2. The First Hospital of Fuzhou Medical Association, Fuzhou 350009, China;
3. Beijing Tiantan Hospital, Capital Medical University, Beijing 100050, China;
4. College of Biological Science and Engineering, Fuzhou University, Fuzhou 350116, China
Research direction: 3D printing and biomedical materials research, implantation and interventional medical device industry transformation
He has led and participated in research teams as an investigator in 8 academic projects, including the National High-Technology Research and Development Program (863 program) and the National Natural Science Foundation of China. He possesses more than 25 patents. He has published over 30 papers in international journals and presented them at conferences, and more than 12 papers have been indexed by SCI.
Yuhua Jiang, E-mail:jxy_200321@163.com.
Cardiovascular and cerebrovascular diseases cause 17.5 million deaths worldwide every year. Stroke is the third leading cause of death in the United States; approximately 795, 000 Americans experience a new or recurrent stroke each year, with more than two-thirds of cases representing the first instance of the disease. Since 2009, the direct and indirect economic burdens due to stroke have been estimated at 6.89 billion US dollars[1]. China also has a high incidence of stroke—the second highest in the world.Epidemiological studies have suggested that there are 3 million cases of acute stroke in China each year and that approximately 1.8 million cases result in death. In addition, approximately 7 million people survive stroke annually, 75% of whom are left with a permanent disability (hemiplegia, aphasia, confusion, coma, incontinence, and so on), representing a significant burden on society. Stroke is a group of diseases characterized by local neurological deficits caused by disturbances in the brain's blood circulation. Hypertension and atherosclerosis are the main pathogenic factors of the disease. These factors are more common in old people[2]. In recent years, China's population has been ageing quite obviously. One predicted result of this is a doubling of the incidence of cardiovascular morbidity and mortality. Therefore, preventing the initial and recurrence onset of cerebrovascular disease is a hot topic in medical research.
The efficacy of traditional conservative treatment (pharmaceutical options) is very limited. With the development of interventional technology and materials, the techniques used in cerebrovascular interventional therapy are maturing. In general, the efficacy of intervention is superior to that of conservative and surgical treatment (carotid endarterectomy, intracranial and extracorporeal vascular bypass surgery), especially for patients with severe cerebrovascular stenosis. Intracranial stenting is particularly effective. Intracranial stenting is thought to be a life-saving treatment for patients with refractory acute cerebral stroke[3]. In addition, cerebrovascular intervention has a number of advantages, such as its minimally invasive procedure and rapid patient recovery. As a result, the technique has been adopted more widely in clinical practice. Intracranial stents have two main clinical applications: intracranial aneurysms and intracranial stenosis. This article focuses on the use of these stents for the treatment of intracranial stenosis.
2 CommentaryCerebrovascular interventional techniques mainly include carotid stenting, intracranial vascular stenting, and the embolization of brain aneurysms. Unlike in Caucasian people in Europe and the United States, atherosclerotic stenosis in the brains of Chinese people is mostly found in the cranial area. Intravascular stenting was first proposed Dotter and Judkins in 1964[4] and was initially used in the treatment of cardiovascular disease. The development of intracranial vascular stenting technology was slow due to the anatomical structure of the intracranial vascular system and the morphological peculiarities of the affected region. Intracranial stents refer to scaffolds placed in the area of the internal carotid artery, the internal branch of the vertebra, the vertebral artery, and the basal artery and its branches. In the past, only the most compliant coronary artery stents were applied intracranially. Many studies have reported that coronary stents cannot be used to treat neoplasia, intraoperative vascular rupture, or spasm occlusion. Therefore, the research and development of intracranial stents is important. In 2002, the US FDA approved the Boston Scientific Corp to develop the Neuroform Microdelivery system for the treatment of intracranial aortic aneurysms. This system is used in the United States and Europe. Subsequently, Guidan introduced the intracranial Neurolink stent system in the United States on October 3, 2002. This system is specifically designed to treat atherosclerotic stenosis in the cerebral arteries.
With the continued development of vascular stent technology and materials, current intracranial vascular stents have become an important method of treating intracranial vascular diseases (such as intracranial artery stenosis caused by ischemic stroke)[5]. These systems have become increasingly effective, safe, and widely recognized. Although stent angioplasty reduces the rate of restenosis in postoperative blood vessels, in-stent restenosis (ISR) may still occur in diseased vessels, leading to stroke. This significantly affects the clinical utility of these systems. Studies have shown that the metal stent restenosis rate is as high as 50%[6, 7]. ISR treatment is more difficult than the treatment of simple postoperative (postoperative or perioperative) restenosis and has become a key factor affecting the efficacy of stent implantation[8]. The cause of ISR is different from that of postoperative restenosis, mainly due to the inflammatory response after stent implantation and hyperplasia of smooth muscle cells (SMC) during the self-repair process and migration to the intima (Figure 1).
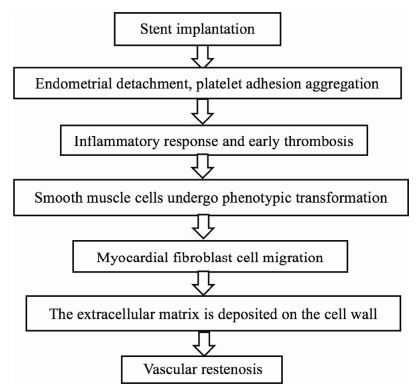
|
| Figure 1 Schema of interventional treatment of vascular restenosis. |
The phenomenon of restenosis after stent implantation has seriously affected the clinical application value of these stents in the context of interventional therapy. Since the discovery of the risks of ISR, methods for the prevention and treatment of ISR have been researched and a variety of new technologies have been developed, including systemic drug therapy, mechanical therapy, topical radiotherapy, and drugeluting stents (DESs). DES technology is the most successful of these new applications. Research by Vajda et al. demonstrated that angioplasty and DESs are safe for the treatment of intracranial atherosclerotic disease and effective in reducing the incidence of ISR[9].
2.1 Classification of intracranial vascular stentsVascular stents are classified differently according to the classification criteria used. Depending on the design of the stent, stents can be divided into braided wire brackets, bending wire stents, winding brackets, laser cutting plate brackets, laser cutting tubular brackets, and ring brackets. According to surface characteristics, they can be divided into bare stents, drug-coated stents, DESs, radioactive stents, surface coated stents, and artificial vascular wall scaffolds. According the degradation characteristics of the stent, they can be divided into permanent stents and biodegradable stents. According to the mechanism of action, they can be divided into self-expansion stents, shape memory expansion stents, and balloon dilatation stents. According to the material used to make the scaffold, they can be divided into 316L stainless steel brackets, titanium nickel alloy stents, cobalt-based alloy stents, metal tantalum stents, magnesium alloy stents, and polymer stents. Cerebrovascular stents are common in clinical practice and include balloon dilatation stents, new balloon dilatation stents, selfexpanding stents, braided stent, and DESs[10].
Balloon expansionary stents, such as AVE, BX, S670, Palmaz, and 316L stainless steel tube laser carved stents, have good compliance, stable ability throughout the disease, and strong support, among other beneficial characteristics. However, they can be easily deformed after being subjected to external forces, particularly if they are composed of a cobalt-chromium alloy.
A new type of balloon dilatation stent can reduce the rate of restenosis. However, these stents are more expensive and suitable for intracranial blood vessels with smaller diameters.
Self-expanding stents, such as mesh Ni-Ti alloy stents, provide a high degree of supportive force, adaptability, adaption to angular lesions, adhesion, resistance to the formation of a dead space, and shortening after the release of the stent. However, there is a danger of expansion after continuous point ruptures.
Braided stents provide good flexibility. They are fully released when recycled. The mesh diameter is small and they are more suitable for the treatment of ulcerous lesions. Various degrees of shortening occur after release and they can be difficult to accurately locate. However, the radial support provided by these stents is strong, carotid artery stretching is easy, and adherence is poor.
Drug-loaded stents can be divided into many types according to the composition or types of drugs delivered: (1) anti-thrombotic heparin-coated scaffolds; (2) anti-inflammatory dexamethasone-coated scaffolds; and (3) anti-proliferative stents: (a) paclitaxel-coated scaffolds and (b) rapamycin-coated scaffolds (Figure 2) : rapamycin takes the form of a large circle with vinegar immunosuppressive agents that can effectively prevent cytokine metastases and inhibit tissue proliferation. This molecule inhibits the cell cycle to prevent SMC proliferation, primarily by inhibiting cell division and changes. It can prevent SMC proliferation and intimal thickening. The final types are (4) gene drug-coated scaffolds and (5) nano-drugcoated stents.
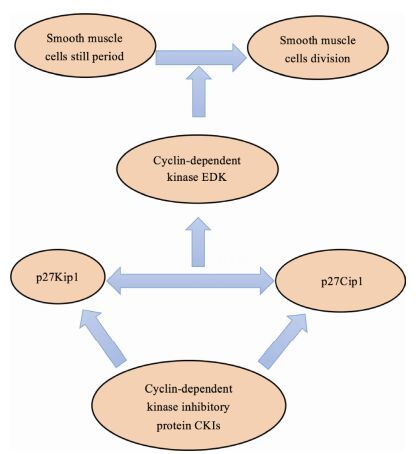
|
| Figure 2 Rapamycin inhibits the proliferation of vascular smooth muscle cells. |
The materials used for the preparation of intracranial intravascular stents include 316L stainless steel, nickeltitanium alloy, cobalt-base alloy, magnesium alloy, tantalum, biodegradable materials, and so on.
Cobalt-based alloys have a density of 8.3-9.15 g/cm3 and a melting point of 1, 290-1, 425 ℃. These allows contain major ingredients such as Co, Cr, Ni, Mo, Mn, and so on. At present, the main cobalt-based alloys used for producing intravascular stents are L605, Elgiloy, MP305, and so on. Cobalt-based alloys have been used as implant materials in clinical surgery since 1937. Laminated self-expansion stents and newtype Walltent self-expansion stents are made from these cobalt-based alloys and used for the treatment of artery hemangioma and peripheral artery stenosis, respectively, as intravascular stents. Benefiting from its superiority over traditional 316L stainless steel in some key areas, cobalt-based alloys are now considered better candidates for the production of drug-eluting/coated stents.
Additionally, as an iron-based corrosion-resistant alloy, the main components of 316L stainless steel are Fe, Cr, Ni, Mo, Mn, and so on. Its metallographic structure is that of a classical austenite with a density of 7.5-8.0 g/cm3 and melting point of 1, 380-1, 420 ℃. It is therefore mostly used to prepare balloon dilatation intravascular stents. As one of the initially developed biomedical alloys, 316L stainless steel provides good mechanical performance and has seen wide application due to its low cost and traceable characteristics. However, the poor corrosion resistance and biocompatibility of intravascular stents made from 316L stainless steel, in addition to toxicity caused by Cr and Ni, have limited the clinical application of stainless steel in this medical context.
Tantalum is a rare metal with an atomic coefficient of 73, density of 16.65 g/cm3, and melting point of 2, 996 ℃. Tantalum has some merits as a material for intravascular stents: (1) high density, giving it good resistance to radiation and facilitating location and release of the stent; (2) non-magnetic properties, giving it high compatibility with nuclear magnetic resonance (NMR), thus facilitating follow-up; (3) superior corrosion resistance and biocompatibility; (4) a negatively charged oxide layer on its surface, preventing interaction with and adhesion of blood platelets, which suppresses the formation of thrombi, thus accelerating endothelialization of the blood vessel. However, the weak radial supportive strength of stents woven from tantalum wire reduces the employment of tantalum in clinical practice.
Nickel-titanium alloy has good shape-memory characteristics and excellent mechanical performance, while its corrosion resistance is relatively poor, leading to commonly reported toxic reactions.
Magnesium alloy has a high degree of biodegradability, mechanical quality, and biocompatibility, but can easily be corroded and shows poor plasticity and development effects, making it hard to trace.
Biodegradable vascular stents are usually made from polymer materials[11], of which polylactic acid, polylactide, and polycaprolactone have been approved by the US FDA as implantable human degradable materials. Degradable polymer materials also have good biocompatibility and superior mechanical and physical properties[12, 13].
2 Structure and principleIntracranial vascular stents are mesh-like capillaries approximately 0.8-3 mm in diameter, 0.03-0.6 mm in thickness, and 8-20 mm in length with a 20-150 μmwide lattice beam[14].
The three-dimensional structure of current stents can be described as follows. The most basic component of the bracket is the bracket metal wire (or bracket support tendons). The other structures are also made of the same bracket metal wire.
The stent hole is a small structure that regularly repeats on the scaffold and is the most basic structural unit of the stent. The structure of the hole is divided into the open type and closed type. The open-hole structure is more complicated than the closed-hole structure and the geometric mechanism of the closedhole type is simple.
The stent ring and the crown are composed of a set of holes that can form a complete stent segment. Many stent segments are connected to each other to form a complete stent. The complexity of the brackets, holes, rings, and scaffolds ranges from low to high. Together, these components constitute a complex three-dimensional bracket structure.
Most commonly used balloon dilatation stents (Figure 3) are made of 316L stainless steel and cobaltbased alloys. The bracket unit section generally employs a sine wave design. Each bracket section has a number of connecting rods. The connecting rod can be formed into various shapes according to the requirements of the stent. The stent is pressed using a special process on a balloon of approximately 11.5 mm in diameter. Since the stent is plastically deformed, it is tightly pressed against the surface of the balloon. The stent will not fall off during the surgical procedure.
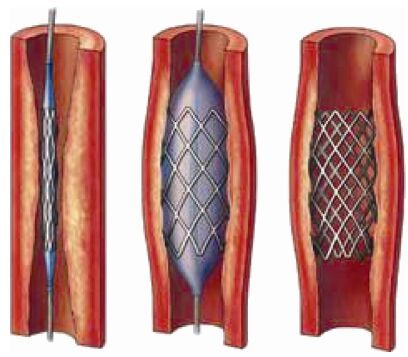
|
| Figure 3 Balloon dilatation stent implantation for the treatment of stenosis. |
Drug-coated stents for the prevention of ISR are composed of a stent, stroma, and drug component (Figure 4). The main body is the stent, which is mostly made from stainless steel or an alloy in addition to biological materials. The matrix is prepared from biodegradable polymers, which are highly biocompatible materials designed to reduce inflammatory responses. In the case of biodegradable polymer materials, the release rate of drugs is controlled by the solubility of the drug and the degradation rate of the polymer carrier. If the degradation rate is much lower than the diffusion rate, diffusion becomes a stronger controlling factor of drug release. Conversely, if the drug is difficult to move within the carrier, degradation becomes a primary controlling factor of release. In drug-controlled release systems with a non-biodegradable polymer as the carrier, the solubility of the drug is the main factor controlling its release rate. The slow release of drugs described above leads to the inhibition of vascular SMC proliferation, halting the mechanism of restenosis.
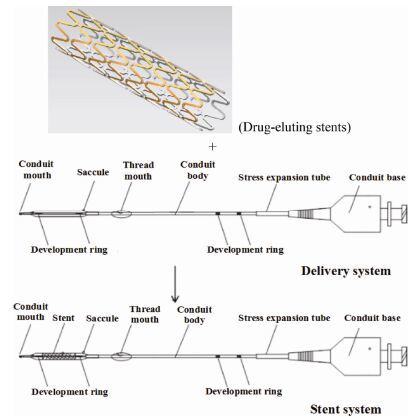
|
| Figure 4 Cerebrovascular drug-eluting stent. |
(1) Laser cutting technology[15]
Because the support bar is small and the feature size is < 110 μm, it is difficult to produce stents using traditional machining methods. Laser cutting has a number of advantages, such as a smaller slit width, good incision parallelism, minimal surface roughness, and high dimensional accuracy. In addition, deformation of the stent is limited and the heat-affected zone is small. There are no issues related to mechanical stress or surface damage, while it is possible to produce the complex shapes needed to manufacture thin-walled metal tubes from a complex network of components.
The quality of the cutting surface is important and determined by a number of characteristics. The width of the incision, roughness of the incision area, width of the heat-affected zone, ripple of the incision section, and cut surface or lower surface are all important and determined by the material, shape, size, surface state, and so on. The performance of the processing system itself is another key issue in the preparation of DESs (accuracy, wavelength, output power, beam mode, beam shape, diameter, divergence angle, focal length, focal position, depth of focus, spot diameter, and so on). The processing parameters are also key factors, and include material feed rate, accuracy, auxiliary gas parameters, nozzle shape, hole size, laser cutting path setting, and so on.
(2) Drug combination process
The two commonly used drug-coated scaffold preparation methods are dip coating and spraying[16].
(A) Dip coating method
This is the main method used in the preparation of DESs. The bare stainless steel stent is placed in a solution containing the carrier polymer and drug. The drug is adsorbed to the stent via the binding force between the polymer carrier and the surface of the metal stent. Heparin drug-coated scaffolds are prepared using dip coating. Modified polymethacrylic resin (obtained by copolymerizing methacrylic acid methyl ester with methacrylic acid and butyl methacrylate) is prepared by a solution polymerization method and used as a drug carrier. The stainless steel stent is then dipped in specific concentrations of carrier polymer and drug blends. Then, 30 min after removing the stent from the solution, the stent is placed in a vacuum drying oven to remove the solvent and leave the drug-coated surface exposed. The properties of the copolymers are then analyzed using infrared spectroscopy and NMR spectroscopy. The physical properties, bioactivity, and drug-releasing properties of the composites are then evaluated. The prepared coatings have good biostability. Methyl methacrylate is then added to improve the physical properties of the polymer, especially when the goal is to produce stents with the following properties: metal matrix binding force, slow drug release, and release cycle of >15 d. The methylated acrylic resin coating improves biological stability, physical properties, and drug release properties. The dip coating process is relatively simple. The coating is firm and the process is also easy to adjust, making it practical. However, more complex stents are prone to material aggregation or condensation. Material aggregation or condensation can be reduced, making it possible to re-modify the coating. However, this slows down the process, requires air and environmental control, and increases material consumption.
(B) Spraying
In this method, the appropriate quantities of drugs and polymers are mixed with solvent tetrahydrofuran after ultrasonic dissolution to form a drug-containing solution for coating the stents.
The use of an ultrasonic atomization spray system is outlined in Figure 5. The control panel on the injector can be used to set the flow rate of the spray solution and the spray volume. The computer controls the switch status of the injector. The sprayed liquid is atomized using an ultrasonic atomization generator. The liquid is then sprayed onto the inner and outer surfaces of the stent using computer-aided focusing and the speed and mode of movement of the stent are controlled by a computer. The thickness of the spray coating and the amount of spray applied can be controlled by adjusting the number of spray cycles.
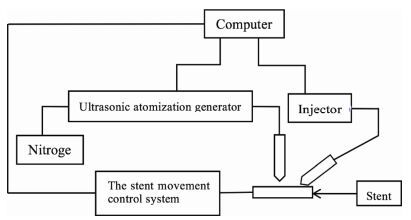
|
| Figure 5 Principle of ultrasonic atomization spraying |
There are many factors that affect the quality of the coating, including the flow rate of the sample, flux, the power of the ultrasonic generator, and the nature of the solution. The flow rate and the concentration of the solution have the greatest effects on coating quality. If the spray velocity is too high, the coating can be uneven and it is easy to produce cross-links due to the accumulation of coating and reduced flow rate. This can lead to incomplete coating coverage and inefficient spraying. The concentration of the solution affects stickiness and fluidity, influencing the volatilization of the solvent and the uniformity of the coating.
(C) Assembly of drug-loaded nanoparticles on the stent using dielectric swimming
The self-assembly of drug-loaded nanoparticles on stents is achieved by the application of a micro-force, such as dielectric activity, capillary action, and hydrophobicity. First, by controlling the size and direction of the electrophoretic force, the movement of the nanoparticles to the surface of the stent is controlled so that the nanoparticles are brought into contact with the surface of the scaffold. Then, the nanoparticles are affected by the capillary force and hydrophobic force and self-assembly is completed.
The following are performance indicators of intracranial vascular stents:
The ideal vascular stent should meet the requirements of the evaluation index of the vascular stent. The specific performance should have the following characteristics[17-20]:
1. Good biocompatibility. Biocompatibility refers to the tolerance of the body's immune system to foreign objects, such as medical equipment. This is the most basic and important performance indicator. It is important that no strong immune reaction is induced so that harmful effects are minimized.
2. Good traceability. The ideal vascular stent should have good X-ray impermeability. The stent should have high visibility on X-ray and it should be easy to facilitate the positioning of the vascular stent, release the stent, and perform follow-up observation. The ability to detect the stent on X-ray is determined not just by impermeability but also the density of the material and the design of the stent itself (wall thickness of the bracket). If a material with poor traceability is used, the position of the stent can usually be determined using additional markings.
3. Good adaptability. The compliance of vascular stents is a reflection of both flexibility and rigidity. Flexible vascular stents are easy to push through tortuous blood vessels to the lesion site but have reduced stability. Rigid vascular stents have good geometric stability but transportation is more difficult. Therefore, the ideal vascular stent should have good compliance to facilitate both transport and fixation.
4. Good expansion. The expansion rate of the ideal vascular stent (the diameter of the stent after compression and the fully expanded diameter) should be >6. Stents with expansion rates < 4 should not be used to ensure that the stent can be delivered to vessels with a small diameter.
5. Good radial flexibility and supportive force. The flexibility of the stent makes it easier to force the stent through tortuous blood vessels and prevents play after deployment in blood vessels that are straight or damaged. Radial supportive force refers to the resistance of the stent to lateral pressure. This determines whether the stent attaches to the vessel wall and adequately prevents the elastic retraction of the wall after deployment.
6. Low axial shortening rate. After the release of vascular stents, the length should not be changed or shortened. This ensures optimal coverage of vascular lesions. Shortening is not conducive to implantation, so the appropriate length and diameter of the stent should be determined preoperatively.
7. Good blood compatibility. Vascular stents are directly in contact with blood, so the stent design must take into account several factors, such as shape, surface roughness, surface tension, surface hydrophilic surface energy, surface charge, and so on. Ideally, the stent should not cause thrombosis or coagulation reactions. The stent should also not cause vascular intimal hyperplasia and vascular wall inflammation or affect the normal blood flood within the vessel.
8. Metal coverage. The degree of coverage of the surface of the stent is an important factor affecting the stent connection within the vessel. The implanted scaffold should completely cover the lesion area. The ability to completely cover the lesion, in addition to the length and visibility of the stent, depends on the surface area of the stent. The metal coverage area of the stent should be moderate. The metal coverage area should be 10%-20%; a smaller metal area than the stent is not conducive to the coverage of the lesion, making the patient prone to plaque shedding, while a larger area of metal coverage leads to thrombosis and the stimulation of blood vessels.
9. Elastic recovery. Elastic radial recovery occurs after removal of the stent expansion balloon and is a necessary stent feature because a small rebound rate can reduce the maximum diameter that must be expanded in order to obtain the final release diameter. The rebound of the stent and the smaller stent need to be expanded to a larger diameter to obtain the final release diameter. In practice, excessive dilation of the implanted scaffold may result in tissue damage that reduces the short and long-term efficacy of the stent. The rebound rate of the stent depends on the structure of the stent, the degree of dilation of the stent, and the deformability of the stent material. The resilience of a tube-type stainless steel support is usually 3%-5%, while the recovery rate of modern cobalt-chromium alloy supports is usually 5%.
(D) Problems and Prospects of Intracranial Vascular Stent
Intravascular stenting has become the most effective treatments for ischemic cerebrovascular disease caused by arterial stenosis. It is generally considered that in cases in which the arterial stenosis occludes ≥70% of the vessel, it is necessary to use intravascular stenting regardless of symptoms to eliminate stenosis and prevent stroke onset.
The complications of stent placement include puncture site injuries, carotid sinus reflex, thrombosis, cerebral hemorrhage, overexpression syndrome, cerebral vasospasm, long-term restenosis, stent collapse, deformation, and displacement. Therefore, in the clinical implementation of intravascular stent placement, angiography should be performed to clarify the characteristics (location and extent) of the lesion to allow an appropriate stent to be selected. The factors to consider include the type of stent in addition to its diameter and length. Other indicators should be taken into consideration to reduce the risk of complications. In addition, although the use of DESs in the treatment of intracranial atherosclerosis has been shown to be safe and effective, the rigidity of the stent itself is still an important factor leading to surgical failure[21]. At present, the risk of thrombosis in the stent after the implantation of DESs can be increased by double antiplatelet therapy. However, the duration of application depends on the type of stent and the ischemia and bleeding status of the patient. Thrombosis and stent fragmentation that may result from angioplasty and stent implantation can be treated using a corresponding distal protection device[22].
4 ConclusionsWith scientific progress, the materials and methods used to produce stents will improve, leading to safer and more effective stent implantation. These improvements in intravascular stents will dramatically improve the treatment of cerebrovascular diseases. In the future, intravascular stents will become one of the main treatment modalities for cerebrovascular diseases.
AcknowledgementsThis work was financially supported by the Science and Technology Major Project Foundation of Fujian Province (No. 2015YZ0003).
Conflict of interestsAll contributing authors have no conflict of interests.
| [1] | Velat GJ, Hoh BL, Levy EI, Mocco J. Primary intracranial stenting in acute ischemic stroke. Curr Cardiol Rep, 2010, 12(1): 14–19. DOI:10.1007/s11886-009-0069-x |
| [2] | Mei MJ. Characteristics and clinical application of different types of cerebral vascular scaffolds. Chin J Tissue Eng Res, 2014, 18(25): 4057–4061. |
| [3] | Seo WK, Oh K, Suh SI, Seol HY. Intracranial stenting as a rescue therapy in patients with stroke-in-evolution. J Stroke Cerebrovasc Dis, 2016, 25(6): 1411–1416. DOI:10.1016/j.jstrokecerebrovasdis.2016.01.054 |
| [4] | Dotter CT, Judkins MP. Transluminal treatment of arteriosclerotic obstruction:description of a new technic and a preliminary report of its application. Circulation, 1964, 30(5): 654–670. DOI:10.1161/01.CIR.30.5.654 |
| [5] | Fields JD, Liu KC, Barnwell SL, Clark WM, Lutsep HL. Indications and applications of arterial stents for stroke prevention in atherosclerotic intracranial stenosis. Curr Cardiol Rep, 2010, 12(1): 20–28. DOI:10.1007/s11886-009-0070-4 |
| [6] | Taylor RA, Siddiq F, Suri MFK, Martin CO, Hayakawa M, Chaloupka JC. Risk factors for in-stent restenosis after vertebral ostium stenting. J Endovasc Ther, 2008, 15(2): 203–212. DOI:10.1583/07-2175.1 |
| [7] | Hokimoto S, Tabata N, Yamanaga K, Sueta D, Akasaka T, Tsujita K, Sakamoto K, Yamamoto E, Yamamuro M, Izumiya Y, Kaikita K, Kojima S, Matsui K, Ogawa H. Prevalence of coronary macro-and micro-vascular dysfunctions after drug-eluting stent implantation without in-stent restenosis. Int J Cardiol, 2016, 222: 185–194. DOI:10.1016/j.ijcard.2016.07.221 |
| [8] | Mitra AK, Agrawal DK. In stent restenosis:bane of the stent era. J Clin Pathol, 2006, 59(3): 232–239. DOI:10.1136/jcp.2005.025742 |
| [9] | Vajda Z, Güthe T, Perez MA, Kurre W, Schmid E, Bäzner H, Henkes H. Prevention of intracranial in-stent restenoses:predilatation with a drug eluting balloon, followed by the deployment of a self-expanding stent. Cardiovasc Intervent Radiol, 2013, 36(2): 346–352. DOI:10.1007/s00270-012-0450-9 |
| [10] | Zhang ZY, Yang SY. Endovascular stent and its materials science. J Clin Rehabilit Tissue Eng Res, 2011, 15(8): 1471–1474. |
| [11] | Liu DZ, Xi TF. Current situation and development tendency of Interventional medical device & material industry. China Med Dev Inform, 2006, 12(12): 1–14. |
| [12] | Agrawal CM, Haas KF, Leopold DA, Clark HG. Evaluation of poly (L-lactic acid) as a material for intravascular polymeric stents. Biomaterials, 1992, 13(3): 176–182. DOI:10.1016/0142-9612(92)90068-Y |
| [13] | Wang CN, Gao RL, Cheng SJ, Song LF, Zheng JG, Zi ZY, Ruan YM, Meng L. Biocompatibility study of poly-D, L-lactide/glycolic acid coated coronary stent in porcine model. Chin J Cardiol, 2003, 31(7): 528–531. |
| [14] | Liu B, Li SM, Zhang JC, Shi YF. A novel stent coated with antibody against CD34 carried by the degradable polymer reduces restenosis in canine. Chin J Lab Diagn, 2007, 11(3): 369–371. |
| [15] | Zhou YH, Liao JH, Meng HY, Liu SH. Laser microfabrication of endovascular stent. Appl Laser, 2005, 25(3): 161–164, 154. |
| [16] | Gu XZ, Ni ZH. The fabrication of micro-hole stent by laser cutting. J Huazhong Univ Sci Technol (Nat Sci Ed), 2007, 35(S1): 143–146. |
| [17] | Ni ZH, Yi H, Gu XZ. Mechanism and method of drugloading Nano-particles and stent self-assembly. Chin J Mech Eng, 2005, 41(8): 190–194. DOI:10.3901/JME.2005.08.190 |
| [18] | Henry M, Klonaris C, Amor M, Henry I, Tzvetanov K. State of the art:which stent for which lesion in peripheral interventions?. Texas Heart Inst J, 2000, 27(2): 119–126. |
| [19] | Hehrlein C. Promises of biodegradable stents. Catheter Cardiovasc Intervent, 2007, 69(5): 739. DOI:10.1002/(ISSN)1522-726X |
| [20] | Yu ZT, Zhou L, Huang FQ, Yuan SB, Zhang YF, Niu JL, He XJ. R & D and application on Ti alloy capillary material used in vessel intervention. China Med Dev Inform, 2006, 12(12): 15–20. |
| [21] | Vajda Z, Aguilar M, Göhringer T, Horváth-Rizea D, Bäzner H, Henkes H. Treatment of intracranial atherosclerotic disease with a balloon-expandable paclitaxel eluting stent. Clin Neuroradiol, 2012, 22(3): 227–233. DOI:10.1007/s00062-011-0125-y |
| [22] | Janardhan V. Distal embolic protection devices with a variable thickness microguidewire and methods for their use. U. S. Patent 9, 034, 007, May 19, 2015. |



