain-wide long-distance axonal projections in mice at a one-micron voxel resolution. NeuroImage, 2013, 74: 87–98. DOI:10.1016/j.neuroimage.2013.02.005 |
| [164] | Osten P, Margrie TW. Mapping brain circuitry with a light microscope. Nat Methods, 2013, 10(6): 515–523. DOI:10.1038/nmeth.2477 |
| [165] | Pan C, Liu N, Zhang P, Wu Q, Deng H, Xu F, Lian LF, Liang QM, Hu Y, Zhu SQ, Tang ZP. EGB761 ameliorates neuronal apoptosis and promotes angiogenesis in experimental intracerebral hemorrhage via RSK1/GSK3β pathway. Mol Neurobiol, in Press, DOI10. 1007/s12035-016-0363-8. |
| [166] | Janowski M, Walczak P, Date I. Intravenous route of cell delivery for treatment of neurological disorders:A meta-analysis of preclinical results. Stem Cells Dev, 2010, 19(1): 5–16. DOI:10.1089/scd.2009.0271 |
| [167] | Konala VBR, Mamidi MK, Bhonde R, Das AK, Pochampally R, Pal R. The current landscape of the mesenchymal stromal cell secretome:A new paradigm for cell-free regeneration. Cytotherapy, 2016, 18(1): 13–24. DOI:10.1016/j.jcyt.2015.10.008 |